Advanced Manufacturing of Enantiomerically Enriched Bisphospholane Ligands for Asymmetric Catalysis
Advanced Manufacturing of Enantiomerically Enriched Bisphospholane Ligands for Asymmetric Catalysis
The landscape of asymmetric catalysis is continually evolving, driven by the demand for highly efficient and stereoselective ligand systems that can operate under industrially viable conditions. Patent CN1882600A introduces a groundbreaking methodology for the preparation of enantiomerically enriched bisphospholane ligands, specifically targeting the limitations found in traditional synthetic routes. This technology focuses on the synthesis of compounds of general formula (I), which serve as critical bidentate ligands in various metal-catalyzed transformations. By shifting away from the reliance on hazardous and difficult-to-handle alkyllithium reagents in the early stages of synthesis, this invention offers a robust pathway that enhances both safety profiles and overall process economics. The core innovation lies in the strategic generation of reactive phosphorus intermediates using elemental lithium and dichlorophenylphosphine, which subsequently react with chiral di-leaving group precursors to construct the phospholane ring system with high fidelity.
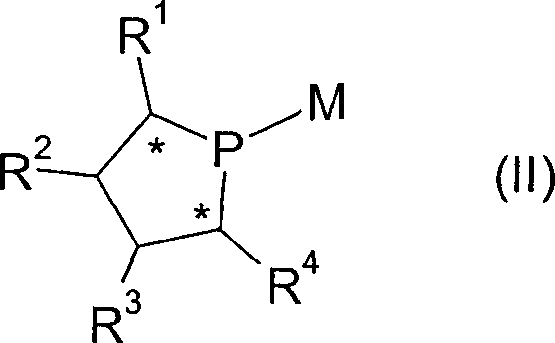
For R&D directors and process chemists, the structural integrity and electronic tunability of these ligands are paramount. The general formula (I) depicted above highlights the versatility of the scaffold, where R1 through R4 can be independently selected from a wide range of alkyl, aryl, and alkoxy groups, allowing for fine-tuning of the steric and electronic environment around the metal center. This adaptability is crucial for optimizing catalytic performance in complex asymmetric syntheses, such as hydrogenation or cross-coupling reactions used in the production of active pharmaceutical ingredients (APIs). The patent explicitly details how controlling the flexibility of the ligand system serves as an inherent limitation of asymmetric induction, thereby ensuring that the chiral information is effectively transferred to the substrate. This level of control is essential for achieving the high enantiomeric excess (ee) values required in modern drug manufacturing, making this technology a valuable asset for any organization focused on high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral phospholanes has relied heavily on the use of strong, pyrophoric bases such as methyllithium or butyllithium to deprotonate phenylphosphine or to generate nucleophilic phosphorus species. As described in the background art of the patent, methods like those in EP528865 or JOC 2003 require the handling of these aggressive reagents at cryogenic temperatures, which imposes severe constraints on industrial scalability. The use of alkyllithium compounds necessitates expensive safety infrastructure, including specialized containment systems and rigorous temperature control protocols, to prevent runaway reactions and ensure operator safety. Furthermore, these conventional routes often suffer from moderate yields and inconsistent reproducibility when scaled up, as seen in comparative examples where yields hovered around 52% to 67%. The reliance on such hazardous materials not only increases the operational expenditure due to safety compliance but also complicates the supply chain logistics, as the storage and transport of these reagents require stringent regulatory adherence.
The Novel Approach
In stark contrast, the novel approach outlined in CN1882600A circumvents these challenges by utilizing elemental lithium metal in conjunction with dichlorophenylphosphine to generate the necessary dilithium phenylphosphine intermediate in situ. This methodological shift eliminates the immediate need for pre-formed alkyllithium bases during the critical ring-closing steps, thereby drastically simplifying the reaction setup and reducing the associated safety risks. The process allows for the reaction to be conducted in common aprotic polar solvents like THF or dimethoxyethane at more manageable temperature ranges, typically between -25°C and +40°C, rather than the extreme cryogenic conditions often demanded by traditional methods. By streamlining the reagent profile and enabling a potential one-pot synthesis strategy, this new route significantly enhances the economic attractiveness of producing these high-value ligands. The result is a process that is not only safer and more environmentally benign but also capable of delivering superior yields, with the patent reporting overall yields exceeding 70% from the starting halophosphines, representing a substantial improvement over prior art.
Mechanistic Insights into Lithium-Mediated Phospholane Ring Formation
The mechanistic pathway of this synthesis is centered on the nucleophilic substitution of chiral di-leaving group compounds by a highly reactive dilithio-phosphorus species. Initially, dichlorophenylphosphine reacts with lithium metal to form dilithium phenylphosphine, a potent nucleophile that attacks the electrophilic carbon centers of the chiral substrate, such as (2S,5S)-2,5-hexanediol bismesylate. This double displacement reaction effectively closes the five-membered phospholane ring while retaining the stereochemical configuration of the starting diol, a critical feature for maintaining the enantiomeric purity of the final ligand. The choice of leaving groups (Y in Formula IV), such as mesylates, tosylates, or halides, plays a pivotal role in the kinetics of this transformation, with mesylates being particularly preferred for their balance of reactivity and stability. The reaction proceeds through a concerted mechanism where the phosphorus atom displaces the leaving groups, forming stable P-C bonds that define the rigid chiral backbone of the ligand.
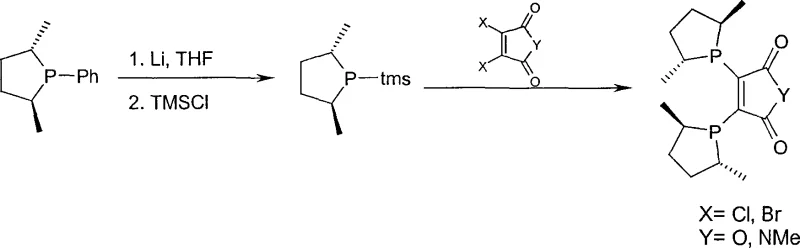
Following the formation of the monomeric phospholane unit (Formula II), the process advances to the coupling stage where two such units are linked via a bridging group (A in Formula III) to create the bidentate ligand system. As illustrated in the reaction scheme above, the phospholane intermediate, often stabilized as a trimethylsilyl derivative or a lithium salt, reacts with di-electrophiles like dichloromaleic anhydride or dibromomaleimide. This step constructs the final C2-symmetric or pseudo-C2-symmetric architecture that is characteristic of high-performance ligands like DuPhos analogues. The ability to perform these transformations in a sequential manner, potentially within a single reactor vessel, minimizes material loss and reduces the generation of waste streams. For quality control teams, understanding this mechanism is vital for monitoring impurity profiles, as side reactions such as over-alkylation or elimination can be mitigated by precise control of stoichiometry and temperature, ensuring the production of high-purity bisphospholane ligands suitable for sensitive catalytic applications.
How to Synthesize Bisphospholane Ligands Efficiently
The synthesis of these advanced chiral ligands requires precise adherence to the optimized conditions described in the patent to ensure maximum yield and stereochemical integrity. The process begins with the careful preparation of the phosphorus nucleophile, followed by its reaction with the chiral diol derivative under inert atmosphere conditions. Detailed standard operating procedures regarding solvent drying, temperature ramping, and workup protocols are essential for replicating the success of the laboratory-scale examples on a larger production floor. The patent emphasizes the importance of selecting the appropriate leaving groups and solvents to facilitate smooth ring closure without compromising the chiral centers.
- Preparation of Dilithium Phenylphosphine by reacting dichlorophenylphosphine with lithium metal in THF.
- Reaction of the dilithio species with chiral di-mesylates (e.g., 2,5-hexanediol bismesylate) to form the phospholane ring.
- Final coupling with bridging agents (Formula III) to generate the bidentate bisphospholane ligand system.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers transformative benefits that extend beyond mere technical performance. By eliminating the dependency on hazardous alkyllithium reagents for the primary ring-forming steps, manufacturers can significantly reduce the capital expenditure associated with specialized safety equipment and hazard mitigation systems. This reduction in operational complexity translates directly into lower manufacturing costs, making the final ligand products more price-competitive in the global market. Furthermore, the use of readily commercially available starting materials, such as dichlorophenylphosphine and common chiral diols, ensures a stable and resilient supply chain that is less susceptible to the volatility often seen with niche organometallic reagents. This stability is crucial for long-term production planning and securing reliable contracts with downstream pharmaceutical clients who demand consistent availability of critical catalyst components.
- Cost Reduction in Manufacturing: The streamlined process design inherently lowers production costs by removing the need for expensive cryogenic infrastructure and the high-cost reagents associated with traditional lithiation methods. The ability to achieve higher yields (>70%) compared to conventional routes means that less raw material is wasted, directly improving the material efficiency and reducing the cost of goods sold (COGS). Additionally, the potential for one-pot synthesis reduces the number of isolation and purification steps, which further cuts down on solvent consumption, energy usage, and labor hours. These cumulative efficiencies allow for a more lean manufacturing model, providing substantial cost savings that can be passed on to customers or reinvested into further process optimization initiatives.
- Enhanced Supply Chain Reliability: Sourcing strategies are greatly improved by the reliance on bulk commodity chemicals rather than specialized, hazardous reagents that may have limited suppliers or long lead times. The robustness of the reaction conditions, which tolerate a broader temperature window and utilize common ether solvents, reduces the risk of batch failures due to minor environmental fluctuations. This reliability ensures that production schedules can be met consistently, reducing lead time for high-purity chiral ligands and preventing bottlenecks in the customer's own manufacturing timelines. For supply chain heads, this means a more predictable inventory management system and the ability to scale production volumes rapidly in response to market demand without facing significant raw material constraints.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, featuring reaction steps that are exothermic but manageable, and workup procedures that are straightforward, such as simple filtration and distillation. This ease of scale-up facilitates the transition from pilot plant to commercial production, supporting the commercial scale-up of complex pharmaceutical intermediates without the need for extensive re-engineering. Moreover, the reduction in hazardous waste generation and the avoidance of toxic by-products align with increasingly stringent environmental regulations, simplifying the permitting process and reducing waste disposal costs. This environmental compliance not only mitigates regulatory risk but also enhances the corporate sustainability profile, which is becoming a key differentiator in B2B negotiations with eco-conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bisphospholane synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN1882600A, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this new bisphospholane synthesis route?
A: The process described in CN1882600A eliminates the need for dangerous alkyllithium bases like methyllithium or butyllithium in the initial steps, significantly improving safety and reducing equipment costs while achieving higher yields (>70%).
Q: Can this process be scaled for industrial production?
A: Yes, the method utilizes readily commercially available starting materials and allows for one-pot synthesis strategies, making it highly suitable for commercial scale-up of complex chiral ligands without expensive safety precautions.
Q: What is the stereochemical outcome of this reaction?
A: The process produces enantiomerically enriched compounds with high fidelity, retaining the chirality from the starting diol derivatives (such as 2S,5S-2,5-hexanediol) through the nucleophilic substitution mechanism.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Bisphospholane Ligands Supplier
The technological advancements detailed in CN1882600A represent a significant leap forward in the field of asymmetric catalysis, offering a pathway to produce high-performance ligands with greater efficiency and safety. NINGBO INNO PHARMCHEM stands at the forefront of adopting such innovative methodologies, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch of bisphospholane ligands meets the exacting standards required for pharmaceutical and fine chemical applications. We understand that the consistency of chiral ligands is critical for the success of downstream catalytic processes, and our state-of-the-art facilities are equipped to handle the nuanced requirements of organophosphorus chemistry.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this novel manufacturing process. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to drive innovation and efficiency in your supply chain, ensuring a steady source of high-quality chiral building blocks for your most demanding synthetic challenges.
