Advanced Synthesis of Fimasartan Key Intermediate for Scalable API Production
Advanced Synthesis of Fimasartan Key Intermediate for Scalable API Production
The pharmaceutical landscape for Angiotensin II Receptor Blockers (ARBs) continues to evolve, driven by the demand for more efficient and cost-effective manufacturing processes for active pharmaceutical ingredients (APIs). Patent CN102666496A introduces a significant technological breakthrough in the synthesis of 2-(2-n-butyl-4-hydroxy-6-methyl-pyrimidin-5-yl)-N,N-dimethylacetamide, a critical advanced intermediate in the production of Fimasartan. This novel methodology addresses longstanding chemoselectivity issues inherent in previous synthetic routes, offering a pathway that significantly suppresses the formation of stubborn urea byproducts while simultaneously enhancing overall production yields. For R&D directors and process chemists, this represents a pivotal shift away from cumbersome purification protocols towards a more streamlined, industrially viable protocol. The strategic implementation of chloroformate-mediated activation or optimized condensation reactions provides a robust framework for ensuring high-purity outputs essential for regulatory compliance in global markets.
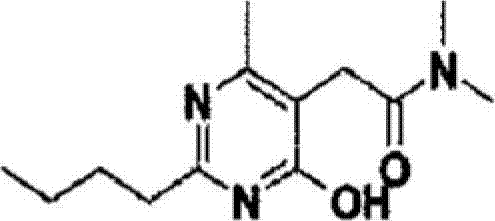
As a reliable pharmaceutical intermediate supplier, understanding the nuances of such process improvements is vital for maintaining competitive advantage in the supply chain. The ability to produce high-purity Fimasartan intermediates without the baggage of difficult-to-remove impurities directly translates to reduced operational expenditures and shorter lead times for downstream API manufacturers. This report delves deep into the mechanistic advantages and commercial implications of adopting this patented technology, providing actionable insights for procurement managers and supply chain heads looking to optimize their sourcing strategies for antihypertensive drug components.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
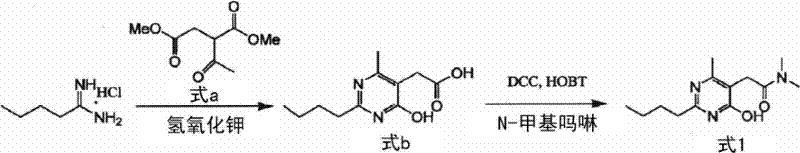
Prior art methodologies, such as those described in Korean Patent No. 10-521980, rely heavily on the condensation of pentanamidine hydrochloride with dimethyl acetylsuccinate in the presence of strong bases like potassium hydroxide. A fundamental flaw in this conventional approach is the inevitable hydrolysis of the terminal methyl ester functionality during the basic reaction conditions, leading to the formation of a carboxylic acid derivative rather than the desired ester. When this acidic intermediate is subsequently coupled using reagents like N-hydroxybenzotriazole (HOBT) and dicyclohexylcarboimide (DCC) to introduce the dimethylamide group, it generates dicyclohexylurea as a stoichiometric byproduct.
This urea byproduct possesses high moisture absorption properties and notoriously poor filtration characteristics, often clogging standard centrifuges and requiring extensive, yield-reducing washing procedures to remove. Furthermore, the reliance on expensive coupling reagents increases the raw material cost profile significantly, while the complex workup procedures extend the batch cycle time, creating bottlenecks in commercial scale-up of complex pharmaceutical intermediates. The accumulation of these inefficiencies makes the conventional route less attractive for high-volume manufacturing where cost reduction in API manufacturing is a primary directive.
The Novel Approach
The innovative process disclosed in CN102666496A circumvents these pitfalls through two distinct yet equally effective strategies. The first method utilizes a pre-functionalized beta-keto ester derivative (Formula 2) reacting directly with pentamidine, avoiding the harsh hydrolysis conditions that plague the older route. Alternatively, the second method employs a mixed anhydride strategy, activating the carboxylic acid precursor (Formula 5) with a haloformate compound (Formula 6) such as ethyl chloroformate. This activation step proceeds under mild conditions, typically between -10°C and 35°C, generating a reactive intermediate that readily undergoes aminolysis with dimethylamine. By bypassing the carbodiimide coupling entirely, this novel approach eliminates the formation of urea byproducts at the source, simplifying the purification process to a straightforward crystallization. This represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing, offering a cleaner, faster, and more economically sustainable pathway to the target molecule.
Mechanistic Insights into Mixed Anhydride Activation and Cyclization
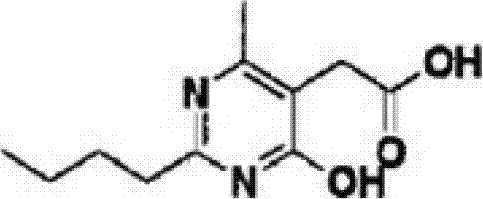
The mechanistic elegance of the second preparation method lies in the generation of a mixed carbonic-carboxylic anhydride intermediate. When 2-(2-n-butyl-4-hydroxy-6-methyl-pyrimidin-5-yl)-acetic acid is treated with ethyl chloroformate in the presence of a tertiary amine base like triethylamine, the carboxylate anion attacks the carbonyl carbon of the chloroformate. This nucleophilic acyl substitution displaces the chloride ion, forming a highly electrophilic mixed anhydride species. This intermediate is significantly more reactive towards nucleophiles than the parent carboxylic acid but avoids the over-activation risks associated with acid chlorides. The subsequent addition of dimethylamine, preferably generated in situ from dimethylamine hydrochloride and triethylamine to ensure controlled release, results in a rapid nucleophilic attack at the carbonyl carbon adjacent to the pyrimidine ring.
Critical to this mechanism is the suppression of side reactions; the use of equimolar amounts of base and chloroformate minimizes the formation of symmetric anhydrides or carbonate byproducts. The reaction temperature is carefully modulated, starting low to control exotherm during activation and rising to reflux (e.g., 50-60°C in dichloromethane) to drive the amidation to completion. From an impurity control perspective, this route is superior because the byproducts—primarily ethanol and triethylamine hydrochloride—are either volatile or water-soluble, allowing for easy removal during the aqueous workup. This contrasts sharply with the solid urea waste of the DCC method, ensuring that the final crude product possesses a much higher purity profile before the final crystallization step, thereby reducing the load on downstream purification units.
In the first preparation method, the mechanism involves a classic Hantzsch-like pyrimidine synthesis where the amidine nitrogen attacks the beta-keto ester functionality. The presence of the dimethylamide side chain on the starting material (Formula 2) ensures that the final product already possesses the required amide functionality, negating the need for a separate amidation step. This convergent synthesis strategy reduces the total number of unit operations, which is a key factor in improving overall process mass intensity (PMI). The choice of solvent, preferably ethanol or methanol, facilitates the solubility of both the organic substrate and the inorganic base, ensuring homogeneous reaction conditions that promote consistent crystal growth and morphology in the final isolation step.
How to Synthesize 2-(2-n-butyl-4-hydroxy-6-methyl-pyrimidin-5-yl)-N,N-dimethylacetamide Efficiently
Implementing this synthesis requires strict adherence to the optimized reaction parameters defined in the patent to maximize yield and minimize impurity carryover. The process begins with the precise activation of the pyrimidine acetic acid derivative, followed by a controlled amidation reaction that leverages the reactivity of mixed anhydrides. Operators must maintain rigorous temperature control during the addition of chloroformates to prevent thermal runaway, and the subsequent reflux period must be sufficient to ensure complete conversion of the intermediate. The workup procedure involves a pH-swing extraction, where basification to pH 9 ensures the product remains in the organic phase while acidic impurities are sequestered in the aqueous layer. Detailed standardized operating procedures for each unit operation are critical for reproducibility.
- Activate 2-(2-n-butyl-4-hydroxy-6-methyl-pyrimidin-5-yl)-acetic acid using ethyl chloroformate and triethylamine in dichloromethane at -10°C to 35°C.
- React the resulting mixed anhydride intermediate with dimethylamine (generated in situ from dimethylamine hydrochloride and triethylamine) under reflux conditions.
- Perform aqueous workup involving basification to pH 9, extraction with dichloromethane, and crystallization using ethyl acetate and n-hexane to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond simple chemistry. The elimination of expensive coupling reagents like DCC and HOBT directly impacts the bill of materials, leading to substantial cost savings in raw material procurement. Furthermore, the removal of the difficult filtration step associated with urea byproducts drastically reduces processing time and equipment occupancy, allowing for higher throughput in existing manufacturing facilities. This efficiency gain translates into a more resilient supply chain capable of meeting fluctuating market demands for antihypertensive medications without the risk of production bottlenecks.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the complete avoidance of carbodiimide coupling agents, which are not only costly but also generate significant solid waste. By switching to chloroformate activation, the process utilizes cheaper, commodity-grade reagents that are widely available from multiple global suppliers, reducing supply risk. Additionally, the simplified workup reduces solvent consumption and energy usage associated with extended drying and filtration cycles, contributing to a lower overall cost of goods sold (COGS) and improved margin potential for the final API.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as ethyl chloroformate and pentamidine salts ensures a stable supply base, mitigating the risk of shortages associated with specialized reagents. The robustness of the reaction conditions, which tolerate a reasonable range of temperatures and solvent grades, means that production is less susceptible to minor variations in raw material quality or environmental conditions. This reliability is crucial for maintaining continuous supply to downstream API manufacturers, ensuring that critical medication pipelines remain uninterrupted.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process is inherently greener due to the reduction in solid waste generation. The absence of dicyclohexylurea waste simplifies effluent treatment and disposal, aligning with increasingly stringent environmental regulations. The scalability is further enhanced by the use of common organic solvents like dichloromethane and ethanol, which are standard in most multipurpose pharmaceutical plants, facilitating easy technology transfer from pilot scale to multi-ton commercial production without the need for specialized equipment modifications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and advantageous effects reported in the patent documentation, providing clarity on process feasibility and quality outcomes. Understanding these details is essential for technical teams evaluating the integration of this route into their existing manufacturing portfolios.
Q: Why does the conventional DCC/HOBT coupling method pose purification challenges?
A: Conventional methods utilizing dicyclohexylcarboimide (DCC) generate dicyclohexylurea as a stoichiometric byproduct. This urea derivative exhibits high moisture absorption and poor solubility characteristics, making it extremely difficult to remove via standard centrifugal filtration, thereby compromising overall process efficiency and purity profiles.
Q: What are the critical reaction parameters for the chloroformate activation step?
A: The activation step requires precise temperature control between -10°C and 35°C to prevent side reactions. The molar ratio of the carboxylic acid to the chloroformate activating agent is optimally maintained between 1:2 and 1:3, with triethylamine serving as the base to scavenge generated hydrogen chloride and drive the formation of the mixed anhydride intermediate.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the described methodology eliminates the need for expensive coupling reagents and complex filtration steps associated with urea removal. The use of commercially available chloroformates and standard organic solvents like dichloromethane facilitates straightforward scale-up from kilogram to multi-ton production capacities while maintaining robust impurity control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(2-n-butyl-4-hydroxy-6-methyl-pyrimidin-5-yl)-N,N-dimethylacetamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient intermediate synthesis in the broader context of pharmaceutical development. Our team of expert process chemists has extensively evaluated the technology described in CN102666496A and confirmed its potential for delivering high-quality intermediates with superior purity profiles. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch meets the exacting standards required for global regulatory submissions.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic impact of switching to this urea-free methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a collaborative approach to advancing the availability of high-quality antihypertensive therapies.
