Advanced Low-Temperature Synthesis of Aminoimidazole Derivatives for Commercial Pharmaceutical Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for critical oncology intermediates, particularly those leading to alkylating agents like dacarbazine and temozolomide. Patent CN101663266B introduces a transformative methodology for producing N-(2-amino-1,2-dicyanovinyl)imidates and their subsequent cyclization into aminoimidazole derivatives. This technology addresses long-standing challenges in the synthesis of 5-amino-1H-imidazole-4-carbonitrile (AICN) and 5-amino-1H-imidazole-4-carboxamide (AICA), which are pivotal precursors in the manufacture of hepatoprotective and anticancer medications. By leveraging strong acid catalysis under mild thermal conditions, this process achieves superior yields while mitigating the decomposition issues prevalent in traditional high-temperature protocols. The strategic implementation of aprotic solvents further refines the impurity profile, ensuring that the final active pharmaceutical ingredient (API) precursors meet stringent global quality standards.
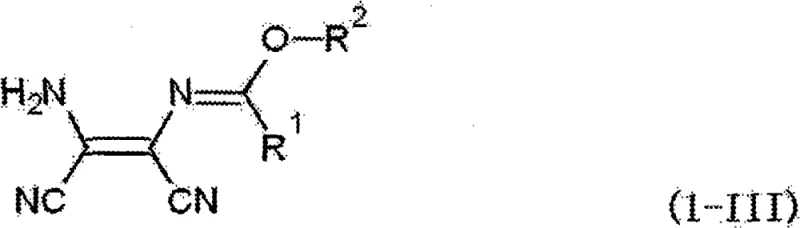
For procurement specialists and supply chain directors, the adoption of this low-temperature synthesis route represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. Traditional methods often require harsh reflux conditions in solvents like dioxane, which not only consume excessive energy but also accelerate the degradation of sensitive starting materials like diaminomaleonitrile (DAMN). The new approach operates efficiently at temperatures ranging from 30°C to 50°C, drastically lowering energy consumption and enhancing operational safety. Furthermore, the compatibility of this method with scalable solvent systems like tetrahydrofuran (THF) simplifies the transition from laboratory benchtop to commercial production, ensuring a reliable aminoimidazole intermediate supplier can maintain consistent delivery schedules without the bottlenecks associated with complex purification steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-(2-amino-1,2-dicyanovinyl)imidates has been plagued by inefficiencies stemming from high-temperature reaction requirements. Conventional protocols typically involve heating diaminomaleonitrile with triethyl orthoformate in dioxane at reflux temperatures. Under these vigorous thermal conditions, the starting material, DAMN, is prone to decomposition and polymerization, leading to the formation of intractable tars and unidentified impurities. Additionally, the product itself, the imidate ester, exhibits thermal instability, meaning that even if formed, a portion inevitably degrades before isolation. This results in suboptimal yields, often necessitating extensive and costly downstream purification processes to remove polymeric byproducts. The reliance on gaseous ammonia for subsequent amidination steps in older methods further complicates the engineering landscape, as the product tends to precipitate at gas inlet points, causing frequent blockages in reactor piping and halting continuous production lines.
The Novel Approach
The innovative process detailed in the patent data circumvents these thermal limitations by employing strong acid catalysts such as methanesulfonic acid, p-toluenesulfonic acid, or trifluoroacetic acid at significantly lower temperatures. By conducting the reaction in aprotic organic solvents like tetrahydrofuran, the method stabilizes the reactive intermediates and suppresses the polymerization pathways that degrade DAMN. This shift allows for rapid conversion times, often completing within one hour, while maintaining high product integrity. Moreover, the adaptation of using aqueous ammonia within an ether solvent system for the amidination step eliminates the engineering hazards of gaseous ammonia handling. This liquid-phase modification ensures homogeneous mixing and prevents the clogging issues seen in legacy systems, thereby facilitating a smoother, more reliable workflow for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Acid-Catalyzed Cyclization and Solvent Effects
The core mechanistic advantage of this synthesis lies in the precise activation of the orthoformate reagent by strong Brønsted acids. In the absence of a potent acid catalyst, the nucleophilic attack of the amino groups in DAMN on the orthoformate carbon is sluggish and requires thermal energy that compromises stability. The introduction of catalytic amounts of strong acids protonates the orthoformate, generating a highly electrophilic species that reacts rapidly with DAMN even at ambient or slightly elevated temperatures (30°C to 50°C). This kinetic acceleration is crucial because it outpaces the competing decomposition reactions of the starting material. The choice of solvent plays an equally pivotal role; aprotic solvents like THF do not participate in hydrogen bonding that could stabilize unwanted transition states leading to byproducts. Instead, they provide a medium where the ionic intermediates involved in the cyclization are sufficiently solvated to react efficiently without promoting hydrolysis or alcoholysis side reactions that are common in protic media.
Impurity control is inherently built into this mechanistic framework through the suppression of thermal degradation pathways. In traditional alcohol-based systems, the presence of protic solvents can lead to transesterification or the formation of stable alkoxy-imidate byproducts that are difficult to separate from the desired nitrile or amide final products. By utilizing an aprotic environment throughout the sequence—from imidate formation to amidination and finally cyclization—the process minimizes the generation of these structural analogs. The use of aqueous ammonia in the amidination step specifically targets the formation of the unsubstituted formamidine (AMD-H), which is the optimal precursor for cyclization into AICN. This specificity ensures that the subsequent ring-closure reaction proceeds with high regioselectivity, yielding the 5-amino-1H-imidazole core with minimal contamination from isomeric impurities or polymeric residues.
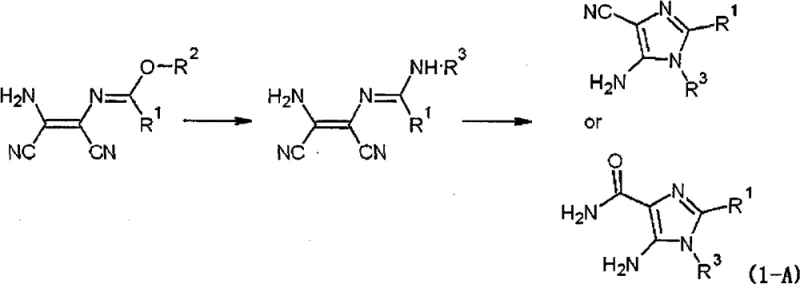
How to Synthesize N-(2-amino-1,2-dicyanovinyl)imidate Efficiently
The practical execution of this synthesis involves a carefully controlled sequence of reagent additions and temperature management to maximize yield and safety. Operators begin by suspending diaminomaleonitrile in tetrahydrofuran and adding the trialkyl orthoformate, followed by the slow introduction of the strong acid catalyst to initiate the exothermic formation of the imidate. Once the imidate slurry is formed, it is treated directly with aqueous ammonia without isolation, leveraging the one-pot capability to reduce solvent waste and handling time. The detailed standardized synthesis steps see the guide below for specific molar ratios and safety protocols.
- React diaminomaleonitrile (DAMN) with trialkyl orthoformate in the presence of a strong acid catalyst (e.g., methanesulfonic acid) in an aprotic solvent like tetrahydrofuran at 30-50°C to form N-(2-amino-1,2-dicyanovinyl)imidate.
- Convert the imidate intermediate to N-(2-amino-1,2-dicyanovinyl)formamidine by reacting with aqueous ammonia in an ether solvent, avoiding gaseous ammonia to prevent equipment clogging.
- Perform cyclization and hydrolysis in an alkaline aqueous solution within the aprotic solvent system to yield 5-amino-1H-imidazole-4-carbonitrile (AICN) or 5-amino-1H-imidazole-4-carboxamide (AICA) with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this manufacturing technology offers substantial cost savings and supply chain resilience for buyers of oncology intermediates. The elimination of high-temperature reflux conditions translates directly into reduced utility costs and lower wear-and-tear on reactor vessels, extending the lifespan of capital equipment. Furthermore, the ability to perform the synthesis in a single solvent system (THF) across multiple steps simplifies solvent recovery and recycling operations, significantly reducing the environmental footprint and disposal costs associated with mixed solvent waste streams. These efficiencies allow manufacturers to offer more competitive pricing structures while maintaining healthy margins, making it an attractive option for long-term supply agreements.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive and hazardous high-temperature operations, which reduces energy consumption and lowers the risk of thermal runaway incidents. By avoiding the use of gaseous ammonia, the requirement for specialized pressure-rated equipment and complex gas scrubbing systems is removed, leading to significant capital expenditure savings. Additionally, the high yield and purity achieved reduce the volume of raw materials required per kilogram of final product, optimizing the overall cost of goods sold.
- Enhanced Supply Chain Reliability: The robustness of the low-temperature protocol ensures consistent batch-to-batch quality, minimizing the risk of production failures that can disrupt supply timelines. The use of readily available and stable reagents like aqueous ammonia and common organic solvents reduces dependency on specialized or volatile raw material markets. This stability allows suppliers to maintain higher inventory levels of finished intermediates, ensuring that downstream API manufacturers can rely on just-in-time delivery without fear of stockouts.
- Scalability and Environmental Compliance: The one-pot nature of the synthesis minimizes the number of unit operations, making it easier to scale from pilot plant to multi-ton commercial production without re-engineering the entire process flow. The reduction in solvent switching and intermediate isolation steps decreases the generation of hazardous waste, facilitating compliance with increasingly strict environmental regulations. This green chemistry approach not only lowers regulatory risk but also aligns with the sustainability goals of major pharmaceutical companies seeking eco-friendly supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on yield expectations, solvent choices, and operational parameters. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing manufacturing portfolios.
Q: Why is tetrahydrofuran (THF) preferred over alcohol solvents for this synthesis?
A: THF is an aprotic solvent that significantly reduces side reactions during the cyclization step compared to protic solvents like alcohols. It also facilitates the one-pot synthesis from diaminomaleonitrile, improving overall yield and purity by minimizing byproduct formation such as dicyanoimidazole derivatives.
Q: What are the advantages of using aqueous ammonia instead of gaseous ammonia?
A: Using aqueous ammonia in an ether solvent prevents the precipitation of the product at the gas inlet, which often clogs industrial reaction piping when gaseous ammonia is used. This modification ensures smoother operation, better mixing, and higher reproducibility in large-scale manufacturing.
Q: How does the low-temperature condition affect the stability of the intermediates?
A: Maintaining reaction temperatures between 30°C and 50°C prevents the thermal decomposition and polymerization of diaminomaleonitrile (DAMN) and the resulting imidate intermediates. This stability is crucial for achieving high yields, as high-temperature conditions typically lead to significant degradation and impurity generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-(2-amino-1,2-dicyanovinyl)imidate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving oncology therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated low-temperature chemistry described in CN101663266B can be successfully translated to industrial volumes. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of aminoimidazole derivative meets the exacting standards required for GMP API synthesis. Our commitment to process excellence means that we can deliver materials with consistent impurity profiles, supporting your regulatory filings and clinical trial timelines with confidence.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this low-temperature protocol for your supply chain. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your production needs, ensuring a seamless partnership that drives value and efficiency in your pharmaceutical manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
