Optimizing N,N'-Carbonyldiazoles Production: A Technical Breakthrough in Phosgene Chemistry
Introduction to Advanced N,N'-Carbonyldiazoles Manufacturing
The chemical industry constantly seeks robust methodologies for synthesizing critical intermediates, particularly those involving hazardous reagents like phosgene. Patent CN1918130A introduces a transformative approach for the preparation of N,N'-carbonyldiazoles, a class of compounds essential for peptide coupling and heterocyclic synthesis in pharmaceutical manufacturing. This innovation addresses long-standing challenges associated with the phosgenation of azoles, specifically the management of byproduct precipitation and reaction homogeneity. By shifting from traditional aromatic solvents to a carefully selected range of polar solvents including ethers, ketones, and chlorinated aliphatics, the process fundamentally alters the physical chemistry of the reaction mixture. The core breakthrough lies in the ability to maintain high reactant concentrations while ensuring that the azole hydrochloride byproduct remains a stirrable, crystalline solid rather than a problematic viscous mass. This technical advancement not only enhances safety profiles by reducing the risk of equipment fouling but also significantly improves the economic viability of large-scale production through superior space-time yields.
For R&D directors and process chemists, the implications of this patent extend beyond simple yield improvements; it represents a paradigm shift in how phosgene reactions are engineered for scalability. The method described in CN1918130A utilizes a precise metering strategy where the azole and phosgene are introduced simultaneously into the reaction vessel. This controlled addition, coupled with the specific solvent environment, prevents the localized supersaturation that typically leads to the formation of sticky, unmanageable sludge in conventional processes. Consequently, manufacturers can operate at reactant concentrations exceeding 40wt%, a substantial increase over the 28-33wt% limits observed in prior art methods utilizing aromatic solvents. This capability allows for significant reductions in solvent usage and reactor volume requirements, directly impacting the capital expenditure and operational efficiency of production facilities dedicated to high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N,N'-carbonyldiazoles has been plagued by significant processing difficulties, primarily stemming from the physical properties of the reaction byproducts. Traditional methods, such as those described in DE-A-198 33 913, rely heavily on aromatic solvents like benzene, toluene, or xylene, often requiring rigorous pre-drying steps via partial distillation. Despite these precautions, a critical failure mode persists: the formation of azole hydrochloride salts as viscous, tacky masses during the reaction. This phenomenon occurs because the solubility characteristics of the salt in aromatic media promote oiling out or the formation of amorphous aggregates rather than defined crystals. These sticky precipitates adhere tenaciously to reactor walls and agitator blades, creating severe mixing inefficiencies that can halt production entirely. In extreme cases, the hardening of these masses into solid spheres poses a direct mechanical threat to internal reactor components, including impellers and dip tubes, leading to costly downtime and maintenance interventions.
Furthermore, the reliance on aromatic solvents imposes strict limitations on reactant loading to mitigate these precipitation issues. To maintain any semblance of stirrability, operators are forced to dilute the reaction mixture, keeping azole concentrations typically between 2wt% and 12wt% in older methods, or marginally higher in slightly improved variations. This low concentration regime drastically reduces the space-time yield of the process, necessitating larger reactor volumes to achieve the same output tonnage. Additionally, conventional processes often exhibit high sensitivity to excess phosgene, which can lead to undesirable discoloration of the final product, thereby complicating downstream purification steps. The combination of poor mixing, low concentration limits, and product quality risks renders these legacy methods economically suboptimal for modern, high-volume commercial manufacturing environments where efficiency and consistency are paramount.
The Novel Approach
The methodology disclosed in CN1918130A offers a definitive solution to these entrenched problems by re-engineering the solvent system and addition protocol. Instead of aromatic hydrocarbons, the invention mandates the use of polar solvents selected from ethers (such as THF, MTBE, or dioxane), ketones (like acetone or methyl ethyl ketone), or chlorinated aliphatic solvents (such as methylene chloride). These solvents possess distinct solvation properties that favor the nucleation and growth of the azole hydrochloride byproduct as discrete, free-flowing crystals. When combined with a simultaneous metering technique—where the azole and phosgene are added in parallel at a controlled molar ratio—the reaction environment remains homogeneous enough to prevent the formation of viscous agglomerates. This ensures that the precipitate remains easily stirrable throughout the entire course of the reaction, even at near-completion stages where byproduct accumulation is highest.
This novel approach unlocks the ability to operate at significantly higher reactant concentrations, consistently achieving levels greater than 40wt% without compromising mixing efficiency. The practical result is a dramatic intensification of the process, allowing manufacturers to produce more product per unit volume of reactor space. Moreover, the crystalline nature of the byproduct facilitates rapid and efficient filtration, often at elevated temperatures, which streamlines the workup procedure. The process also demonstrates remarkable robustness against over-phosgenation; unlike prior art where excess reagent causes discoloration, this system tolerates slight deviations without affecting the color or consistency of the precipitate. This resilience simplifies process control requirements and reduces the risk of batch rejection due to minor operational variances, making it an ideal candidate for reliable commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Polar Solvent-Mediated Phosgenation
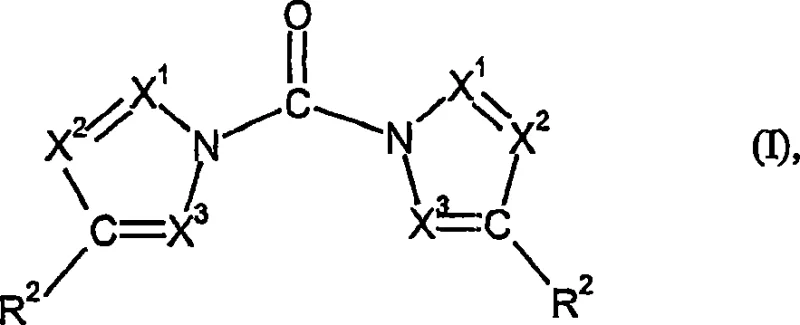
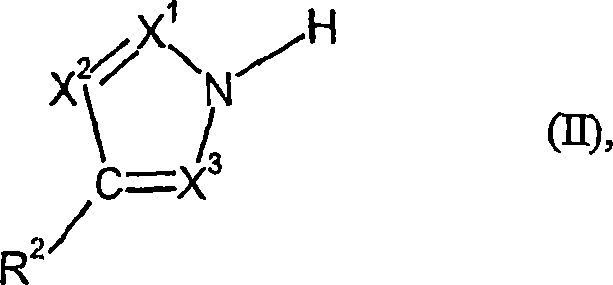
The success of this synthesis route hinges on the intricate interplay between solvent polarity, supersaturation dynamics, and crystal growth kinetics. In the context of reacting an azole of general formula (II) with phosgene to form the N,N'-carbonyldiazole of general formula (I), the choice of solvent dictates the solubility curve of the generated hydrochloride salt. Polar aprotic solvents like tetrahydrofuran (THF) or acetone provide a medium where the ionic character of the azole hydrochloride is sufficiently stabilized to prevent immediate precipitation as an oil, yet the solubility limit is low enough to drive crystallization. The simultaneous addition of reactants ensures that the local concentration of phosgene never vastly exceeds the stoichiometric requirement relative to the available azole, preventing the formation of unstable intermediates that could degrade or polymerize. This controlled environment promotes the formation of a stable crystal lattice for the byproduct, which is crucial for maintaining low viscosity in the slurry.
From an impurity control perspective, the mechanism inherently suppresses side reactions that lead to colored impurities. In conventional aromatic systems, the sticky nature of the precipitate can trap unreacted phosgene or reactive intermediates, leading to prolonged exposure and subsequent degradation pathways that manifest as discoloration. In contrast, the free-flowing crystalline precipitate in the polar solvent system allows for efficient mass transfer and easy separation. The byproduct can be filtered off cleanly at temperatures ranging from 20°C to 100°C, leaving the desired N,N'-carbonyldiazole in the mother liquor. Subsequent cooling of this filtrate induces the crystallization of the target product with high purity, typically exceeding 95% without the need for extensive recrystallization. This mechanistic clarity provides R&D teams with a robust framework for troubleshooting and optimizing the process for specific azole derivatives, ensuring consistent quality across different batches.
How to Synthesize N,N'-Carbonyldiazoles Efficiently
Implementing this synthesis route requires strict adherence to the specified parameters regarding solvent dryness and metering rates to replicate the patented benefits. The process begins with the selection of a suitable polar solvent with a water content strictly controlled to below 0.5wt%, preferably lower, to prevent hydrolysis of the phosgene or the product. The reaction is typically conducted at moderate temperatures between 40°C and 80°C, balancing reaction kinetics with thermal stability. A key operational detail involves the initial charge and subsequent metering: a portion of the solvent is heated, and then the azole and phosgene streams are introduced simultaneously. The molar ratio during this addition phase is critical, maintained between 0.17 and 0.34 moles of phosgene per mole of azole to ensure the correct stoichiometry for the formation of the carbonyl bridge without excessive accumulation of free phosgene. Detailed standardized synthetic steps see the guide below.
- Prepare a polar solvent system (ethers, ketones, or chlorinated aliphatics) with water content below 0.5wt% and heat to 40-80°C.
- Simultaneously meter the azole reactant and phosgene into the solvent, maintaining a molar ratio of 0.17 to 0.34 moles of phosgene per mole of azole during addition.
- Filter the resulting crystalline azole hydrochloride precipitate at reaction temperature, then cool the mother liquor to isolate the pure N,N'-carbonyldiazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the technology described in CN1918130A translates into tangible operational efficiencies and cost optimizations. The primary driver of value is the substantial increase in space-time yield afforded by the ability to run reactions at concentrations exceeding 40wt%. By processing more mass in the same reactor volume, facilities can significantly increase throughput without the need for capital-intensive expansion of tank farms or reaction vessels. This intensification directly lowers the fixed cost allocation per kilogram of product. Furthermore, the elimination of sticky precipitates removes a major bottleneck in batch cycle times; filtration and cleaning operations are drastically accelerated because the crystalline byproduct does not foul equipment. This reliability ensures more predictable production schedules and reduces the risk of unplanned downtime associated with mechanical failures caused by hardened sludge.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from multiple vectors of efficiency. Firstly, the higher reactant concentration means a significant reduction in the volume of solvent required per unit of product, which lowers both raw material costs and the energy burden associated with solvent recovery and distillation. Secondly, the ability to recycle the azole hydrochloride byproduct back into the free azole creates a closed-loop material flow that effectively doubles the theoretical yield based on the purchased azole starting material. This recycling capability transforms a waste stream into a valuable asset, drastically reducing the net consumption of expensive heterocyclic starting materials. Finally, the reduced sensitivity to phosgene excess minimizes batch losses due to off-spec color or purity, ensuring a higher first-pass yield and reducing the costs associated with reprocessing or disposal.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by process variability and equipment maintenance issues, both of which are mitigated by this technology. The formation of non-sticky, crystalline byproducts ensures that agitation systems remain functional throughout the reaction, preventing the catastrophic batch failures that can occur when stirrers seize in viscous masses. This mechanical reliability translates to consistent batch cycle times, allowing supply chain planners to forecast delivery dates with greater confidence. Additionally, the use of common industrial solvents like THF or acetone, rather than specialized aromatic mixtures requiring extensive pre-drying, simplifies raw material sourcing. The robustness of the process against minor fluctuations in phosgene dosing further stabilizes production, making the supply of high-purity intermediates more resilient to operational variances.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies heat transfer and mixing challenges, but this method is inherently designed for scalability. The low viscosity of the reaction slurry ensures efficient heat removal, which is critical for exothermic phosgenation reactions, thereby enhancing safety profiles at large scales. From an environmental standpoint, the reduction in solvent usage directly correlates to a lower carbon footprint and reduced waste generation. The efficient filtration and potential for solvent recycling minimize the volume of liquid waste requiring treatment. Moreover, the ability to convert the hydrochloride byproduct back to the starting material aligns with green chemistry principles by maximizing atom economy. These factors collectively simplify regulatory compliance and environmental reporting, positioning the manufacturer as a sustainable partner in the global supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phosgenation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for their specific product portfolios. The answers highlight the distinct advantages over legacy methods, focusing on yield, purity, and operational safety.
Q: Why does this process prevent the formation of sticky precipitates?
A: By using specific polar solvents like THF or acetone and strictly controlling the phosgene metering rate, the azole hydrochloride byproduct forms as a free-flowing crystalline solid rather than a viscous mass.
Q: What are the advantages regarding reactant concentration?
A: This method allows for reactant concentrations greater than 40wt%, significantly higher than the 28-33wt% limit of conventional aromatic solvent methods, improving space-time yield.
Q: Can the azole hydrochloride byproduct be recycled?
A: Yes, the azole hydrochloride can be converted back to the free azole using alkali metal compounds, effectively doubling the theoretical yield based on the starting azole material.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-Carbonyldiazoles Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated controls required for this phosgenation process are meticulously managed. We understand that high-purity N,N'-carbonyldiazoles demand stringent purity specifications and rigorous QC labs to verify the absence of residual phosgene and colored impurities. Our commitment to quality assurance means that every batch is subjected to comprehensive analytical testing, guaranteeing that the material meets the exacting standards required for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to leverage this advanced manufacturing technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits of switching to this high-efficiency process for your projects. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us demonstrate how our mastery of complex organic synthesis can drive value, reliability, and quality to your operations.
