Advanced Aqueous Synthesis of Macrocyclic Chelating Agents for High-Purity MRI Contrast Intermediates
Advanced Aqueous Synthesis of Macrocyclic Chelating Agents for High-Purity MRI Contrast Intermediates
The pharmaceutical and diagnostic imaging sectors are constantly seeking more efficient and environmentally sustainable pathways for producing critical active pharmaceutical ingredients and their precursors. Patent CN1166649C introduces a groundbreaking methodology for the preparation of macrocyclic chelating agents complexed with paramagnetic metal ions, specifically targeting the synthesis of non-ionic contrast agents used in Magnetic Resonance Imaging (MRI). This technology represents a significant departure from traditional organic solvent-heavy processes, offering a robust, aqueous-based alternative that addresses long-standing issues regarding toxicity, yield consistency, and operational complexity. For R&D directors and procurement specialists alike, understanding the nuances of this patent is crucial for securing a reliable MRI contrast agent intermediate supplier capable of delivering high-purity materials while adhering to stringent environmental regulations. The innovation lies not just in the chemical transformation but in the holistic redesign of the process flow to minimize hazardous waste and maximize throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
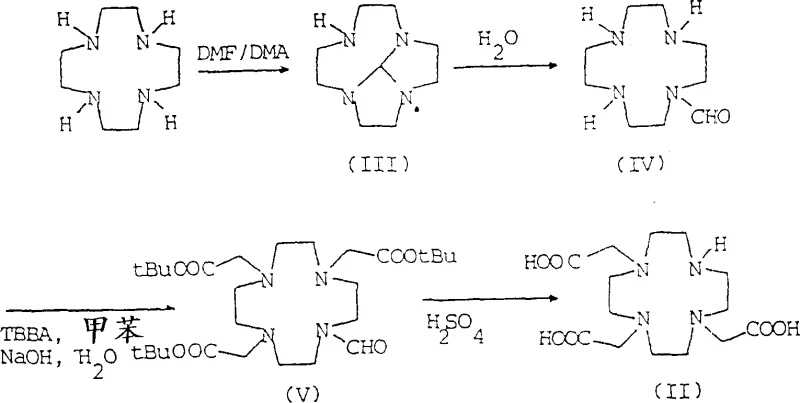
Historically, the synthesis of macrocyclic chelates like Gadoteridol and Gadobutrol has relied on strategies that involve multiple protection and deprotection steps using hazardous organic solvents. As illustrated in the prior art, conventional routes often utilize reagents such as dimethylformamide dimethyl acetal and tert-butyl bromoacetate, which pose significant health and safety risks to personnel and require elaborate waste management systems. Furthermore, these traditional methods are plagued by operational inefficiencies; while they may show promise on a laboratory scale, they frequently suffer from drastic yield reductions when scaled up to industrial volumes. For instance, literature indicates that certain established protocols see overall yields drop significantly, sometimes hovering around 42%, due to the complexity of isolating intermediates and removing salts and solvents at each stage. This inconsistency creates substantial supply chain volatility, making it difficult for procurement managers to forecast costs and ensure continuous availability of high-purity pharmaceutical intermediates.
The Novel Approach
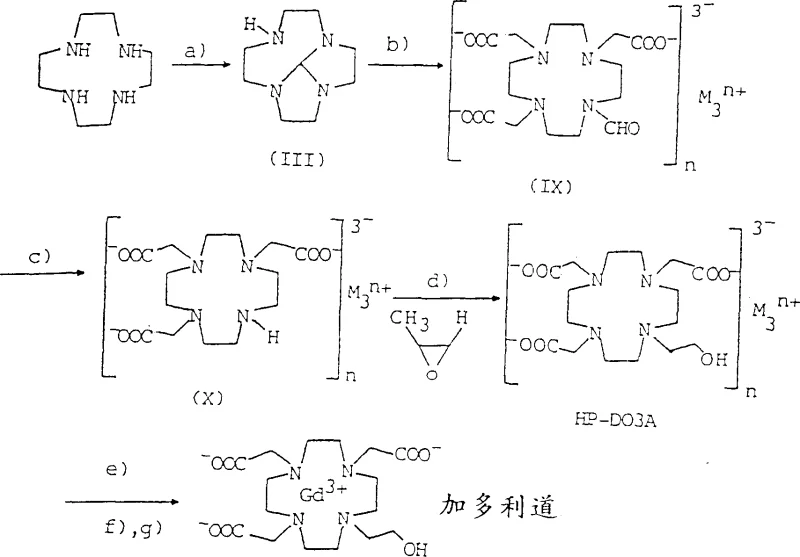
In stark contrast, the methodology disclosed in Patent CN1166649C revolutionizes the production landscape by shifting the reaction medium primarily to water. This novel approach utilizes triethyl orthoformate as a safer, less toxic alternative to traditional protecting group reagents, effectively mitigating the generation of harmful, non-condensable gaseous by-products. The process is designed to be highly selective yet operationally simple, allowing for the direct carboxymethylation and subsequent hydrolysis of intermediates in an aqueous environment without the need for tedious isolation steps between reactions. By maintaining specific pH ranges and temperatures, the reaction progress can be monitored reliably using basic parameters like time and temperature, eliminating the need for constant gas chromatography analysis. This streamlining not only enhances safety but also drastically simplifies the workflow, making it an ideal candidate for cost reduction in pharmaceutical intermediates manufacturing where efficiency and safety are paramount.
Mechanistic Insights into Aqueous Phase Carboxymethylation and Hydrolysis
The core of this technological advancement lies in the precise control of the aqueous reaction environment during the carboxymethylation and hydrolysis phases. The process begins with the formation of a protected intermediate, 5H,9bH-2a,4a,7,9a-octahydrotetraazacycloocta[cd]pentalene, using triethyl orthoformate in the presence of a carboxylic acid catalyst. Following this, the critical carboxymethylation step occurs in water at a controlled pH between 9.5 and 12.5, utilizing haloacetic acid to introduce the acetate arms necessary for metal chelation. Unlike organic phase reactions, this aqueous step allows for excellent heat dissipation and mixing, reducing the risk of thermal runaways. Subsequently, the protecting group is removed via hydrolysis under strongly alkaline conditions (pH > 12.5) at elevated temperatures, yielding the triacetate salt directly in solution. This seamless transition from protection to functionalization without intermediate isolation is a masterclass in process chemistry, ensuring that impurities are minimized and the structural integrity of the macrocyclic ring is preserved throughout the synthesis.
Furthermore, the mechanism ensures high purity by leveraging the solubility differences between the desired product and potential by-products. The subsequent alkylation with epoxides, such as propylene oxide for Gadoteridol, proceeds efficiently in the same aqueous medium, followed by complexation with paramagnetic metal ions like gadolinium. The final purification stages involve sophisticated diafiltration and ion exchange techniques tailored to remove specific ionic impurities while retaining the high molecular weight chelate. This multi-stage purification strategy is essential for meeting the rigorous quality standards required for injectable diagnostic agents. For R&D teams, understanding these mechanistic details provides confidence in the reproducibility of the process, ensuring that every batch meets the stringent specifications necessary for clinical applications. The ability to perform these complex transformations in water rather than volatile organic compounds represents a paradigm shift towards greener chemistry in the fine chemical industry.
How to Synthesize Gadoteridol Efficiently
The synthesis of Gadoteridol, a critical MRI contrast agent, exemplifies the practical application of this patented technology. The process involves a sequence of well-defined steps starting from 1,4,7,10-tetraazacyclododecane, moving through protection, carboxymethylation, hydrolysis, alkylation with propylene oxide, and finally complexation with gadolinium. Each step is optimized for aqueous conditions, ensuring safety and scalability. The detailed standardized synthesis steps below outline the precise conditions required to achieve high yields and purity, serving as a foundational guide for process engineers looking to implement this technology.
- React 1,4,7,10-tetraazacyclododecane with triethyl orthoformate in the presence of an acid catalyst to form the protected intermediate.
- Perform carboxymethylation in water at pH 9.5-12.5 using haloacetic acid, followed by hydrolysis at pH > 12.5 to remove the protecting group.
- Alkylate the resulting triacetate salt with an epoxide in water, complex with a paramagnetic metal salt, and purify via diafiltration and ion exchange.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous synthesis technology offers compelling strategic advantages that extend beyond mere chemical efficiency. The elimination of toxic organic solvents and hazardous reagents translates directly into reduced operational risks and lower costs associated with safety compliance and waste disposal. By simplifying the process flow and removing the need for complex intermediate isolations, manufacturers can achieve faster turnaround times and more predictable production schedules. This reliability is crucial for maintaining a steady supply of high-purity pharmaceutical intermediates in a market where demand for diagnostic imaging agents continues to grow. Moreover, the robustness of the process on an industrial scale ensures that supply continuity is maintained even during periods of high demand, mitigating the risks of shortages that can disrupt downstream pharmaceutical production.
- Cost Reduction in Manufacturing: The shift to an aqueous-based process significantly lowers the cost burden associated with solvent procurement, recovery, and disposal. By avoiding expensive and toxic reagents like tert-butyl bromoacetate, the raw material costs are optimized, and the need for specialized containment equipment is reduced. Additionally, the high selectivity of the reaction minimizes the formation of by-products, leading to higher overall yields and less material waste. These factors combine to create a more economically viable production model, allowing for competitive pricing without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: The simplified operational parameters of this method, such as monitoring reaction progress via time and temperature rather than complex analytics, reduce the likelihood of batch failures. This operational stability ensures a consistent output of high-quality intermediates, fostering trust between suppliers and pharmaceutical clients. The ability to scale the process from laboratory to commercial production without significant yield loss further strengthens the supply chain, providing a dependable source of critical materials for the manufacture of life-saving diagnostic agents.
- Scalability and Environmental Compliance: Designed with industrial scalability in mind, this process aligns perfectly with modern environmental regulations by minimizing the release of volatile organic compounds and hazardous waste. The use of water as the primary solvent simplifies effluent treatment and reduces the environmental footprint of the manufacturing facility. This compliance not only avoids potential regulatory fines but also enhances the corporate social responsibility profile of the supply chain, appealing to increasingly eco-conscious stakeholders and partners in the global pharmaceutical market.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical specifications and commercial viability of the macrocyclic chelating agent synthesis described in Patent CN1166649C. These answers are derived directly from the patent's technical disclosures and experimental data, providing clarity on how this innovative method compares to existing technologies. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this process into their current manufacturing workflows or sourcing strategies.
Q: What are the key safety advantages of the new aqueous synthesis method?
A: The new method eliminates the use of highly toxic reagents such as tert-butyl bromoacetate and dimethylformamide dimethyl acetal, replacing them with safer alternatives like triethyl orthoformate and conducting reactions primarily in water.
Q: How does the yield of the novel process compare to conventional methods?
A: While conventional methods often suffer from yield drops during scale-up (e.g., dropping to 42%), the patented aqueous process demonstrates robust yields equal to or higher than 80% for high-quality end products like Gadoteridol.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for reproducibility on an industrial scale, utilizing simpler operational parameters like temperature and time monitoring without the need for complex gas chromatography checks during reactant addition.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gadoteridol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of advanced diagnostic agents. Our expertise in process chemistry allows us to leverage innovations like the aqueous synthesis method described in Patent CN1166649C to deliver superior products. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-volume supplies without compromise. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Gadoteridol or Gadobutrol intermediate meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through advanced chemical solutions. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Contact us today to request specific COA data and route feasibility assessments, and discover how our commitment to innovation and quality can support your long-term business goals in the competitive landscape of pharmaceutical intermediates.
