Advanced Solvent-Free Synthesis of Macrocyclic Intermediates for Commercial MRI Contrast Agent Production
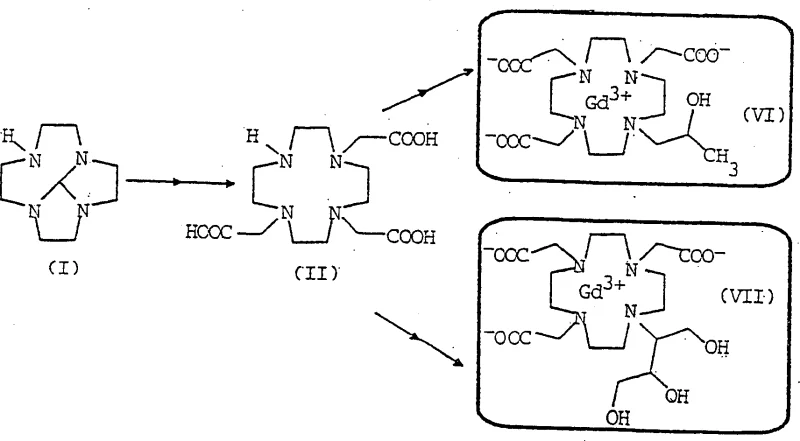
The pharmaceutical industry's demand for high-purity diagnostic agents continues to drive innovation in intermediate synthesis, particularly for magnetic resonance imaging (MRI) contrast media. Patent CN1117094C discloses a groundbreaking process for the preparation of 5H,9bH-2a,4a,7,9a-octahydrotetraazacycloocta[cd]pentalene, a critical macrocyclic intermediate used in the manufacture of gadolinium-based contrast agents such as Gadoteridol and Gadobutrol. This technology addresses long-standing challenges in scalability and purity that have historically plagued the production of these complex nitrogen-containing heterocycles. By shifting from traditional solvent-heavy methodologies to a novel solvent-free condensation approach, the patent establishes a new benchmark for industrial efficiency. For R&D directors and procurement specialists, understanding the nuances of this patented route is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent regulatory standards. The ability to produce this key building block with minimal impurities directly impacts the safety profile and commercial viability of the final diagnostic drug product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this macrocyclic intermediate relied on methods described in earlier literature, such as those by Atkins, which utilized N,N-dimethylformamide-dimethyl acetal in aromatic solvents like benzene. These conventional processes suffer from severe operational drawbacks that hinder their applicability on a commercial scale. The reagents involved are highly reactive towards nucleophiles, necessitating rigorously anhydrous conditions that are difficult and expensive to maintain in large reactors. Furthermore, the reaction generates toxic dimethylamine gas as a by-product, requiring specialized gas scrubbing equipment and posing significant safety risks to plant personnel. The use of large volumes of aromatic solvents not only increases raw material costs but also creates substantial waste disposal burdens and environmental compliance issues. Additionally, the stoichiometry must be controlled with extreme precision; any excess of the acetal reagent leads to rapid by-product formation, while insufficient amounts leave unreacted starting material, complicating downstream purification and reducing overall process yield.
The Novel Approach
In stark contrast, the patented process introduces a robust and economically superior methodology by employing triethyl orthoformate in the absence of solvent. This strategic shift eliminates the need for hazardous aromatic solvents and avoids the generation of toxic gaseous by-products, replacing them with ethanol which can be easily recovered and recycled. The lower reactivity of triethyl orthoformate compared to dimethyl acetals allows for safer handling and less stringent addition protocols, enabling the reagent to be added at the beginning of the reaction without risking runaway exotherms or excessive impurity formation. This simplification of the reaction profile significantly reduces the complexity of process monitoring, as the progress can be tracked simply by measuring the volume of distilled ethanol rather than relying on frequent gas chromatography analysis. Consequently, this approach offers a streamlined pathway that enhances both operator safety and production throughput, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Acid-Catalyzed Cyclization
The core of this technological advancement lies in the acid-catalyzed condensation mechanism between 1,4,7,10-tetraazacyclododecane and triethyl orthoformate. The reaction is facilitated by the presence of a C3-C18 carboxylic acid catalyst, such as propionic acid or pivalic acid, which activates the orthoformate for nucleophilic attack by the secondary amines of the macrocycle. Operating at elevated temperatures between 110°C and 150°C, the system drives the equilibrium forward through the continuous removal of ethanol, a by-product of the transacetalization process. This distillation-driven equilibrium shift is crucial for achieving the high conversion rates reported in the patent examples. The absence of solvent ensures a high concentration of reactants, which kinetically favors the intramolecular cyclization over intermolecular side reactions. Moreover, the exclusion of oxygen and light during the reaction prevents oxidative degradation of the sensitive amine substrate, ensuring the formation of a clean product profile with minimal colored impurities.
Impurity control is inherently built into this mechanistic design, addressing a primary concern for R&D teams focused on purity and impurity profiles. In conventional methods, water contamination leads to the hydrolysis of the intermediate back to the mono-formyl derivative, creating a complex mixture that is difficult to separate. However, the new process incorporates an initial drying step where the starting tetraazacyclododecane is melted and stripped of water using an alcohol azeotrope. This pre-treatment ensures that the reaction environment remains sufficiently dry to prevent hydrolysis without requiring exotic drying agents. The resulting crude product typically exhibits a purity of 95-98% by area normalization, which can be further enhanced to over 90% isolated yield through fractional distillation. This high level of chemical integrity is vital for the subsequent carboxymethylation steps required to produce the final chelating agents.
How to Synthesize 5H,9bH-2a,4a,7,9a-octahydrotetraazacycloocta[cd]pentalene Efficiently
Implementing this synthesis route requires careful attention to the initial drying phase and the precise control of reaction temperature to maximize yield. The process begins with the dissolution of the starting macrocycle in a drying alcohol such as n-butanol or n-pentanol, followed by vacuum distillation to remove water azeotropically until the residual pressure and temperature indicate a dry melt. Once the substrate is prepared, triethyl orthoformate and the selected carboxylic acid catalyst are introduced under a nitrogen blanket to maintain an inert atmosphere. The mixture is then heated to the target range, allowing the generated ethanol to distill off continuously, which serves as a real-time indicator of reaction progress. Detailed standardized synthesis steps see the guide below.
- Dry 1,4,7,10-tetraazacyclododecane by azeotropic distillation with butanol or pentanol under reduced pressure to remove water.
- Add triethyl orthoformate and a C3-C18 carboxylic acid catalyst to the molten substrate under nitrogen atmosphere.
- Heat the mixture to 110-150°C for 4-24 hours, distilling off generated ethanol to drive the reaction to completion.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this patented solvent-free process represents a significant opportunity for cost reduction in pharmaceutical intermediate manufacturing. By eliminating the need for large volumes of aromatic solvents, the process drastically reduces raw material procurement costs and the associated logistics of solvent recovery and disposal. The replacement of toxic dimethylamine-generating reagents with triethyl orthoformate removes the capital expenditure requirement for specialized gas scrubbing systems, thereby lowering the barrier to entry for production facilities. Furthermore, the robustness of the reaction against minor variations in reagent addition reduces the risk of batch failures, enhancing overall supply chain reliability and ensuring consistent availability of this critical high-purity pharmaceutical intermediate. These factors combine to create a more resilient and economically efficient supply model for downstream drug manufacturers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the substitution of expensive and hazardous reagents with cheaper, safer alternatives like triethyl orthoformate. The elimination of aromatic solvents removes significant costs related to solvent purchase, recycling infrastructure, and hazardous waste treatment fees. Additionally, the simplified reaction monitoring reduces labor hours and analytical costs, as the process does not require constant gas chromatography checks to prevent over-reaction. The ability to recycle the co-produced ethanol further contributes to the overall cost efficiency, creating a closed-loop system that minimizes waste and maximizes resource utilization.
- Enhanced Supply Chain Reliability: Supply continuity is strengthened by the use of commercially available and stable reagents that do not require special storage conditions or handling precautions beyond standard industrial practices. The reduced sensitivity to moisture compared to previous methods means that the process is less prone to disruptions caused by environmental humidity fluctuations or minor deviations in raw material quality. This robustness allows for more predictable production scheduling and shorter lead times for high-purity macrocyclic ligands, ensuring that downstream synthesis of MRI contrast agents can proceed without interruption due to intermediate shortages.
- Scalability and Environmental Compliance: Scaling this solvent-free process is inherently simpler than scaling solvent-heavy reactions, as heat transfer and mixing are more efficient in a neat reaction mass. The absence of volatile organic compounds (VOCs) from aromatic solvents significantly eases environmental compliance burdens, reducing the need for complex emission control systems. The generation of ethanol as the only major by-product simplifies waste stream management, aligning with modern green chemistry principles and corporate sustainability goals. This ease of scale-up facilitates the commercial scale-up of complex pharmaceutical intermediates from pilot plants to multi-ton annual production capacities without significant process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific pain points identified in the background art and the beneficial effects demonstrated in the patent examples. Understanding these aspects helps stakeholders evaluate the feasibility of adopting this route for their specific supply chain needs. The answers reflect the objective data provided in the patent documentation regarding yields, conditions, and impurity profiles.
Q: Why is water removal critical in this synthesis?
A: Water hydrolyzes the triethyl orthoformate reagent and the product intermediate, leading to significant yield loss and formation of by-products like 1-formyl-1,4,7,10-tetraazacyclododecane.
Q: What are the advantages of using triethyl orthoformate over DMF-dimethyl acetal?
A: Triethyl orthoformate is less reactive, safer to handle, does not produce toxic dimethylamine gas, and is more cost-effective, eliminating the need for complex gas scrubbing systems.
Q: What yields can be expected from this patented process?
A: The process typically achieves crude yields of 95-98% and purified yields exceeding 90%, which is superior to conventional solvent-based methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5H,9bH-2a,4a,7,9a-octahydrotetraazacycloocta[cd]pentalene Supplier
As a leading CDMO expert, NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like this patented process are executed with precision. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for MRI contrast agent synthesis. We understand the critical nature of this building block in the diagnostic supply chain and are committed to delivering consistent quality that supports your regulatory filings and commercial launch timelines. Our technical team is ready to collaborate on process optimization to further enhance efficiency and yield.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that identifies opportunities to optimize your supply chain without compromising on quality or compliance. Let us help you secure a stable source of high-quality intermediates that drive your diagnostic innovations forward.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
