Advanced Synthetic Route for Ioxilan Intermediates: Enhancing Purity and Commercial Scalability
Advanced Synthetic Route for Ioxilan Intermediates: Enhancing Purity and Commercial Scalability
The pharmaceutical industry's relentless pursuit of safer, non-ionic X-ray contrast media has driven significant innovation in the synthesis of key precursors like Ioxilan intermediates. Patent CN101948404B, filed in 2013, introduces a transformative methodology for preparing 5-acetamido-N-(2,3-dihydroxypropyl)-N'-(2-hydroxyethyl)-2,4,6-triiodo-1,3-benzenedicarboxamide, a critical building block for the final active pharmaceutical ingredient. This technical disclosure moves away from hazardous acyl chloride chemistry, opting instead for a streamlined sequence of esterification, amidation, and controlled iodination. For R&D directors and procurement specialists, this shift represents not merely a chemical curiosity but a viable pathway to cost reduction in pharmaceutical intermediates manufacturing while simultaneously addressing stringent environmental compliance standards. The protocol leverages common reagents like methanol and iron powder to replace toxic thionyl chloride, fundamentally altering the safety profile and economic feasibility of large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex triiodo-benzene derivatives relied heavily on the activation of carboxylic acid groups using thionyl chloride (SOCl2) to generate reactive acyl chlorides. While chemically effective, this traditional approach presents severe operational hazards and supply chain vulnerabilities. The generation of acyl chlorides requires strict anhydrous conditions and produces sulfur dioxide and hydrogen chloride gases as byproducts, necessitating expensive scrubbing systems and specialized corrosion-resistant reactor linings. Furthermore, conventional routes often introduce the expensive iodinating agent, iodine monochloride, at the very beginning of the synthesis. This premature iodination means that if subsequent steps fail or yield low purity, the costly iodine atoms are lost in the waste stream, drastically inflating the cost of goods sold (COGS). The reliance on such aggressive reagents also complicates the purification process, often leading to broader impurity profiles that require extensive downstream processing to meet the rigorous specifications of injectable drugs.
The Novel Approach
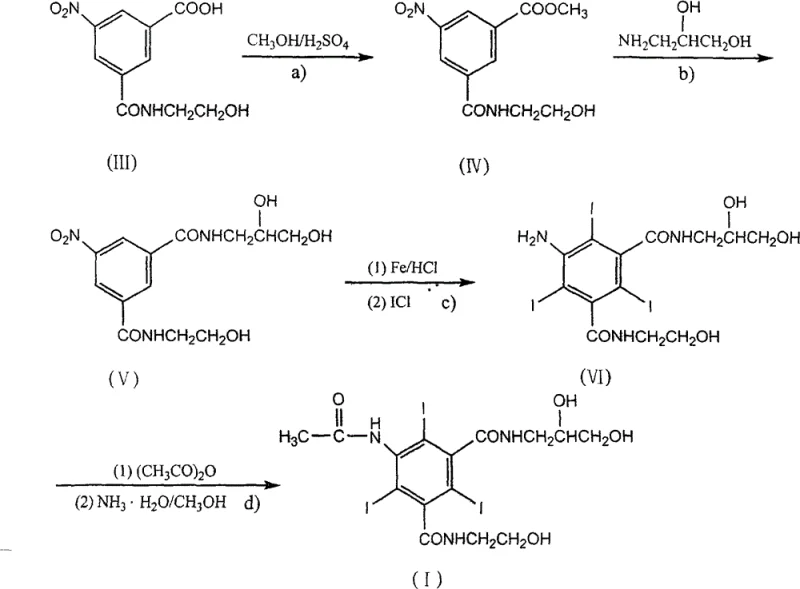
The methodology outlined in the patent data proposes a sophisticated rearrangement of the synthetic sequence to mitigate these risks effectively. By initiating the process with a methyl esterification of the nitro-benzoic acid starting material, the route activates the carboxyl group without generating toxic gases. This is followed by a direct amidation with 3-amino-1,2-propanediol, constructing the complex amide backbone before any iodine is introduced. This strategic sequencing ensures that the expensive iodine monochloride is only utilized on a fully formed molecular scaffold, maximizing atom economy. The final steps involve a gentle acetylation and subsequent hydrolysis using aqueous ammonia, which offers superior selectivity compared to strong alkaline hydrolysis. This novel approach not only simplifies the operational workflow but also aligns perfectly with the goals of a reliable pharmaceutical intermediates supplier seeking to minimize environmental liability while maximizing yield consistency.
Mechanistic Insights into Reductive Iodination and Selective Hydrolysis
The core chemical innovation lies in the tandem reduction-iodination step performed in an aqueous medium. In this phase, the nitro group of the diamide intermediate is reduced to an amino group using iron powder in hydrochloric acid. This in situ generation of the aniline derivative is immediately followed by electrophilic aromatic substitution using iodine monochloride. The presence of the electron-donating amino group activates the benzene ring specifically at the ortho and para positions, facilitating the introduction of three iodine atoms with high regioselectivity to form the 2,4,6-triiodo pattern essential for X-ray opacity. The use of an aqueous system here is particularly advantageous as it dissolves the inorganic salts generated during reduction, allowing for easy filtration of the organic product. This mechanistic precision ensures that the iodine atoms are locked into the most stable positions on the ring, preventing isomerization and ensuring the structural integrity required for the final contrast agent's low osmolarity and high tolerance.
Following iodination, the acetylation and hydrolysis sequence serves as a critical purification and protection strategy. The amino group is acetylated using acetic anhydride to prevent unwanted side reactions, and subsequently, the ester groups are hydrolyzed. Crucially, the patent specifies the use of aqueous ammonia for this hydrolysis rather than sodium hydroxide or other strong bases. Strong bases can induce nucleophilic attack on the amide bonds or even cause de-iodination of the sensitive triiodo-benzene core under heated conditions. Ammonia, being a weaker base, provides the necessary nucleophilicity to cleave the methyl esters while maintaining a pH environment that preserves the carbon-iodine bonds. This subtle control over reaction conditions is what allows the process to achieve high-purity pharmaceutical intermediates with minimal degradation products, directly addressing the purity concerns of R&D teams developing parenteral formulations.
How to Synthesize 5-Acetamido-N-(2,3-dihydroxypropyl)-N'-(2-hydroxyethyl)-2,4,6-triiodo-1,3-benzenedicarboxamide Efficiently
The execution of this synthesis requires precise control over stoichiometry and temperature to ensure optimal conversion at each stage. The process begins with the dissolution of the nitro-acid starting material in methanol, followed by the careful addition of catalytic sulfuric acid to drive the esterification equilibrium forward. Once the methyl ester is isolated, it undergoes amidation with 3-amino-1,2-propanediol, a step that requires prolonged reflux to overcome the lower reactivity of the ester compared to an acyl chloride. The subsequent reduction-iodination is exothermic and must be managed by controlled addition of iron powder and iodine monochloride to prevent thermal runaway. Finally, the acetylation is performed in excess acetic anhydride, and the hydrolysis is allowed to proceed at room temperature or mild heating with ammonia water. For detailed standard operating procedures and specific quality control checkpoints, please refer to the standardized synthesis guide below.
- Perform methyl esterification of 5-nitro-N-(2-hydroxyethyl)-formamide benzoic acid using methanol and sulfuric acid catalyst at 70°C.
- Conduct amidation reaction with 3-amino-1,2-propanediol in methanol solution at reflux temperatures to form the nitro-di-amide intermediate.
- Execute reductive iodination using iron powder in hydrochloric acid followed by iodine monochloride addition at 80-90°C.
- Finalize with acetylation using acetic anhydride followed by mild hydrolysis with aqueous ammonia to yield the target triiodo compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this synthetic route offers profound advantages in terms of raw material security and operational expenditure. By eliminating the need for thionyl chloride, manufacturers remove a highly regulated and hazardous material from their inventory, reducing the costs associated with storage, handling, and emergency response planning. The shift to methanol and acetic anhydride utilizes commodity chemicals that are widely available and less subject to volatile price fluctuations compared to specialized acylating agents. Furthermore, the avoidance of early-stage iodination means that the bulk of the synthesis can be performed with cheaper, non-iodinated intermediates. This modularity allows for better inventory management; if a batch fails prior to the iodination step, the financial loss is significantly contained. This resilience is crucial for maintaining supply chain reliability in the face of global raw material shortages.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the improved atom economy and the reduction of waste disposal costs. By delaying the introduction of iodine monochloride until the molecule is fully assembled, the process ensures that every gram of expensive iodine contributes to the final product yield, rather than being lost in purification losses of early intermediates. Additionally, the replacement of thionyl chloride eliminates the need for expensive corrosion-resistant equipment (such as glass-lined or Hastelloy reactors) and the associated maintenance downtime. The milder hydrolysis conditions using aqueous ammonia also reduce energy consumption, as intense heating or cooling is not required to manage violent exotherms typical of strong base hydrolysis. These factors combine to create a substantially lower cost base for the production of high-value contrast media intermediates.
- Enhanced Supply Chain Reliability: The reliance on robust, commodity-grade reagents like iron powder, methanol, and ammonia enhances the stability of the supply chain. Unlike specialized catalysts or hazardous gases that may face shipping restrictions or regulatory bottlenecks, these materials are staples of the chemical industry with multiple qualified suppliers globally. This diversification of the supply base mitigates the risk of production stoppages due to single-source failures. Moreover, the simplified work-up procedures, such as the easy filtration of iron sludge and the crystallization of products from aqueous mixtures, reduce the cycle time per batch. Faster turnaround times mean that manufacturing capacity can be utilized more efficiently, allowing suppliers to respond more agilely to sudden spikes in demand from downstream API manufacturers without compromising on delivery schedules.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of gaseous byproducts like SO2, which are difficult to manage at large scales. The aqueous nature of the reduction-iodination step makes heat transfer and mixing more manageable in large reactors, reducing the risk of hot spots that could lead to safety incidents. From an environmental standpoint, the process generates significantly less hazardous waste. The iron sludge can often be recycled or treated easily, and the aqueous mother liquors are less toxic than those containing chlorinated solvents or heavy metal catalysts. This alignment with green chemistry principles not only reduces waste disposal fees but also future-proofs the manufacturing site against increasingly stringent environmental regulations, ensuring long-term operational continuity and social license to operate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to legacy processes. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term value proposition of suppliers utilizing this advanced chemistry.
Q: How does this new method reduce environmental impact compared to traditional acyl chloride routes?
A: The novel process eliminates the use of thionyl chloride (SOCl2) for carboxyl activation, replacing it with a safer methyl esterification step. This significantly reduces the generation of corrosive sulfur dioxide gas and minimizes the need for complex waste gas treatment systems, thereby lowering the environmental footprint of the manufacturing facility.
Q: What represents the primary cost-saving advantage in this synthetic pathway?
A: The strategic delay of the iodination step until after the formation of the diamide backbone prevents the wasteful consumption of expensive iodine monochloride on impure intermediates. Additionally, replacing harsh acyl chloride reagents with methanol and acetic anhydride reduces raw material costs and equipment corrosion maintenance.
Q: Why is aqueous ammonia preferred for the final hydrolysis step?
A: Using aqueous ammonia for the hydrolysis of the acetylated intermediate provides much milder reaction conditions compared to strong inorganic bases. This gentleness preserves the integrity of the sensitive triiodo-benzene structure, preventing de-iodination side reactions and ensuring the final product achieves the high purity levels required for injectable contrast media applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ioxilan Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is critical for the future of the pharmaceutical industry. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101948404B are fully realized in practical manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with HPLC and NMR capabilities to verify that every batch of Ioxilan intermediate meets the exacting standards required for injectable contrast media. Our commitment to quality assurance ensures that the impurity profiles are tightly controlled, minimizing the burden on your downstream purification processes and guaranteeing the safety of the final medical product.
We invite global partners to collaborate with us to leverage these advanced manufacturing capabilities. By choosing NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our optimized synthesis of complex pharmaceutical intermediates can enhance your supply chain resilience and drive down your overall production costs while maintaining the highest standards of safety and efficacy.
