Optimizing Iohexol Manufacturing: Advanced Synthetic Routes for Commercial Scale-up and Purity Control
The global demand for non-ionic X-ray contrast agents continues to surge, driven by the increasing prevalence of diagnostic imaging procedures in modern healthcare systems. At the forefront of this market is Iohexol, a critical pharmaceutical intermediate known for its high safety profile and efficacy. However, the manufacturing landscape has long been challenged by complex synthesis routes that struggle to balance high purity with cost efficiency. A pivotal advancement in this domain is detailed in patent CN1907961A, which outlines a novel preparation method for Iohexol, specifically 5-[N-(2,3-dihydroxypropyl)acetyl]-N,N'-bis(2,3-dihydroxypropyl)-2,4,6-triiodo-1,3-benzamide. This technology addresses the persistent issue of O-alkylation by-products that plague conventional synthesis, offering a pathway to significantly enhanced yield and purity. For R&D directors and procurement specialists, understanding the mechanistic advantages of this protected intermediate route is essential for securing a reliable supply chain of high-quality contrast media.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing processes for Iohexol typically involve a direct N-alkylation step where the nitrogen atom of the benzamide core is alkylated with a dihydroxypropyl group. While conceptually straightforward, this approach suffers from a significant chemical drawback: the competitive O-alkylation of the hydroxyl groups present on the side chains. When the alkylating agent, such as 3-chloro-1,2-propylene glycol, is introduced, it does not distinguish exclusively between the nitrogen and oxygen nucleophiles. Consequently, a mixture of N-alkylated and O-alkylated products is formed. These O-alkylated impurities possess physicochemical properties very similar to the target Iohexol molecule, making their removal extremely difficult and costly. Conventional purification relies heavily on repeated recrystallization, which not only consumes vast amounts of solvent and energy but also results in substantial product loss, driving down the overall yield to levels that are economically suboptimal for large-scale production.
The Novel Approach
The innovative strategy presented in the referenced patent fundamentally alters the reaction sequence to circumvent the O-alkylation problem at its source. Instead of attempting to separate impurities after they form, this method employs a protective group strategy. By introducing an amino-acetonide group to protect the side-chain hydroxyls prior to the N-alkylation step, the oxygen atoms are effectively masked and rendered non-nucleophilic. This ensures that the alkylating agent reacts selectively with the nitrogen atom, drastically minimizing the formation of O-alkylated by-products. Furthermore, the deprotection step utilizes a cationic resin in a methanol-water system, which simplifies the post-reaction workup. This shift from complex purification to preventive chemistry represents a paradigm shift in process efficiency, offering a cleaner reaction profile and a more robust manufacturing protocol that is highly attractive for commercial scale-up.
Mechanistic Insights into Protective Group Strategy and N-Alkylation
The core of this technological breakthrough lies in the precise manipulation of functional group reactivity through temporary protection. In the initial stages, 5-amino-2,4,6-triiodo-1,3-benzoyl chloride is reacted to form an acetylamino intermediate. Crucially, the subsequent step involves the reaction with amino-acetonide under basic conditions, typically using catalysts like triethylamine at controlled low temperatures. This forms a stable ketal-like structure on the side chains. When the N-alkylation is subsequently performed using 3-chloro-1,2-propylene glycol in the presence of sodium hydroxide, the protected hydroxyl groups are inert. The steric bulk and electronic nature of the acetonide group prevent the oxygen from attacking the electrophilic carbon of the alkylating agent. This selectivity is the key to the high purity observed in the final product, as confirmed by HPLC analysis showing a marked reduction in related substances compared to unprotected routes.
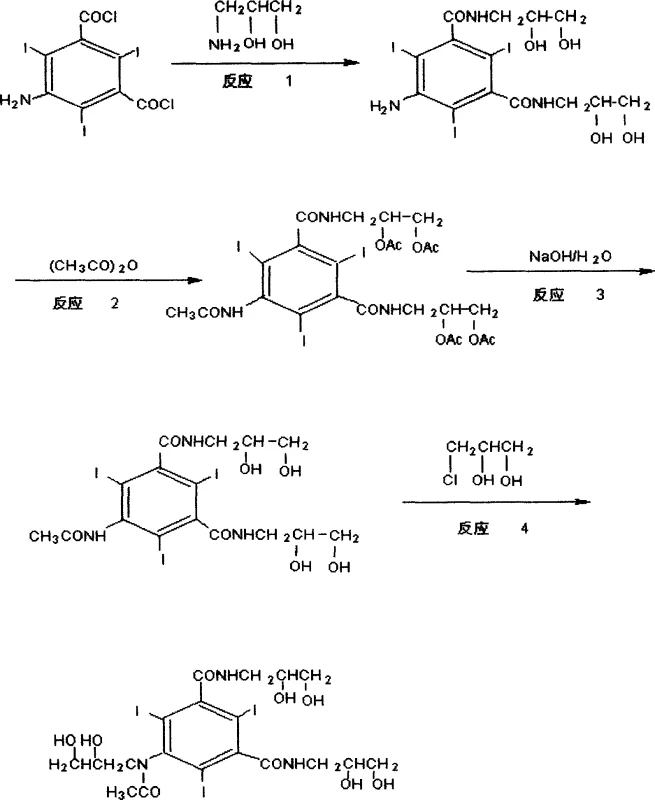
Following the alkylation, the removal of the protecting group is achieved through a mild acid-catalyzed hydrolysis facilitated by a cationic resin. This heterogeneous catalysis method offers distinct advantages over homogeneous acid hydrolysis. The resin acts as a solid acid source, releasing protons to cleave the acetonide bond and regenerate the free hydroxyl groups necessary for the final Iohexol structure. The use of a resin allows for easy separation via filtration, eliminating the need for neutralization steps that generate salt waste. Moreover, the resin system helps in scavenging residual ionic impurities, further polishing the crude product before the final drying stage. This mechanistic elegance ensures that the final API intermediate meets stringent purity specifications required for injectable contrast agents, reducing the burden on downstream quality control laboratories.
How to Synthesize Iohexol Efficiently
Implementing this optimized synthesis route requires careful attention to reaction conditions and stoichiometry to maximize the benefits of the protective group strategy. The process begins with the acetylation of the triiodobenzoyl chloride derivative, followed by the critical protection step with amino-acetonide. Once the protected intermediate is secured, the N-alkylation can proceed with confidence in its selectivity. The final deprotection using the cationic resin system is the culmination of the process, yielding the target molecule with minimal impurity load. For process chemists looking to adopt this methodology, the detailed standardized synthetic steps outlined below provide a clear roadmap for laboratory validation and pilot plant trials. Adhering to these parameters ensures reproducibility and safety while leveraging the full potential of this patented technology.
- Acetylate 5-amino-2,4,6-triiodo-1,3-benzoyl chloride using acetyl chloride and a catalyst like quinoline to form the acetylamino intermediate.
- Protect the side-chain hydroxyl groups by reacting with amino-acetonide in the presence of a base catalyst such as triethylamine at low temperatures.
- Perform N-alkylation with 3-chloro-1,2-propylene glycol, followed by deprotection using a cationic resin system to yield pure Iohexol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this advanced synthesis route offers tangible economic and operational benefits that extend beyond simple yield metrics. The primary advantage lies in the drastic simplification of the purification workflow. By preventing the formation of hard-to-remove O-alkylated impurities, the need for multiple, solvent-intensive recrystallization cycles is eliminated. This reduction in downstream processing directly translates to lower consumption of organic solvents and reduced energy usage for heating and cooling, contributing to significant cost reduction in contrast agent manufacturing. Furthermore, the streamlined process shortens the overall production cycle time, allowing for faster turnover and improved responsiveness to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of complex purification steps significantly lowers the operational expenditure associated with solvent recovery and waste disposal. Since the reaction selectively produces the desired N-alkylated product, the yield loss typically associated with aggressive purification is minimized. This efficiency gain means that less raw material is required to produce the same amount of finished goods, effectively lowering the cost of goods sold (COGS). Additionally, the use of resin-based deprotection reduces the consumption of liquid acids and bases, further decreasing the cost of consumables and the environmental footprint of the manufacturing facility.
- Enhanced Supply Chain Reliability: A more robust synthesis route inherently reduces the risk of batch failures due to out-of-specification impurity profiles. In the context of pharmaceutical intermediates, consistency is paramount. This method's ability to consistently deliver high-purity Iohexol reduces the likelihood of production delays caused by failed quality control tests. For supply chain planners, this reliability ensures a steady flow of materials, reducing the need for excessive safety stock and mitigating the risks associated with supply disruptions. The simplified process also makes it easier to transfer technology between manufacturing sites, enhancing overall supply chain resilience.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind. The use of heterogeneous catalysis (cationic resin) simplifies reactor design and operation, making it easier to scale from pilot batches to multi-ton commercial production. From an environmental perspective, the reduction in solvent use and waste generation aligns with increasingly stringent global regulations on chemical manufacturing. This compliance advantage not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is becoming a critical factor in vendor selection for major pharmaceutical companies seeking green supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Iohexol synthesis technology. These insights are derived directly from the patent data and are intended to clarify the operational benefits for potential partners. Understanding these details is crucial for making informed decisions about process adoption and supplier qualification. The answers reflect the consensus on how protective group chemistry can solve long-standing manufacturing bottlenecks in the contrast agent sector.
Q: How does the new process reduce O-alkylated impurities in Iohexol?
A: The process introduces an amino-acetonide protective group on the side-chain hydroxyls before N-alkylation. This steric and electronic protection prevents the oxygen atoms from attacking the alkylating agent, significantly reducing the formation of O-alkylated by-products compared to conventional methods.
Q: What are the yield improvements in the final deprotection step?
A: By utilizing a cationic resin/methanol-water system for deprotection, the method achieves a yield of approximately 96% in the final step. This is a substantial improvement over traditional methods which often suffer from product loss during complex recrystallization processes required to remove impurities.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the method is designed for scalability. It avoids harsh conditions and complex purification steps like repeated recrystallization. The use of resin-based deprotection simplifies the workup, making it safer, easier to operate, and more energy-efficient for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iohexol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the theoretical advantages of a patent must be translated into practical, commercial reality. As a leading CDMO and supplier in the fine chemical sector, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our engineering teams are adept at adapting complex synthetic routes, such as the protected intermediate method for Iohexol, to fit our state-of-the-art manufacturing infrastructure. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to ensure that every batch of Iohexol meets the exacting standards required for pharmaceutical applications. Our commitment to quality ensures that the impurity profiles remain well within the limits defined by international pharmacopoeias.
We invite procurement leaders and R&D directors to collaborate with us to optimize your supply chain for contrast agent intermediates. By leveraging our technical expertise, you can achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can establish a long-term partnership that secures your supply of high-purity Iohexol while driving down costs through process innovation and operational excellence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
