Breakthrough Catalyst-Free Synthesis of Bioactive Isochromene Derivatives for Commercial Drug Development
Breakthrough Catalyst-Free Synthesis of Bioactive Isochromene Derivatives for Commercial Drug Development
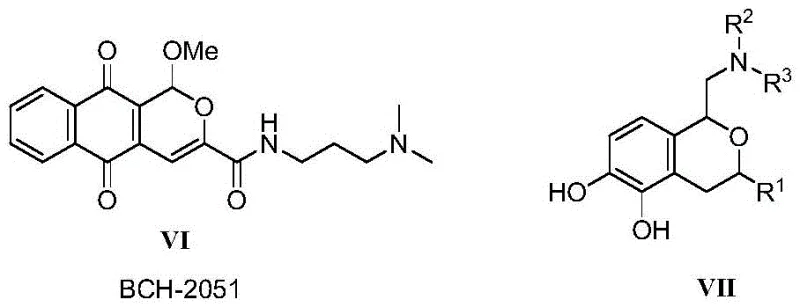
The pharmaceutical industry is constantly seeking efficient pathways to access complex heterocyclic scaffolds that serve as critical building blocks for next-generation therapeutics. Patent CN113135884B introduces a significant advancement in this domain by disclosing a novel class of isochromene compounds and, more importantly, a robust preparation method that eliminates the need for transition metal catalysts. This innovation addresses a major pain point in medicinal chemistry, where traditional synthetic routes often rely on expensive and toxic metals such as palladium or gold. By leveraging a thermal cyclization strategy between nitro-quinone ethylene derivatives and cyclic 1,3-diones, this technology offers a streamlined approach to generating high-purity intermediates. For R&D directors and procurement specialists, this represents a tangible opportunity to optimize supply chains for SIRT inhibitor development, ensuring both cost efficiency and regulatory compliance regarding heavy metal residues in final drug substances.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the isochromene core has been heavily dependent on transition metal catalysis, which introduces several bottlenecks for commercial manufacturing. Literature precedents often describe the use of Palladium(II) or Gold(I) complexes to facilitate the addition of nucleophiles to alkynyl aldehydes, followed by intramolecular cyclization. While chemically effective on a small scale, these methods suffer from significant drawbacks when translated to industrial settings. The reliance on precious metals drastically inflates the bill of materials, and more critically, necessitates rigorous purification protocols to reduce metal content to parts-per-million levels required by regulatory agencies. Furthermore, these catalytic systems often require stringent anhydrous conditions or specific ligands that are sensitive to air and moisture, complicating the operational workflow and increasing the risk of batch failure due to catalyst deactivation.
The Novel Approach
In stark contrast, the methodology outlined in CN113135884B bypasses these complexities entirely by utilizing a catalyst-free thermal reaction. The process involves the direct reaction of (E)-β-nitro-p-benoquinone ethylene with cyclic 1,3-diones or 4-hydroxycoumarins in common organic solvents like toluene. This approach not only removes the cost burden of catalysts but also simplifies the reaction setup to a simple mix-and-heat operation. The absence of metal contaminants means that downstream processing is significantly streamlined, reducing the number of unit operations required for purification. This shift from catalytic dependency to thermal activation represents a paradigm shift in how these valuable heterocycles can be produced, offering a greener and more economically viable pathway for the synthesis of pharmaceutical intermediates targeting metabolic and oncological indications.
Mechanistic Insights into Thermal Cyclization and Nitro-Group Activation
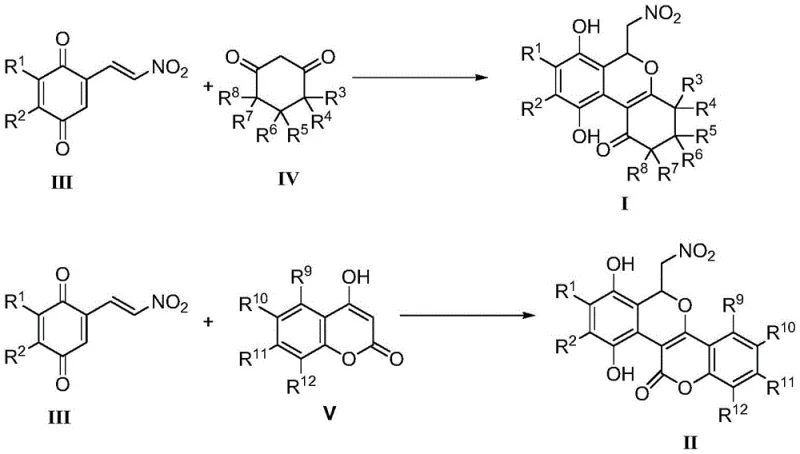
The success of this catalyst-free protocol lies in the unique electronic properties of the reactants, specifically the activation provided by the nitro group on the quinone ethylene moiety. In the absence of a Lewis acid catalyst to activate the carbonyl or alkyne groups, the reaction relies on the inherent electrophilicity of the nitro-alkene system. The mechanism likely proceeds through a Michael-type addition where the enol form of the cyclic 1,3-dione attacks the electron-deficient beta-carbon of the nitro-ethylene. This initial adduct then undergoes an intramolecular cyclization, driven by the nucleophilic attack of the phenolic or enolic oxygen onto the adjacent electrophilic center, eventually leading to the formation of the fused isochromene ring system. The nitro group serves a dual purpose: it activates the double bond for the initial addition and stabilizes the transition states through resonance, allowing the reaction to proceed smoothly at moderate temperatures without external promotion.
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. Metal-catalyzed reactions often generate side products resulting from beta-hydride elimination, homocoupling of alkynes, or ligand degradation. In this thermal process, the reaction pathway is more deterministic, primarily governed by the stoichiometry and thermodynamics of the cycloaddition. The major byproducts are typically unreacted starting materials or simple hydrolysis products, which are far easier to separate from the desired product than organometallic complexes. This cleaner reaction profile translates directly to higher crude purity, reducing the load on chromatographic purification steps and improving the overall mass balance of the manufacturing process, which is a key metric for process chemists evaluating scalability.
How to Synthesize Isochromene Derivatives Efficiently
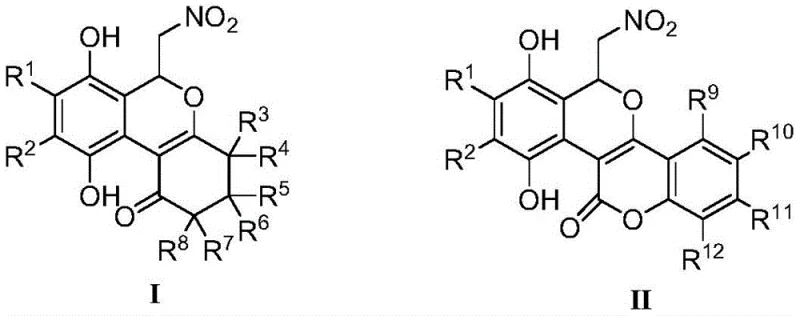
The practical implementation of this synthesis is designed for operational simplicity, making it accessible for both laboratory discovery and pilot plant operations. The standard protocol involves dissolving the cyclic dione precursor in a solvent such as toluene, followed by the addition of the nitro-quinone ethylene reagent. The mixture is then heated to a moderate temperature range, typically between 30°C and 70°C, and maintained for a period of 24 to 36 hours to ensure complete conversion. Following the reaction, the solvent is removed under reduced pressure, and the crude solid is purified using standard silica gel column chromatography. This straightforward procedure eliminates the need for inert atmosphere gloveboxes or specialized catalytic reactors, lowering the barrier to entry for producing these high-value intermediates.
- Dissolve the cyclic 1,3-dione or 4-hydroxycoumarin starting material in an organic solvent such as toluene under stirring.
- Add (E)-β-nitro-p-benzoquinone ethylene to the reaction mixture and maintain the temperature between 30°C and 70°C for 24 to 36 hours.
- Remove the solvent under reduced pressure and purify the resulting residue via column chromatography to isolate the high-purity isochromene product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalyst-free technology offers compelling economic and logistical benefits that extend beyond simple yield metrics. The most immediate impact is seen in the reduction of raw material costs, as the elimination of precious metal catalysts removes a volatile and expensive line item from the budget. Additionally, the simplified purification process reduces the consumption of chromatography media and solvents, further driving down the cost of goods sold. From a supply chain reliability standpoint, the use of commodity chemicals like toluene and readily available cyclic diones ensures that production is not held hostage by the availability of specialized ligands or custom-synthesized catalysts, thereby enhancing the resilience of the supply chain against market fluctuations.
- Cost Reduction in Manufacturing: The removal of transition metal catalysts such as palladium or gold fundamentally alters the cost structure of isochromene production. Without the need for expensive metal salts and phosphine ligands, the direct material costs are significantly lowered. Furthermore, the absence of heavy metals eliminates the requirement for costly scavenging resins or specialized filtration steps designed to meet strict ICH Q3D guidelines for elemental impurities. This streamlining of the downstream process results in substantial operational savings, allowing for more competitive pricing of the final pharmaceutical intermediate while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: Relying on thermal activation rather than sensitive catalytic systems greatly improves the robustness of the manufacturing process. Traditional metal-catalyzed reactions can be susceptible to trace impurities in solvents or reagents that poison the catalyst, leading to stalled reactions and delayed batches. In contrast, this thermal method is tolerant of minor variations in reagent quality, ensuring consistent batch-to-batch performance. Moreover, the starting materials are stable and commercially available in bulk quantities, reducing the risk of supply disruptions. This reliability allows for more accurate production planning and shorter lead times for delivering high-purity intermediates to downstream drug substance manufacturers.
- Scalability and Environmental Compliance: The mild reaction conditions and lack of toxic metal waste make this process inherently scalable and environmentally friendly. Scaling up exothermic metal-catalyzed reactions often requires complex engineering controls to manage heat release and prevent runaway scenarios. The gentle thermal profile of this synthesis minimizes such risks, facilitating a smoother transition from kilogram to ton-scale production. Additionally, the reduction in hazardous waste generation aligns with green chemistry principles, simplifying waste disposal compliance and reducing the environmental footprint of the manufacturing facility. This sustainability angle is increasingly important for pharmaceutical companies aiming to meet corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these isochromene compounds. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the technology's capabilities. Understanding these details is crucial for partners looking to integrate this chemistry into their existing drug development pipelines or sourcing strategies.
Q: What are the primary advantages of this catalyst-free synthesis method?
A: The primary advantage is the elimination of expensive transition metal catalysts like Palladium or Gold, which significantly reduces raw material costs and simplifies downstream purification by removing the need for heavy metal scavenging steps.
Q: What is the biological activity profile of these isochromene compounds?
A: These compounds act as potent inhibitors of SIRT1 and SIRT5 enzymes, showing IC50 values in the low micromolar range, making them highly relevant for developing treatments for metabolic diseases, tumors, and neurodegenerative disorders.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes mild reaction temperatures (30°C-70°C) and common organic solvents like toluene, avoiding harsh conditions and specialized catalytic equipment, which facilitates straightforward scale-up from laboratory to industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isochromene Compound Supplier
As the demand for targeted SIRT inhibitors continues to grow in the fields of oncology and metabolic disease, securing a reliable source of high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands ready to support your development needs with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the thermal cyclization processes described in CN113135884B with precision, ensuring that every batch meets stringent purity specifications. We understand the critical nature of these intermediates in the drug discovery value chain and employ rigorous QC labs to verify identity and potency, guaranteeing that our clients receive materials that accelerate rather than hinder their research timelines.
We invite you to engage with our technical team to discuss how this catalyst-free technology can be integrated into your specific project requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this novel synthetic route. We encourage you to contact our technical procurement team today to request specific COA data for our isochromene library and to discuss route feasibility assessments tailored to your target molecules. Let us help you optimize your supply chain and bring your therapeutic candidates to the clinic faster and more efficiently.
