Advanced Microbial Fermentation Technology for Scalable Glycyrrhetinic Acid Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking sustainable alternatives to traditional plant extraction methods, particularly for high-value natural product derivatives like glycyrrhetinic acid. Patent CN102363796B introduces a groundbreaking method for producing glycyrrhetinic acid through microbial fermentation transformation, utilizing a specifically mutated strain of Aspergillus flavus (CGMCC No. 5144). This technology represents a significant leap forward in biocatalysis, addressing the critical bottlenecks of resource scarcity and environmental pollution associated with conventional licorice root processing. By leveraging advanced fermentation techniques combined with precise membrane separation technologies, this process achieves a fermentation yield of 4.38 g/L and a product purity exceeding 98%, with a conversion rate greater than 90%. For R&D directors and procurement specialists, this patent outlines a robust pathway to secure a reliable glycyrrhetinic acid supplier capable of meeting stringent quality specifications without relying on fluctuating agricultural outputs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the production of glycyrrhetinic acid has relied heavily on the extraction of glycyrrhizic acid from licorice roots followed by acid hydrolysis. This conventional approach suffers from severe limitations that impact both cost efficiency and supply chain stability. Firstly, licorice is a slow-growing medicinal plant primarily distributed in arid regions, and over-harvesting has led to ecological deterioration and a sharp decline in raw material availability. Secondly, the acid hydrolysis process typically requires harsh reaction conditions, often involving strong acids and high temperatures, which not only degrade the product quality but also generate significant amounts of hazardous waste liquid that requires expensive treatment. Furthermore, the specificity of chemical hydrolysis is often poor, leading to complex impurity profiles that necessitate rigorous and costly purification steps to meet pharmaceutical grade standards. These factors collectively result in high production costs and unpredictable lead times, posing a substantial risk to manufacturers seeking cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the novel microbial fermentation method described in the patent offers a sustainable and highly efficient alternative. By employing Aspergillus flavus CGMCC No. 5144, which has been optimized through UV and LiCl mutagenesis, the process utilizes the organism's secreted beta-glucuronidase to specifically catalyze the hydrolysis of glycyrrhizic acid under mild conditions. This biological transformation eliminates the need for corrosive acids and extreme temperatures, thereby preserving the structural integrity of the target molecule and minimizing the formation of by-products. Additionally, the process ingeniously utilizes licorice leachate cooking liquid as a culture medium component, effectively turning a potential waste stream into a valuable carbon and nitrogen source. This circular economy approach not only reduces raw material costs but also significantly lowers the environmental footprint of the manufacturing process, aligning perfectly with modern green chemistry principles and regulatory requirements for industrial scale-up.
Mechanistic Insights into Enzymatic Hydrolysis and Membrane Separation
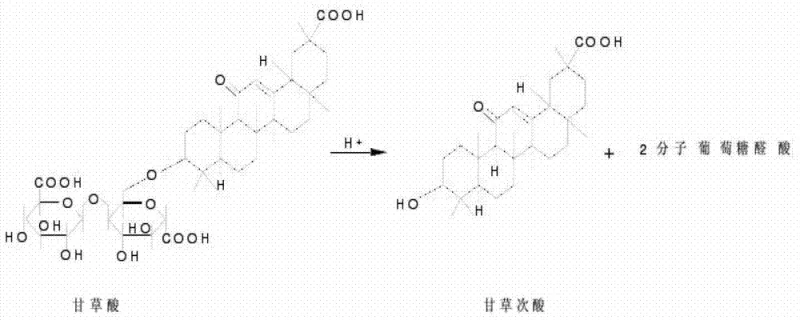
The core of this technological breakthrough lies in the precise enzymatic mechanism and the subsequent downstream processing strategy. The mutated Aspergillus flavus strain secretes high levels of beta-glucuronidase, an enzyme that specifically targets the beta-glucuronide glycosidic bonds in glycyrrhizic acid. This enzymatic cleavage converts glycyrrhizic acid into glycyrrhetinic acid and two molecules of glucuronic acid, mirroring the metabolic pathway found in the human liver but occurring in a controlled bioreactor environment. The specificity of this enzymatic reaction ensures that the aglycone structure remains intact, avoiding the degradation often seen in chemical hydrolysis. Following fermentation, the recovery of the product is achieved through a sophisticated membrane separation combined process. Ultrafiltration using PES-20 membranes removes macromolecular impurities and cellular debris with molecular weights between 500 and 500,000 Daltons, while subsequent nanofiltration with NF-270 membranes concentrates the target product by retaining lower molecular weight substances in the 200 to 1,000 Dalton range. This dual-membrane strategy effectively fractionates impurities based on size, drastically simplifying the purification load.
Following the membrane concentration steps, the crude product undergoes further purification using macroporous adsorbent resin H103 and silica gel column chromatography. The use of macroporous resin allows for the selective adsorption of glycyrrhetinic acid from the aqueous fermentation broth, effectively removing polar impurities and salts. The final polishing step involves silica gel column chromatography using a petroleum ether and ethyl acetate gradient, which separates the target compound from any remaining structurally similar impurities. This multi-stage purification protocol ensures that the final product meets the high-purity glycyrrhetinic acid standards required for pharmaceutical applications, with purity levels consistently reaching above 98%. The integration of biological catalysis with advanced physical separation techniques creates a synergistic effect that maximizes yield while minimizing solvent consumption and waste generation, providing a compelling value proposition for supply chain heads focused on sustainability and efficiency.
How to Synthesize Glycyrrhetinic Acid Efficiently
The synthesis of glycyrrhetinic acid via this microbial route involves a carefully orchestrated sequence of fermentation and separation steps designed to maximize enzyme activity and product recovery. The process begins with the activation of the Aspergillus flavus strain on solid media, followed by expansion in a liquid seed culture optimized with corn powder saccharification liquid and wheat bran juice to stimulate rapid mycelial growth. Once the seed culture reaches the vegetative phase, it is inoculated into the main fermentation tank containing the glycyrrhizic acid substrate. The fermentation parameters, including temperature (20-40°C), pH (4-5), and dissolved oxygen, are tightly controlled to maintain optimal enzyme expression. Detailed standardized synthesis steps see the guide below.
- Cultivate Aspergillus flavus CGMCC No. 5144 on solid PDA slants, then inoculate 1% into liquid seed medium containing corn powder saccharification liquid and wheat bran juice for 24 hours.
- Transfer 30% of the seed culture into a fermentation tank containing glycyrrhizic acid substrate, maintaining temperature at 20-40°C and stirring at 130-170 rpm for 2-4 days.
- Separate the fermentation broth using ultrafiltration (PES-20) and nanofiltration (NF-270) membranes, followed by purification via macroporous resin H103 and silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this microbial fermentation technology offers transformative advantages over traditional extraction methods. The primary benefit is the decoupling of production from agricultural cycles; since the process relies on fermentation tanks rather than field-grown licorice roots, manufacturers can ensure a continuous, year-round supply of high-purity intermediates regardless of seasonal weather patterns or crop failures. This reliability is crucial for maintaining uninterrupted production schedules for downstream pharmaceutical formulations. Furthermore, the utilization of licorice leachate as a fermentation substrate significantly reduces raw material costs, as it valorizes a low-value by-product of the initial extraction process. This internal recycling of materials creates a closed-loop system that drives down the overall cost of goods sold (COGS) without compromising on quality.
- Cost Reduction in Manufacturing: The elimination of harsh acid hydrolysis reagents and the associated neutralization wastes leads to substantial cost savings in waste treatment and disposal. Additionally, the high conversion rate (>90%) means that less raw glycyrrhizic acid is required to produce the same amount of final product, directly improving material efficiency. The use of membrane filtration instead of energy-intensive evaporation steps for concentration further reduces utility costs, making the process economically superior for large-scale operations.
- Enhanced Supply Chain Reliability: By shifting the production bottleneck from agricultural harvesting to industrial fermentation, the supply chain becomes far more resilient to external shocks such as droughts or trade restrictions on raw herbal materials. The stability of the mutated Aspergillus flavus strain ensures consistent batch-to-batch performance, reducing the risk of production delays caused by variable raw material quality. This predictability allows procurement teams to negotiate better long-term contracts and optimize inventory levels with confidence.
- Scalability and Environmental Compliance: The fermentation process is inherently scalable, with the patent demonstrating successful operation in 100L fermenters, indicating a clear path to multi-ton commercial production. The mild reaction conditions and reduced solvent usage align with increasingly strict environmental regulations, minimizing the risk of compliance-related shutdowns. The simplified downstream processing also reduces the volume of organic solvents required for purification, contributing to a greener manufacturing profile that appeals to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the microbial production of glycyrrhetinic acid. These answers are derived directly from the technical specifications and experimental data provided in patent CN102363796B, ensuring accuracy and relevance for industry professionals evaluating this technology for potential adoption or sourcing.
Q: How does microbial fermentation improve the purity of glycyrrhetinic acid compared to acid hydrolysis?
A: The microbial method utilizes specific beta-glucuronidase enzymes from Aspergillus flavus to selectively hydrolyze glycyrrhizic acid, avoiding the harsh conditions and side reactions associated with traditional acid hydrolysis, resulting in purity levels >= 98%.
Q: What are the key advantages of the membrane separation technology used in this process?
A: The combination of ultrafiltration and nanofiltration allows for the precise removal of impurities based on molecular weight ranges (500-500000 Da and 200-1000 Da respectively), significantly enhancing yield and reducing downstream purification costs.
Q: Is the Aspergillus flavus strain stable for large-scale industrial production?
A: Yes, the strain CGMCC No. 5144 has been optimized through UV and LiCl mutagenesis to ensure hereditary stability and high enzyme secretion, making it suitable for consistent commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glycyrrhetinic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of microbial fermentation technologies in revolutionizing the supply of complex natural product derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN102363796B can be seamlessly transitioned from the lab to the factory floor. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced fermentation capabilities, allowing us to meet stringent purity specifications for glycyrrhetinic acid and other high-value intermediates. We are committed to delivering consistent quality and reliability, helping our partners navigate the complexities of modern pharmaceutical supply chains.
We invite you to collaborate with us to optimize your supply chain for glycyrrhetinic acid and related compounds. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
