Advanced Manufacturing of N-Phenyl Bis(Trifluoromethanesulfonimide) for Global Pharmaceutical Supply Chains
Advanced Manufacturing of N-Phenyl Bis(Trifluoromethanesulfonimide) for Global Pharmaceutical Supply Chains
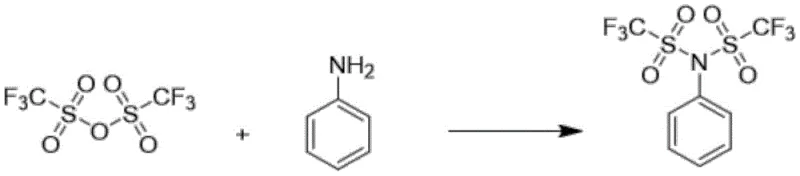
In the rapidly evolving landscape of fine chemical manufacturing, the efficient production of high-value trifluoromethanesulfonylation reagents remains a critical bottleneck for pharmaceutical developers. Patent CN115028557A introduces a groundbreaking preparation method for N-phenyl bis(trifluoromethanesulfonimide), often referred to as PhNTf2, which addresses longstanding issues regarding cost, safety, and scalability. This reagent is indispensable in modern organic synthesis, particularly for the installation of trifluoromethanesulfonyl groups in complex drug molecules such as Abiraterone Acetate, a key treatment for prostate cancer. The disclosed technology shifts the paradigm from hazardous gas-phase reactions and expensive anhydride-based routes to a robust liquid-phase process utilizing common alkali metal salts. For global supply chain leaders, this innovation represents a significant opportunity to secure a more reliable pharmaceutical intermediate supplier while drastically simplifying the manufacturing workflow.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of N-phenyl bis(trifluoromethanesulfonimide) has been plagued by severe economic and safety constraints inherent to traditional chemical pathways. As illustrated in the background art, conventional routes typically rely on the reaction of aniline with trifluoromethanesulfonic anhydride, a process that generates stoichiometric amounts of trifluoromethanesulfonic acid salts as waste. This not only results in poor atom economy but also creates a massive burden for waste treatment, driving up the operational expenditure for any fine chemical manufacturing facility. Alternative methods involving trifluoromethanesulfonyl fluoride gas present even greater challenges, requiring specialized high-pressure reactors and rigorous leak-proof systems to handle the highly corrosive and toxic nature of the gas. Furthermore, these gas-phase reactions often suffer from low reaction activity and catalyst deactivation, leading to inconsistent yields and necessitating complex purification steps to remove organic impurities and unreacted starting materials.
The Novel Approach
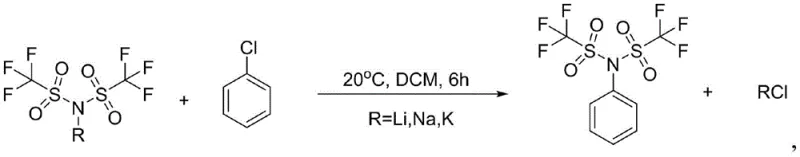
The methodology described in patent CN115028557A offers a transformative solution by utilizing alkali metal bistrifluoromethanesulfonimide salts (such as lithium, sodium, or potassium salts) reacting directly with chlorobenzene. This approach completely eliminates the need for hazardous fluorine-containing gases or expensive anhydrides, replacing them with stable, commercially available solid raw materials that are cheap and easy to source. The reaction proceeds under mild conditions, typically between 20°C and 60°C, without the requirement for inert gas protection or extreme temperature controls that characterize older technologies. By shifting to this nucleophilic substitution pathway, the process avoids the generation of fluorine-containing byproducts, thereby simplifying the downstream purification process to simple aqueous washing and alcohol recrystallization. This fundamental shift in synthetic strategy not only enhances operator safety but also aligns perfectly with the principles of green chemistry, making it an ideal candidate for cost reduction in fine chemical manufacturing.
Mechanistic Insights into Alkali Metal Salt Mediated Trifluoromethanesulfonylation
The core of this technological breakthrough lies in the unique reactivity of the alkali metal bistrifluoromethanesulfonimide salt acting as a nucleophile towards the chlorobenzene electrophile. Unlike traditional electrophilic trifluoromethanesulfonylation which activates the substrate, this method leverages the stability of the nitrogen-sulfur bond in the salt to transfer the bis(trifluoromethanesulfonyl)imide group efficiently. The reaction mechanism likely involves a nucleophilic aromatic substitution or a metal-catalyzed coupling pathway facilitated by the specific solvent system, such as dichloromethane or acetonitrile. The use of an ice bath during the initial addition of chlorobenzene ensures controlled exothermicity, preventing thermal runaway and maintaining the structural integrity of the sensitive imide functionality. This controlled environment allows for the formation of the C-N bond with high selectivity, minimizing the formation of side products that typically plague high-temperature aromatic substitutions.
From an impurity control perspective, the choice of reactants inherently limits the complexity of the impurity profile. Since the reaction does not involve free fluorine gas or anhydride hydrolysis, the primary impurities are limited to unreacted chlorobenzene and inorganic lithium or sodium salts, both of which are easily removed. The patent details a purification protocol involving multiple water washes to extract the inorganic salts followed by reduced pressure distillation to remove the solvent. The final recrystallization step using alcohol solvents like ethanol or methanol effectively removes trace organic impurities, ensuring the final product meets the stringent purity specifications required for API synthesis. This robust impurity profile is critical for R&D directors who need to ensure that the trifluoromethanesulfonylation reagent does not introduce difficult-to-remove contaminants into the final drug substance.
How to Synthesize N-Phenyl Bis(Trifluoromethanesulfonimide) Efficiently
Implementing this synthesis route requires precise adherence to the reaction parameters outlined in the patent to maximize yield and purity. The process begins with the dissolution of the alkali metal salt in a suitable organic solvent, followed by the controlled addition of chlorobenzene under cooling conditions to manage reaction kinetics. Following the reaction period, the workup procedure is straightforward, relying on phase separation and crystallization rather than complex chromatographic techniques. For detailed operational parameters including specific molar ratios, solvent volumes, and temperature ramps, please refer to the standardized synthesis guide below which encapsulates the critical process steps.
- Dissolve the alkali metal salt (Li, Na, or K) in a solvent like dichloromethane and stir in an ice bath.
- Slowly add chlorobenzene to the reaction mixture and gradually raise the temperature to 20-60°C for 6 hours.
- Wash the reaction mixture with water to remove lithium salts, separate the organic phase, and distill off the solvent.
- Purify the crude product by washing with an alcohol solvent such as ethanol to obtain the refined final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers compelling strategic advantages that extend far beyond simple unit price reductions. By transitioning away from specialty gases and anhydrides, manufacturers can decouple their production from volatile raw material markets and complex logistics chains associated with hazardous chemical transport. The use of common solvents and stable solid salts means that production can be scaled up rapidly using standard glass-lined or stainless steel reactors found in most multipurpose chemical plants, eliminating the need for capital-intensive infrastructure upgrades. This flexibility ensures a more resilient supply chain capable of responding quickly to fluctuations in demand for high-purity pharmaceutical intermediates without the risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of expensive trifluoromethanesulfonic anhydride and the avoidance of stoichiometric waste generation lead to a drastic simplification of the cost structure. Without the need for specialized gas handling equipment or extensive waste neutralization processes for corrosive acids, the overall operational expenditure is significantly lowered. This efficiency allows for a more competitive pricing model for the final reagent, providing substantial cost savings that can be passed down the value chain to API manufacturers.
- Enhanced Supply Chain Reliability: Sourcing common alkali metal salts and chlorobenzene is far more reliable than securing high-purity fluorine gases which often have limited global suppliers. This diversification of raw material sources reduces the risk of supply disruptions and ensures continuous production capability. Additionally, the mild reaction conditions reduce equipment wear and tear, leading to higher asset availability and more consistent delivery schedules for customers relying on just-in-time inventory models.
- Scalability and Environmental Compliance: The process is inherently scalable from laboratory benchtop to multi-ton commercial production without encountering the heat transfer or mass transfer limitations typical of gas-liquid reactions. Furthermore, the absence of fluorine-containing byproducts and the use of recyclable alcohol solvents for purification greatly simplify environmental compliance and waste disposal. This alignment with green chemistry principles reduces the regulatory burden and facilitates smoother permitting for capacity expansion in regions with strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this cost-saving preparation method. These insights are derived directly from the experimental data and technical disclosures within the patent documentation to provide clarity on process feasibility. Understanding these details is essential for technical teams evaluating the integration of this reagent into their existing synthetic workflows.
Q: What are the primary safety advantages of this new synthesis method compared to traditional routes?
A: Unlike traditional methods that utilize hazardous trifluoromethanesulfonyl fluoride gas or generate corrosive hydrogen fluoride byproducts, this novel process utilizes stable solid salts and chlorobenzene, eliminating the need for specialized high-pressure gas handling equipment and significantly reducing operator risk.
Q: How does this process impact the cost structure of N-phenyl bis(trifluoromethanesulfonimide) production?
A: The process replaces expensive trifluoromethanesulfonic anhydride with common, low-cost alkali metal salts and chlorobenzene. Furthermore, the atom economy is improved by avoiding the generation of stoichiometric amounts of trifluoromethanesulfonic acid waste, leading to substantial reductions in raw material and waste disposal costs.
Q: What purity levels can be achieved using this preparation method?
A: Experimental data from the patent indicates that through simple aqueous washing and alcohol recrystallization, the refined product achieves purity levels exceeding 99%, which meets the stringent specifications required for high-value pharmaceutical intermediate applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Phenyl Bis(Trifluoromethanesulfonimide) Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality trifluoromethanesulfonylation reagents play in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify every batch. By leveraging the innovative synthesis route described in CN115028557A, we can offer a superior balance of cost-efficiency and quality assurance that meets the exacting standards of the global pharmaceutical industry.
We invite you to engage with our technical procurement team to discuss how this optimized manufacturing process can benefit your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new drug candidates, we are ready to provide the support you need. Contact us today to request a quotation and discover how our advanced capabilities can accelerate your time to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
