Scalable Electrochemical Production of 2,4-Dimethylanisole: A Cost-Effective Route for Global Supply Chains
The landscape of fine chemical manufacturing is undergoing a profound transformation driven by the urgent need for greener, more sustainable synthetic pathways. A pivotal development in this arena is documented in Chinese Patent CN102337555A, which discloses a novel method for synthesizing 2,4-dimethylanisole, a valuable intermediate widely utilized in the flavor, fragrance, and fuel additive industries. This patent introduces a groundbreaking electrochemical catalytic oxidation technology that fundamentally shifts the paradigm from traditional, hazard-prone chemical alkylation to a cleaner, electricity-driven process. By leveraging a sophisticated reaction system composed of flat graphite electrodes, m-xylene, and methanol, alongside a proprietary solid acid catalyst derived from zirconium oxychloride, this invention offers a compelling solution to long-standing inefficiencies in aromatic ether synthesis. For R&D directors and procurement strategists alike, this technology represents not merely a laboratory curiosity but a viable, scalable alternative that promises to enhance purity profiles while drastically simplifying the operational complexity associated with conventional methylation reactions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted anisoles such as 2,4-dimethylanisole has relied heavily on classical Williamson ether synthesis or acid-catalyzed alkylation using aggressive reagents. As illustrated in prior art, such as the work by Bates and Siahaan published in the Journal of Organic Chemistry in the mid-1980s, traditional routes often involve the methylation of phenolic precursors using dimethyl sulfate or methyl iodide. These conventional methodologies are fraught with significant drawbacks that hinder modern commercial viability. Firstly, the use of dimethyl sulfate poses severe health and safety risks due to its high toxicity and carcinogenicity, necessitating expensive containment systems and rigorous waste treatment protocols that inflate operational expenditures. Secondly, these reactions frequently suffer from poor regioselectivity and low yields, often requiring prolonged reaction times and complex purification steps to isolate the desired 2,4-isomer from a mixture of ortho-, meta-, and para-substituted byproducts. The reliance on stoichiometric amounts of hazardous alkylating agents also results in poor atom economy, generating substantial quantities of inorganic salt waste that complicates environmental compliance and disposal logistics.
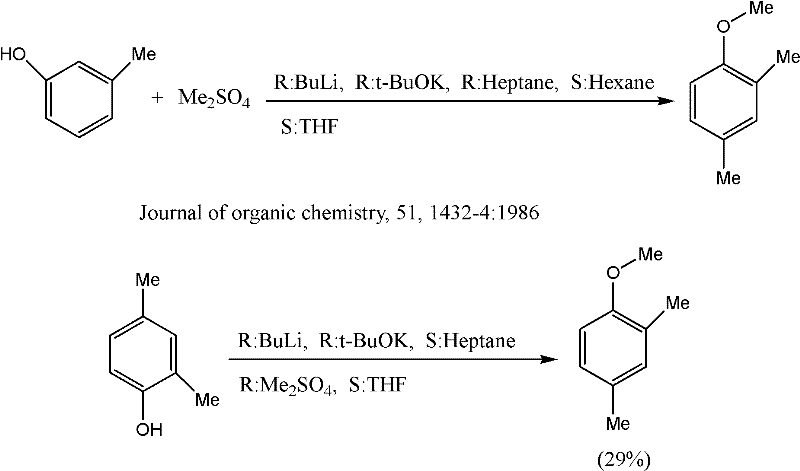
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN102337555A pioneers a direct electrochemical coupling of m-xylene and methanol, bypassing the need for toxic alkylating agents entirely. This innovative approach utilizes an electrochemical cell where m-xylene and methanol serve as both reactants and solvent media, activated by a specialized SO4(2-)/ZrO2 solid acid catalyst under the influence of a direct electric current. The reaction proceeds under remarkably mild conditions, typically at ambient temperature (298K) and atmospheric pressure (101325Pa), which significantly reduces the energy footprint compared to high-temperature thermal processes. By employing plate graphite electrodes and optimizing the catalyst composition—potentially modified with transition metals like molybdenum or copper—the process achieves high selectivity and yield, reportedly reaching up to 62.2% in optimized scenarios. This direct oxidative methylation not only streamlines the synthetic route by eliminating multiple protection-deprotection steps but also aligns perfectly with the principles of green chemistry by utilizing methanol, a readily available and inexpensive C1 building block, as the methyl source.
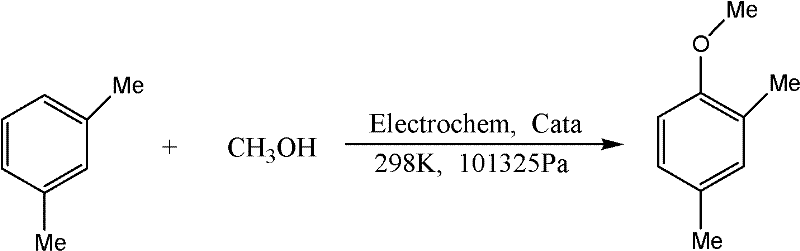
Mechanistic Insights into Electrochemical Catalytic Oxidation
The core of this technological breakthrough lies in the synergistic interaction between the electrochemical field and the superacidic properties of the sulfated zirconia catalyst. The SO4(2-)/ZrO2 catalyst, prepared through a meticulous process of hydrolysis, sulfation, and high-temperature calcination, possesses strong Lewis and Brønsted acid sites that are crucial for activating the methanol molecule. In the presence of the electric field, the methanol is likely oxidized at the anode to generate reactive methylating species, such as methoxy radicals or carbocation intermediates, which are then stabilized by the catalyst surface. Simultaneously, the electron-rich aromatic ring of m-xylene undergoes electrophilic attack at the para-position relative to the existing methyl group, driven by the electronic directing effects and the specific adsorption characteristics of the catalyst. The addition of supporting electrolytes like potassium monofluoride further enhances the conductivity of the reaction medium, ensuring efficient charge transfer and minimizing side reactions such as over-oxidation or polymerization. This precise control over the reaction trajectory allows for the selective formation of the 2,4-dimethylanisole isomer, effectively suppressing the formation of unwanted 2,5- or 3,4-isomers that typically plague non-catalytic thermal methods.
Furthermore, the impurity profile of the final product is significantly improved due to the heterogeneous nature of the catalytic system. Unlike homogeneous acid catalysts which can lead to extensive tar formation and difficult-to-remove residues, the solid acid catalyst can be physically separated from the reaction mixture via simple filtration upon completion. This ease of separation means that downstream purification, primarily achieved through rectification to collect the 190-193°C fraction, is far more efficient and yields a product of exceptional purity. The ability to tune the catalyst properties by doping with metal nitrates (such as ammonium molybdate or copper nitrate) during the preparation phase offers an additional layer of process control, allowing manufacturers to optimize the catalyst's surface area and acidity for specific batch requirements. This mechanistic robustness ensures consistent quality across different production runs, a critical factor for pharmaceutical and flavor applications where trace impurities can have significant sensory or biological impacts.
How to Synthesize 2,4-Dimethylanisole Efficiently
The practical implementation of this electrochemical synthesis route is designed to be straightforward and adaptable to existing chemical infrastructure. The process begins with the in-house preparation of the solid acid catalyst, involving the precipitation of zirconium hydroxide followed by sulfation and calcination, ensuring a fresh and highly active catalytic surface for each campaign. Once the catalyst is prepared, it is introduced into an electrochemical reactor along with m-xylene and methanol in a molar ratio of approximately 1:2. The reaction is initiated by applying a controlled DC voltage of less than 30V and a current below 3A, with continuous magnetic stirring to maintain a uniform slurry of the catalyst within the liquid phase. After a reaction period of roughly 3 hours, the mixture is filtered to recover the catalyst, washed with water and sodium bicarbonate to remove acidic residues, dried, and finally distilled to isolate the pure 2,4-dimethylanisole product. For a comprehensive, step-by-step technical guide including specific reagent grades and equipment specifications, please refer to the standardized protocol below.
- Preparation of the SO4(2-)/ZrO2 solid acid catalyst by precipitating Zr(OH)4 from ZrOCl2, followed by sulfuric acid impregnation and calcination at over 500°C.
- Assembly of the electrochemical reactor using plate graphite electrodes, adding m-xylene, methanol, the prepared catalyst, and potassium monofluoride as the electrolyte support.
- Application of direct current voltage below 30V and current below 3A at room temperature for approximately 3 hours, followed by filtration, washing, and distillation to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical synthesis method offers transformative economic and logistical benefits that extend far beyond simple yield improvements. By shifting the raw material base from specialized, hazardous alkylating agents to commodity chemicals like m-xylene and methanol, manufacturers can achieve substantial cost reduction in fine chemical intermediates manufacturing. Methanol is one of the most produced organic chemicals globally, ensuring a stable and resilient supply chain that is immune to the volatility often seen in niche reagent markets. Furthermore, the elimination of toxic dimethyl sulfate removes the need for costly safety infrastructure, specialized storage facilities, and expensive hazardous waste disposal services, thereby drastically lowering the total cost of ownership for the production facility. The mild operating conditions also translate to reduced energy consumption for heating and cooling, contributing to a lower carbon footprint and aligning with increasingly stringent corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic argument for this technology is compelling, primarily driven by the substitution of expensive and regulated reagents with low-cost bulk commodities. Traditional methylation routes incur high costs not only from the reagents themselves but also from the extensive safety measures required to handle carcinogenic substances. By utilizing an electrochemical cell with graphite electrodes and a reusable solid catalyst, the process minimizes consumable costs and maximizes resource efficiency. The atom economy of directly coupling m-xylene with methanol is inherently superior to substitution reactions that generate stoichiometric salt byproducts, meaning less raw material is wasted. Additionally, the simplified workup procedure—filtration followed by distillation—reduces labor hours and solvent usage during purification, further driving down the variable costs per kilogram of finished product.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on m-xylene and methanol, both of which are produced on a massive petrochemical scale with well-established global distribution networks. Unlike specialized phenolic starting materials or sensitive alkylating agents that may face supply disruptions or long lead times, these feedstocks are readily available from multiple suppliers worldwide. This diversification of supply sources mitigates the risk of production stoppages due to raw material shortages. Moreover, the robustness of the solid acid catalyst, which can be prepared in-house from zirconium salts, reduces dependency on external catalyst vendors. The ability to operate at ambient pressure and temperature also simplifies equipment requirements, allowing for production in a wider range of facilities without the need for high-pressure reactors, thus increasing overall manufacturing flexibility and capacity.
- Scalability and Environmental Compliance: Scaling this electrochemical process from pilot to commercial production is facilitated by the modular nature of electrochemical cells, which can be expanded by increasing electrode surface area or numbering up reactors. The environmental profile of the process is exceptionally clean, generating minimal hazardous waste compared to traditional methods. The absence of heavy metal catalysts (using instead zirconium-based solids) and toxic alkylating agents simplifies wastewater treatment and ensures compliance with rigorous environmental regulations such as REACH or TSCA. The solid catalyst can potentially be regenerated and reused multiple times, further reducing the volume of solid waste generated. This eco-friendly profile not only reduces regulatory burden but also enhances the marketability of the final product to end-users in the food and fragrance industries who prioritize sustainably sourced ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrochemical synthesis of 2,4-dimethylanisole, based on the detailed specifications and experimental data provided in the patent literature. These insights are intended to clarify the feasibility and advantages of implementing this technology within your existing manufacturing framework. Understanding these nuances is essential for making informed decisions about process adoption and supplier qualification.
Q: What are the primary advantages of the electrochemical method over traditional alkylation for 2,4-dimethylanisole?
A: The electrochemical method described in CN102337555A eliminates the need for hazardous alkylating agents like dimethyl sulfate, operates under mild room temperature and pressure conditions, and utilizes abundant raw materials like m-xylene and methanol, resulting in a safer and more atom-economical process.
Q: How does the sulfated zirconia catalyst contribute to the reaction efficiency?
A: The SO4(2-)/ZrO2 solid acid catalyst provides strong acidic sites that facilitate the activation of methanol and the subsequent electrophilic substitution on the m-xylene ring, while the electrochemical environment further enhances the oxidation potential, leading to improved selectivity and yield compared to thermal methods.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is designed for scalability, utilizing simple graphite electrodes and standard electrolytic cell configurations. The use of a heterogeneous solid acid catalyst allows for easier separation and potential recycling, which are critical factors for reducing operational costs in commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Dimethylanisole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to maintain competitiveness in the global fine chemicals market. Our team of expert chemists has thoroughly analyzed the electrochemical pathway described in CN102337555A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative route to life. We are committed to delivering high-purity 2,4-dimethylanisole that meets the most stringent purity specifications, utilizing our rigorous QC labs to ensure every batch is free from the impurities associated with older synthetic methods. Our state-of-the-art facilities are equipped to handle electrochemical processes safely and efficiently, ensuring a consistent supply of this valuable intermediate for your flavor, fragrance, or agrochemical applications.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this greener synthetic route for your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data for our electrochemically synthesized batches and to discuss detailed route feasibility assessments tailored to your supply chain objectives. Let us help you optimize your sourcing strategy with a reliable, cost-effective, and sustainable solution for 2,4-dimethylanisole.
