Revolutionizing 2,3-Diaminoindole Production: A Metal-Free Electrochemical Strategy for Global Supply Chains
The pharmaceutical industry is currently witnessing a paradigm shift towards sustainable and efficient synthetic methodologies, particularly for complex heterocyclic scaffolds like indoles. Patent CN114656395A introduces a groundbreaking electrochemical strategy for the synthesis of 2,3-diaminoindole compounds, addressing critical pain points in modern drug discovery and process chemistry. This innovative approach leverages constant current electrolysis to drive the oxidative cyclization of o-ethynylanilines and arylamines, completely bypassing the need for traditional transition metal catalysts. By utilizing electricity as a traceless reagent, this method not only aligns with green chemistry principles but also resolves the persistent issue of metal residue contamination that plagues conventional catalytic cycles. For R&D directors and process chemists, this represents a significant advancement in accessing high-purity intermediates essential for the development of next-generation therapeutics and bioactive alkaloids.
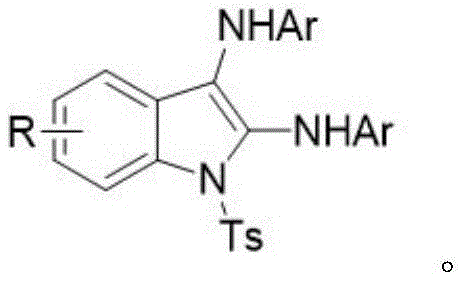
The limitations of conventional synthetic routes for aminoindoles are well-documented and pose substantial challenges for large-scale manufacturing. Historically, the construction of the indole core functionalized with amino groups has relied heavily on copper(II)-catalyzed hydroxylamination or rhodium-mediated intermolecular oxyamidation reactions. These traditional pathways invariably require expensive noble or transition metals, which not only inflate the raw material costs but also introduce severe downstream purification burdens. The removal of trace metal impurities to meet stringent pharmaceutical standards often necessitates additional scavenging steps, resulting in yield erosion and extended production timelines. Furthermore, these thermal processes typically demand elevated reaction temperatures, which can compromise the stability of sensitive functional groups and limit the substrate scope to only robust molecules. The reliance on stoichiometric chemical oxidants in these methods further exacerbates waste generation, creating environmental liabilities that modern supply chains strive to minimize.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Existing methodologies for constructing the 2,3-diaminoindole scaffold are fraught with inefficiencies that hinder their economic viability for commercial production. The dependence on catalysts such as copper or rhodium creates a bottleneck in the supply chain, as the availability and price volatility of these metals can disrupt manufacturing schedules. Beyond the cost implications, the presence of metal residues in the final active pharmaceutical ingredient (API) is a regulatory red flag, requiring rigorous analytical testing and potentially costly remediation protocols. High-temperature conditions associated with these thermal reactions also increase energy consumption and safety risks, particularly when dealing with potentially unstable intermediates or exothermic processes. Additionally, the use of harsh chemical oxidants generates significant amounts of toxic byproducts, complicating waste treatment and increasing the overall environmental footprint of the synthesis. These cumulative factors render conventional methods less attractive for companies aiming to optimize their cost of goods sold (COGS) and enhance their sustainability profiles.
The Novel Approach
In stark contrast, the electrochemical method disclosed in the patent offers a transformative solution by replacing chemical oxidants and metal catalysts with electrical energy. This metal-free protocol operates under remarkably mild conditions, specifically at room temperature, which preserves the integrity of diverse functional groups on the substrate. The reaction utilizes a simple undivided cell setup with a platinum anode and a carbon cathode, facilitating the generation of reactive radical species necessary for the cyclization without introducing foreign metallic elements. The solvent system, comprising a mixture of water and acetonitrile, is not only cost-effective but also reduces the reliance on volatile organic compounds (VOCs). This approach fundamentally simplifies the reaction profile, allowing for a broader substrate scope that includes complex natural product derivatives which might decompose under harsher thermal conditions. The elimination of metal catalysts ensures that the resulting 2,3-diaminoindole products are inherently free from heavy metal contamination, streamlining the quality control process.
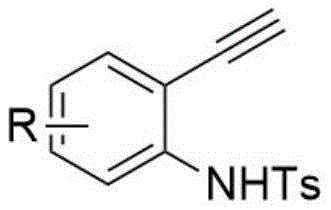
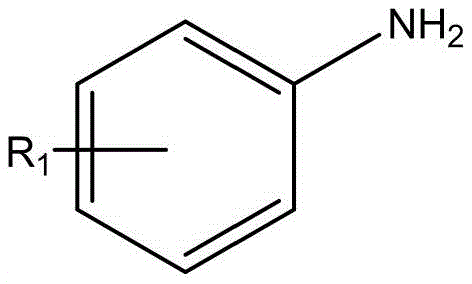
Mechanistic Insights into Electrochemical Oxidative Cyclization
The mechanistic pathway of this electrochemical transformation is driven by anodic oxidation, which initiates a radical cascade leading to the formation of the indole ring. In the presence of the electrolyte lithium perchlorate and the additive tetrabutylammonium iodide, the application of a constant current facilitates the generation of iodine radicals or cationic species at the anode surface. These active species interact with the tetramethylpiperidine oxide (TEMPO) mediator to abstract hydrogen atoms or activate the alkyne moiety of the o-ethynylaniline substrate. This activation triggers a nucleophilic attack by the arylamine, followed by an intramolecular cyclization that constructs the five-membered pyrrole ring characteristic of the indole core. The precise control over the current density allows for the fine-tuning of the reaction rate, preventing over-oxidation and ensuring high selectivity for the desired 2,3-diamino product. This radical-mediated mechanism avoids the formation of metal-complex intermediates, thereby sidestepping the kinetic traps and side reactions often associated with transition metal catalysis.
From an impurity control perspective, the absence of metal catalysts significantly simplifies the impurity profile of the crude reaction mixture. In traditional metal-catalyzed reactions, side products often arise from metal-ligand interactions or incomplete catalyst turnover, leading to complex mixtures that are difficult to separate. The electrochemical method primarily generates byproducts derived from the solvent or minor over-oxidation of the amine, which are typically more polar and easier to remove during the aqueous workup. The use of a water-acetonitrile solvent system further aids in partitioning non-polar impurities away from the target molecule during extraction. Consequently, the purification process via silica gel column chromatography becomes more efficient, yielding products with higher purity levels suitable for direct use in subsequent coupling reactions. This clean reaction profile is particularly advantageous for the synthesis of pharmaceutical intermediates where strict limits on genotoxic impurities and heavy metals are enforced by global regulatory agencies.
How to Synthesize 2,3-Diaminoindole Efficiently
The practical execution of this synthesis is designed for operational simplicity, making it accessible for both laboratory-scale optimization and pilot-plant operations. The procedure involves charging a reaction vessel with the o-ethynylaniline and arylamine substrates in a specific molar ratio, typically ranging from 1:3 to 1:4 to ensure complete conversion of the limiting reagent. Supporting electrolytes and mediators such as lithium perchlorate, tetrabutylammonium iodide, and TEMPO are added to facilitate charge transfer and radical propagation. The reaction is conducted under a constant current regime, typically around 5 mA for small-scale preparations, for a duration of 8 to 12 hours at ambient temperature. Following the electrolysis, the reaction mixture undergoes a straightforward workup involving extraction with ethyl acetate, concentration under reduced pressure, and final purification. Detailed standardized operating procedures for scaling this electrochemical process are outlined below.
- Prepare the reaction mixture by combining o-ethynylaniline, arylamine, lithium perchlorate, tetrabutylammonium iodide, and TEMPO in a water-acetonitrile solvent system.
- Set up the electrochemical cell with a platinum anode and carbon cathode, applying a constant current at room temperature for 8-12 hours.
- Upon completion, extract the reaction mixture with ethyl acetate, concentrate via rotary evaporation, and purify the crude product using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this electrochemical technology presents a compelling value proposition centered on cost efficiency and supply reliability. The most immediate impact is the drastic reduction in raw material costs associated with the elimination of precious metal catalysts like palladium, rhodium, or copper salts, which are subject to significant market price fluctuations. By removing these expensive inputs, the overall cost of goods sold (COGS) for the intermediate is substantially lowered, improving margin potential for downstream API manufacturers. Furthermore, the simplified purification workflow reduces the consumption of silica gel and solvents, contributing to additional operational savings. The mild reaction conditions also translate to lower energy expenditures, as there is no need for heating mantles or cryogenic cooling systems, aligning with corporate sustainability goals and reducing the carbon footprint of the manufacturing process.
- Cost Reduction in Manufacturing: The exclusion of transition metal catalysts removes the necessity for expensive metal scavengers and rigorous purification steps designed to meet residual metal specifications. This qualitative shift in the process design leads to significant cost savings by shortening the production cycle and reducing the volume of waste generated. The use of inexpensive and abundant reagents like lithium perchlorate and iodide salts further stabilizes the input costs, shielding the supply chain from the volatility associated with critical raw materials. Additionally, the high atom economy of the electrochemical approach ensures that a larger proportion of the starting materials are converted into the desired product, minimizing material loss.
- Enhanced Supply Chain Reliability: The reliance on electricity as the primary reagent decouples the synthesis from the supply constraints of specialized chemical oxidants or catalysts that may face logistical bottlenecks. Since the reaction proceeds at room temperature, the safety profile is enhanced, reducing the risk of shutdowns due to thermal runaway incidents or hazardous material handling violations. The robustness of the method across a wide range of substrates means that a single production line can be adapted to manufacture various derivatives of 2,3-diaminoindoles, increasing asset utilization and flexibility. This adaptability ensures a continuous supply of high-quality intermediates even when specific precursor availability fluctuates.
- Scalability and Environmental Compliance: Electrochemical synthesis is inherently scalable through the implementation of flow reactors or parallel plate cells, allowing for seamless transition from gram-scale R&D to tonnage-level commercial production. The aqueous solvent component reduces the load on solvent recovery systems and minimizes the emission of volatile organic compounds, facilitating compliance with increasingly stringent environmental regulations. The absence of heavy metal waste streams simplifies effluent treatment, lowering the costs and complexities associated with waste disposal. This environmentally friendly profile enhances the company's reputation and meets the growing demand from partners for sustainable and responsible sourcing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis method. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of the technology. Understanding these aspects is crucial for stakeholders evaluating the integration of this route into their existing manufacturing portfolios.
Q: Why is the electrochemical method superior to traditional copper-catalyzed synthesis for indoles?
A: Traditional methods rely on expensive transition metals like copper or rhodium and often require high temperatures, leading to metal residue issues. The electrochemical approach operates at room temperature without metal catalysts, fundamentally eliminating heavy metal contamination risks critical for pharmaceutical applications.
Q: What are the key safety advantages of this synthetic route?
A: The process utilizes mild reaction conditions at room temperature and avoids hazardous chemical oxidants by using electricity as the driving force. The solvent system of water and acetonitrile is relatively benign compared to harsh organic solvents often required in thermal cyclizations.
Q: Can this method be scaled for industrial production of API intermediates?
A: Yes, electrochemical synthesis is inherently scalable through flow chemistry or increased electrode surface area. The simplicity of the workup, involving standard extraction and chromatography, supports robust commercial scale-up without complex purification bottlenecks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Diaminoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in delivering high-value pharmaceutical intermediates with unparalleled purity and efficiency. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this metal-free technology are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2,3-diaminoindole meets the exacting standards required for global drug development. Our commitment to innovation allows us to offer customized solutions that optimize both the technical performance and the economic viability of your supply chain.
We invite you to collaborate with us to leverage this advanced synthetic route for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to reach out for specific COA data and route feasibility assessments to understand how this electrochemical method can enhance your product portfolio. Let us help you navigate the complexities of modern chemical manufacturing with a partner dedicated to excellence and sustainability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
