Advanced Manufacturing of Sutent Intermediate: A Technical Breakdown for Global Supply Chains
Advanced Manufacturing of Sutent Intermediate: A Technical Breakdown for Global Supply Chains
The pharmaceutical landscape for oncology treatments continues to evolve, with Sunitinib (Sutent) remaining a cornerstone therapy for gastrointestinal stromal tumors and advanced renal cell carcinoma. At the heart of this therapeutic value chain lies the critical intermediate, 2,4-dimethyl-5-formyl-1H-pyrrole-3-carboxylic acid (CAS 253870-02-9). Recent intellectual property disclosures, specifically patent CN101759625A, have illuminated a refined synthetic pathway that addresses long-standing manufacturing bottlenecks. This technical report analyzes the disclosed methodology, focusing on its implications for process chemistry optimization, cost efficiency, and supply chain resilience. By shifting away from hazardous reagents and high-pressure conditions, this novel approach offers a compelling alternative for reliable pharmaceutical intermediates supplier networks seeking to enhance their production capabilities while adhering to stringent environmental and safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrrole-based intermediates for kinase inhibitors has relied heavily on classical formylation strategies such as the Vilsmeier-Haack reaction or the Gattermann-Koch reaction. The Vilsmeier-Haack protocol, while chemically effective, necessitates the use of phosphorus oxychloride (POCl3), a reagent notorious for its severe corrosivity and the generation of substantial acidic waste streams. Furthermore, achieving the desired regioselectivity often requires a precursor like 2,4-dimethyl-3,5-diethoxycarbonyl-1H-pyrrole, which must undergo selective decarboxylation. This additional step introduces complexity, as controlling mono-decarboxylation versus di-decarboxylation is challenging, frequently leading to difficult-to-separate byproducts that compromise the purity profile essential for high-purity pharmaceutical intermediates. Similarly, the Gattermann-Koch approach demands high-pressure carbon monoxide environments and specialized catalytic systems, creating significant barriers to safe commercial scale-up of complex pharmaceutical intermediates in standard multipurpose plants.
The Novel Approach
In stark contrast, the methodology outlined in patent CN101759625A presents a streamlined route that circumvents these hazardous and operationally intensive steps. The innovation lies in the construction of the pyrrole core via a modified Knorr condensation using readily accessible beta-keto esters, followed by a mild acidolytic formylation. Instead of corrosive POCl3, the process utilizes trifluoroacetic acid (TFA) and triethyl orthoformate under atmospheric pressure and near-ambient temperatures. This shift not only mitigates the risk of equipment corrosion but also simplifies the downstream processing, as the reaction mixture can be directly quenched into water to precipitate the product. The result is a process characterized by robust operational parameters, where intermediates exist as filterable solids, drastically reducing lead time for high-purity pharmaceutical intermediates by eliminating tedious extraction and distillation sequences typically associated with liquid reaction masses.
Mechanistic Insights into Modified Knorr Condensation and Acidolytic Formylation
The synthetic strategy initiates with the nitrosation of tert-butyl acetoacetate using sodium nitrite to form an oxime intermediate, which is subsequently reduced in situ using zinc powder. This generates a reactive amino-ketone species that immediately undergoes condensation with ethyl acetoacetate. This Knorr-type cyclization is pivotal, as it constructs the 2,4-dimethyl-1H-pyrrole scaffold with high regiocontrol, installing the necessary ester functionality at the 3-position and a tert-butyl ester at the 5-position. The choice of the tert-butyl protecting group is strategic; it is stable during the cyclization but labile under the acidic conditions of the subsequent step. Following the formation of the pyrrole ring, the introduction of the formyl group at the 5-position is achieved through an acid-catalyzed reaction with triethyl orthoformate. The trifluoroacetic acid serves a dual purpose: it cleaves the tert-butyl ester to reveal the carboxylic acid (which is later re-esterified or managed) and activates the orthoformate for electrophilic aromatic substitution, ensuring the aldehyde is installed precisely at the electron-rich 5-position of the pyrrole ring.
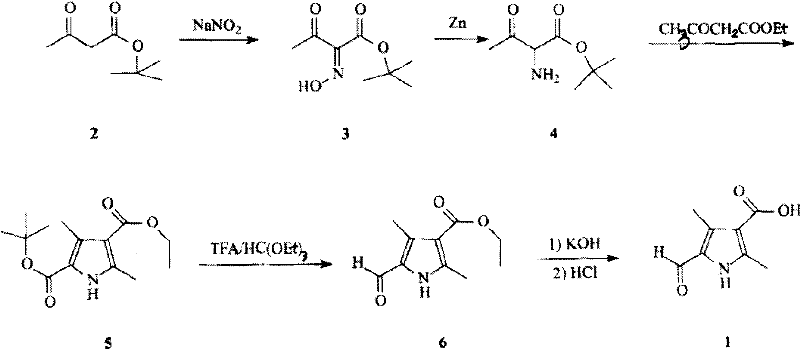
Following the formylation, the final transformation involves the hydrolysis of the ethyl ester to yield the target carboxylic acid. The patent data indicates that the intermediate ethyl ester (Compound 6) is obtained as a solid with a melting point of 163–164°C, confirming its high crystallinity and ease of isolation. The final hydrolysis using potassium hydroxide proceeds rapidly, with the patent noting completion within approximately 40 minutes under reflux. Acidification of the resulting salt precipitates the final product, 2,4-dimethyl-5-formyl-1H-pyrrole-3-carboxylic acid, as an off-white solid with a high melting point of 276–277°C. This thermal stability and solid-state nature throughout the synthesis are critical quality attributes, as they allow for rigorous purification via recrystallization, ensuring the impurity profile meets the exacting standards required for oncology drug substances. The overall yield of approximately 44 percent across the sequence is competitive, particularly when weighed against the reduced operational costs and safety hazards.
How to Synthesize 2,4-dimethyl-5-formyl-1H-pyrrole-3-carboxylic acid Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the nitrosation and reduction steps, as well as temperature control during the formylation to prevent polymerization of the sensitive pyrrole aldehyde. The process is designed to be telescoped where possible, minimizing the isolation of unstable intermediates. For process chemists looking to adopt this route, the key lies in the efficient work-up of the zinc reduction step and the precise quenching of the TFA reaction to maximize recovery of the solid intermediates. The detailed standardized synthesis steps, including specific solvent volumes, addition rates, and purification protocols, are outlined below to ensure reproducibility and safety in your pilot or production facilities.
- Perform nitrosation on tert-butyl acetoacetate using sodium nitrite to generate the oxime intermediate.
- Reduce the oxime with zinc powder and condense with ethyl acetoacetate to form the pyrrole ring structure.
- Execute acidolysis and formylation using trifluoroacetic acid and triethyl orthoformate, followed by alkaline hydrolysis to yield the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical yield. The elimination of phosphorus oxychloride removes a major logistical and safety burden, as this reagent requires specialized storage and handling protocols due to its reactivity with moisture and potential for releasing toxic fumes. By replacing it with trifluoroacetic acid and triethyl orthoformate, the manufacturing process becomes significantly more compatible with standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. This compatibility translates directly into cost reduction in pharmaceutical intermediates manufacturing, as it avoids the need for exotic metallurgy or frequent reactor replacements caused by corrosion. Furthermore, the avoidance of high-pressure carbon monoxide cylinders simplifies the site infrastructure requirements, allowing for production in a wider range of facilities without extensive capital investment in pressure-rated equipment.
- Cost Reduction in Manufacturing: The economic viability of this process is bolstered by the use of commodity chemicals such as zinc powder, sodium nitrite, and ethyl acetoacetate, which are globally available and price-stable. The fact that the intermediates precipitate as solids allows for isolation via simple filtration rather than energy-intensive distillation or solvent-heavy extractions. This reduction in unit operations lowers both utility consumption (steam and electricity) and solvent purchase costs. Additionally, the high purity of the solid intermediates reduces the need for costly chromatographic purification, which is often a bottleneck in terms of both time and silica gel consumption. The cumulative effect is a leaner manufacturing process with a lower cost of goods sold (COGS), providing a buffer against raw material price fluctuations.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on specialized or regulated reagents. By utilizing a route based on widely available bulk chemicals, the risk of supply disruption is minimized. The robustness of the reaction conditions—operating at atmospheric pressure and moderate temperatures—means that production is less susceptible to delays caused by equipment maintenance or safety inspections associated with high-pressure or highly corrosive processes. The solid nature of the intermediates also facilitates easier storage and transportation between different production sites if a multi-site manufacturing strategy is employed. This flexibility ensures that reliable pharmaceutical intermediates supplier networks can maintain consistent inventory levels, safeguarding the downstream production of the final API against upstream volatility.
- Scalability and Environmental Compliance: As regulatory scrutiny on pharmaceutical manufacturing waste intensifies, the environmental profile of a synthesis route becomes a critical selection criterion. This method generates significantly less hazardous waste compared to the Vilsmeier-Haack alternative, primarily by avoiding phosphate waste streams. The aqueous work-ups described in the patent are straightforward, and the zinc salts generated can be managed through standard wastewater treatment protocols. The scalability is further evidenced by the exothermic nature of the reactions being manageable; the patent describes additions over specific timeframes to control heat release, a parameter that translates well from kilogram to metric-ton scale. This alignment with green chemistry principles not only reduces disposal costs but also enhances the sustainability credentials of the supply chain, a factor increasingly weighted in vendor selection by top-tier pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this Sutent intermediate. These answers are derived directly from the experimental data and process descriptions found in the referenced patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for making informed decisions about process adoption and vendor qualification.
Q: What are the primary safety advantages of this synthesis route over Vilsmeier-Haack?
A: This method eliminates the need for phosphorus oxychloride (POCl3), a highly corrosive reagent used in conventional Vilsmeier-Haack reactions, thereby significantly reducing equipment corrosion risks and hazardous waste handling requirements.
Q: How does the solid state of intermediates impact purification costs?
A: The patent highlights that key intermediates, such as the pyrrole ester, precipitate as solids. This physical property allows for simple filtration and washing processes, avoiding energy-intensive distillation or complex chromatography typically required for liquid intermediates.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process operates under atmospheric pressure and moderate temperatures without requiring specialized high-pressure reactors. The use of readily available raw materials and straightforward work-up procedures makes it highly adaptable for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-dimethyl-5-formyl-1H-pyrrole-3-carboxylic acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics observed in patent literature are realized in actual manufacturing campaigns. We understand that for oncology intermediates, consistency is paramount. Therefore, our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, utilizing advanced analytical techniques to monitor impurity profiles at every stage of the synthesis. Whether you require custom synthesis services or a steady supply of this critical pyrrole building block, our infrastructure is designed to support your clinical and commercial needs with precision.
We invite you to leverage our technical expertise to optimize your supply chain for Sunitinib production. Our engineering team is prepared to conduct a Customized Cost-Saving Analysis specific to your volume requirements, identifying further opportunities for efficiency within this synthetic route. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. By collaborating with us, you gain access to a supply partner dedicated to delivering high-quality intermediates that drive the success of your final pharmaceutical products.
