Scalable Synthesis of Chiral (-)-5-Azaspiro[2.4]heptane-7-alcohol for Advanced Oncology Intermediates
Scalable Synthesis of Chiral (-)-5-Azaspiro[2.4]heptane-7-alcohol for Advanced Oncology Intermediates
The pharmaceutical industry is constantly seeking robust, scalable pathways for complex chiral intermediates, particularly those serving as the backbone for next-generation oncology therapeutics. Patent CN113072480A introduces a groundbreaking preparation method for chiral (-)-5-azaspiro[2.4]heptane-7-alcohol, a critical building block for the synthesis of the single-isomer drug (-)-AL8326. This tyrosine kinase inhibitor has demonstrated broad-spectrum antitumor activity against ovarian, leukemia, and liver cancer cells, yet its clinical potential has been hindered by the challenges of isolating the active levorotatory configuration. The disclosed technology overcomes historical bottlenecks by utilizing a sophisticated chemical resolution strategy that leverages the hydroxyl group on the chiral carbon, followed by a mild catalytic debenzylation. This approach not only ensures exceptional optical purity but also establishes a foundation for the targeted clinical application of (-)-AL8326, marking a significant leap forward in the commercial viability of this high-value pharmaceutical intermediate.
![Chemical structure of chiral (-)-5-azaspiro[2.4]heptane-7-alcohol showing the spirocyclic core and hydroxyl group](/insights/img/chiral-azaspiro-alcohol-synthesis-pharma-supplier-20260308030538-01.png)
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of high-purity chiral azaspiro compounds has been plagued by inefficient resolution techniques and unstable reaction pathways. Traditional biological enzyme reduction methods, while theoretically elegant, often fail in practical industrial settings due to substrate instability. As illustrated in comparative studies within the patent data, enzymatic reduction of the corresponding ketone precursor results in the formation of brown viscous substances that increase over time, severely compromising chemical purity to levels as low as 68%. Furthermore, attempts to separate enantiomers using chiral preparative columns have proven ineffective for this specific molecular scaffold, failing to achieve baseline separation even under optimized mobile phase conditions. These limitations create substantial barriers for reliable pharmaceutical intermediate supplier operations, leading to inconsistent batch quality and prohibitive costs associated with low-yield purification processes.
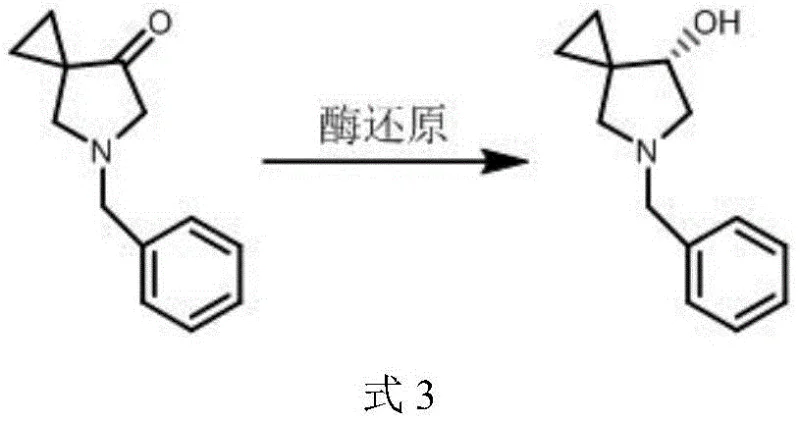
The Novel Approach
In stark contrast to these flawed methodologies, the novel approach detailed in CN113072480A employs a multi-step chemical resolution pathway that is both robust and economically viable. The process begins with the esterification of the racemic alcohol with phthalic anhydride, creating a derivative amenable to resolution. By reacting this benzoate with (-)-alpha-phenylethylamine, the method exploits the drastically different solubilities of the resulting diastereomeric salts in aqueous solutions. This allows for the physical separation of the desired (-)-configuration through simple filtration and recrystallization, bypassing the need for complex chromatography. The subsequent hydrolysis and atmospheric pressure debenzylation complete the transformation, delivering the target molecule with superior purity. This strategic shift from biological or chromatographic dependence to classical chemical resolution represents a paradigm shift in cost reduction in API manufacturing for complex spirocyclic structures.
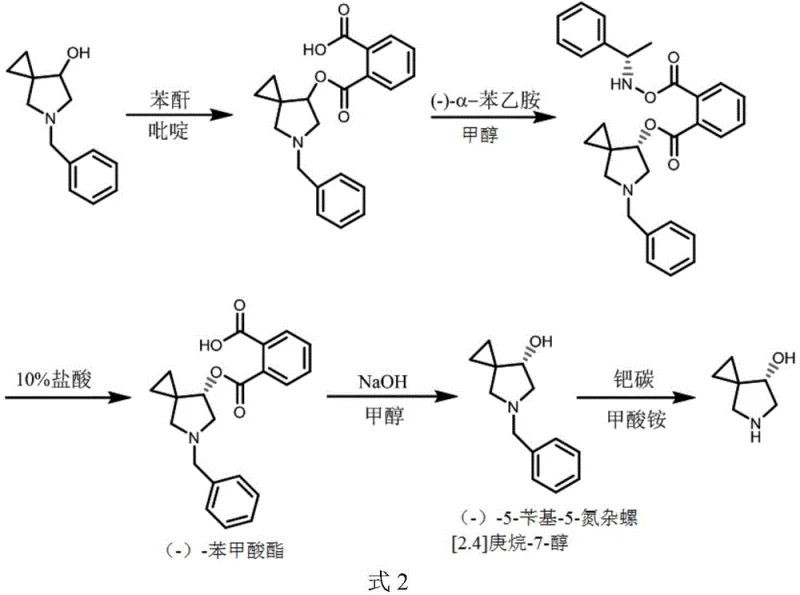
Mechanistic Insights into Chemical Resolution and Transfer Hydrogenation
The core of this technological breakthrough lies in the precise manipulation of stereochemistry through diastereomeric salt formation. Unlike direct resolution of the amine which proved unsuccessful with tartaric acid due to a lack of solid precipitation, the conversion of the hydroxyl group into a phthalate ester introduces a new handle for chiral discrimination. When treated with (-)-alpha-phenylethylamine, the mixture forms two distinct salts. The patent data indicates that the salt corresponding to the desired (-)-enantiomer possesses significantly lower solubility in methanol and water mixtures, precipitating out selectively while the unwanted (+)-enantiomer remains in the mother liquor. This thermodynamic control allows for the enrichment of optical purity to nearly 100% ee after recrystallization. The mechanism ensures that the chiral center at the 7-position is locked and protected during the subsequent harsh chemical treatments, preventing epimerization that often plagues spirocyclic systems.
Furthermore, the final debenzylation step utilizes a transfer hydrogenation mechanism rather than high-pressure hydrogen gas. By employing ammonium formate as the hydrogen donor in the presence of palladium carbon, the reaction proceeds under mild thermal conditions (50-80°C) and atmospheric pressure. Comparative data reveals that high-pressure hydrogenation can induce racemization, likely due to the harsh conditions affecting the strained spiro-ring system or the adjacent nitrogen lone pair. The transfer hydrogenation pathway avoids these high-energy states, ensuring that the optical rotation of the final product remains stable at approximately -75 degrees. This mechanistic nuance is critical for maintaining the stringent purity specifications required for oncology drug substances, where even trace amounts of the wrong isomer can introduce toxicity.
How to Synthesize (-)-5-Azaspiro[2.4]heptane-7-alcohol Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible roadmap for laboratory and pilot-scale production. The process is divided into four distinct operational units: esterification, resolution, hydrolysis, and debenzylation. Each step utilizes commodity chemicals and standard reactor equipment, eliminating the need for specialized bioreactors or high-pressure vessels. The following guide summarizes the critical operational parameters derived from the patent examples, ensuring that technical teams can replicate the high yields and purity profiles reported. For a comprehensive breakdown of the standardized operating procedures, please refer to the detailed synthesis steps provided below.
- Esterify 5-benzyl-5-azaspiro[2.4]heptane-7-alcohol with phthalic anhydride in pyridine at 100°C to form the benzoate derivative.
- Perform chiral resolution using (-)-alpha-phenylethylamine in methanol, followed by filtration and recrystallization to isolate the single-configuration salt.
- Hydrolyze the resolved benzoate with alkaline solution to recover (-)-5-benzyl-5-azaspiro[2.4]heptane-7-alcohol, then remove the benzyl group via catalytic transfer hydrogenation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this chemical resolution methodology offers profound strategic benefits beyond mere technical feasibility. The reliance on bulk commodity chemicals such as phthalic anhydride, pyridine, and alpha-phenylethylamine drastically reduces the raw material cost profile compared to proprietary enzymes or chiral stationary phases. This shift mitigates supply chain risks associated with single-source biological reagents and ensures long-term availability of key inputs. Moreover, the simplification of the purification workflow—from complex chromatography to straightforward filtration and crystallization—significantly accelerates batch cycle times. This operational efficiency translates directly into enhanced supply chain reliability, allowing manufacturers to respond more agilely to the fluctuating demands of the global oncology market without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive chiral columns and unstable biocatalysts removes significant capital and operational expenditures from the production budget. By utilizing a resolution strategy based on solubility differences, the process avoids the high solvent consumption and low throughput inherent in preparative HPLC. Additionally, the use of ammonium formate for debenzylation negates the need for high-pressure hydrogenation infrastructure, further reducing facility safety costs and equipment maintenance requirements. These cumulative factors drive a substantial decrease in the overall cost of goods sold (COGS) for this high-value intermediate.
- Enhanced Supply Chain Reliability: Sourcing stability is paramount for critical API intermediates. This synthetic route relies on widely available organic building blocks that are produced at a global scale, minimizing the risk of raw material shortages. The robustness of the chemical steps ensures consistent batch-to-batch performance, reducing the likelihood of production delays caused by failed enzymatic runs or column degradation. This predictability allows supply chain planners to maintain leaner inventory levels while securing a steady flow of material for downstream drug substance manufacturing, ultimately safeguarding the continuity of the final therapeutic product.
- Scalability and Environmental Compliance: The process is inherently designed for industrial amplification, moving seamlessly from gram-scale optimization to multi-kilogram production. The avoidance of heavy metal catalysts (beyond the recoverable palladium on carbon) and the reduction of hazardous solvent loads align with modern green chemistry principles. The simplicity of the workup procedures, involving standard extractions and filtrations, facilitates easier waste stream management and treatment. This environmental compatibility not only meets rigorous regulatory standards but also future-proofs the manufacturing site against tightening environmental regulations, ensuring sustainable long-term operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of chiral (-)-5-azaspiro[2.4]heptane-7-alcohol. These insights are derived directly from the experimental data and comparative analysis presented in patent CN113072480A, providing clarity on why this specific route is superior to legacy methods. Understanding these nuances is essential for technical teams evaluating process feasibility and for procurement specialists assessing supplier capabilities.
Q: Why is chemical resolution preferred over enzymatic reduction for this specific azaspiro intermediate?
A: According to patent CN113072480A, enzymatic reduction suffers from substrate instability and generates brown viscous impurities over time, resulting in low chemical purity (approx. 68%). In contrast, the chemical resolution method utilizing phthalic anhydride esterification achieves significantly higher chemical and optical purity suitable for industrial GMP production.
Q: How does the novel debenzylation method prevent racemization?
A: The patent demonstrates that traditional high-pressure hydrogenation can lead to racemization (optical rotation dropping to 0). The disclosed method uses ammonium formate as a hydrogen source with palladium carbon at atmospheric pressure and moderate temperatures (50-80°C), which preserves the chiral integrity of the molecule while efficiently removing the benzyl protecting group.
Q: What are the scalability advantages of this resolution technique?
A: This process relies on the distinct solubility differences of diastereomeric phenethylamine salts in aqueous solutions, allowing for separation via simple filtration and recrystallization. This eliminates the need for expensive chiral preparation columns or complex chromatographic separations, making it highly adaptable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (-)-5-Azaspiro[2.4]heptane-7-alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of life-saving oncology therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent literature to industrial reality is seamless. We are equipped with rigorous QC labs and advanced analytical instrumentation capable of verifying stringent purity specifications, including chiral HPLC and NMR analysis, to guarantee that every batch meets the exacting standards required for GMP drug substance synthesis. Our commitment to technical excellence ensures that the complex stereochemical integrity of molecules like (-)-5-azaspiro[2.4]heptane-7-alcohol is preserved throughout the manufacturing lifecycle.
We invite global pharmaceutical partners to collaborate with us to leverage this innovative synthesis technology for your pipeline projects. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific rigor and commercial reliability. Let us help you accelerate the development of (-)-AL8326 and other next-generation therapeutics with our premium intermediate solutions.
