Advanced Aqueous Phase Synthesis of Cephalothin Acid for Scalable API Manufacturing
The pharmaceutical industry is constantly seeking methodologies that balance high-yield synthesis with environmental sustainability and operational safety. Patent CN102040614A introduces a transformative approach to producing cephalothin acid, a critical intermediate for semi-synthetic cephamycin-type antibiotics. This invention discloses a method for synthesizing cephalothin acid using 7-aminocephalosporanic acid (7-ACA) as a raw material, characterized by performing the condensation reaction in a water phase solvent rather than traditional organic media. This shift addresses critical pain points in pharmaceutical intermediate production, specifically regarding environmental impact and operational safety. By utilizing an aqueous system, the process eliminates the need for vast quantities of flammable and volatile organic solvents, thereby fundamentally altering the risk profile of the manufacturing facility. This technical breakthrough offers a robust pathway for producing high-purity cephalothin acid, a vital precursor for semi-synthetic cephamycin-type antibiotics like cefoxitin. The methodology streamlines the workflow into sequential steps of aqueous condensation, centrifugal filtration, refining, decolorization, and crystallization, ensuring a product that meets stringent quality specifications while drastically reducing the ecological footprint of the synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Conventional solvent methods for cephalothin acid production rely heavily on organic media such as acetone, ethyl acetate, and petroleum ether, creating significant logistical and financial burdens. These processes typically involve dissolving 7-ACA in a mixture, followed by acylation using thiophene acetyl chloride in an organic phase, which requires precise temperature control to prevent degradation. The subsequent workup necessitates multiple extraction steps, washing with saline solutions, and extensive drying procedures to remove residual solvents, consuming substantial energy and time. This approach is not only capital intensive due to the high consumption of expensive organic reagents but also poses significant safety hazards associated with handling large volumes of flammable liquids in industrial settings. Furthermore, the environmental burden of disposing of or recycling these solvents adds a layer of regulatory complexity and cost that modern manufacturers strive to avoid to maintain compliance. The reliance on such volatile compounds necessitates specialized explosion-proof equipment and rigorous safety protocols, inflating both CAPEX and OPEX for production facilities.
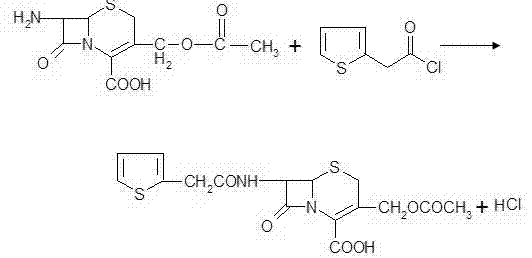
The Novel Approach
The novel aqueous phase approach revolutionizes this landscape by leveraging the unique solubility properties of 7-ACA salts in water under controlled pH conditions, offering a greener alternative. Instead of battling solubility issues with organic co-solvents, the new method dissolves the starting material directly in an aqueous alkaline solution, simplifying the initial setup significantly. The acylation proceeds efficiently in this medium, followed by a straightforward precipitation step induced by pH adjustment or salting-out agents, which avoids the need for complex phase separations. This eliminates the complex liquid-liquid extraction sequences typical of the old method, reducing the number of unit operations and the associated potential for yield loss. The result is a significantly simplified operation flow that reduces labor intensity and minimizes the potential for operator error during batching and discharging. By shifting the reaction medium to water, the process inherently mitigates fire risks and reduces the emission of volatile organic compounds (VOCs), aligning perfectly with green chemistry principles and modern sustainability mandates for responsible chemical manufacturing.
Mechanistic Insights into Aqueous Phase Condensation
The core chemical transformation involves the nucleophilic attack of the amino group at the 7-position of the 7-ACA nucleus on the carbonyl carbon of the 2-thiophene acetyl reagent within the aqueous environment. In the aqueous phase, maintaining the correct pH is critical to ensure the amino group is sufficiently nucleophilic while simultaneously preventing the hydrolysis of the sensitive beta-lactam ring, which is prone to degradation in extreme conditions. The patent specifies using alkaline conditioners like sodium carbonate or bicarbonate to facilitate the dissolution of 7-ACA and activate the amine for the subsequent acylation reaction. Once the condensation is complete, the reaction is terminated, and the product is precipitated, leveraging the decreased solubility of the acid form in the aqueous matrix. The use of water as a solvent also aids in heat dissipation, providing better temperature control compared to viscous organic mixtures, which is vital for exothermic reactions. This thermal stability is crucial for preserving the integrity of the sensitive beta-lactam structure throughout the synthesis, ensuring high chemical purity.
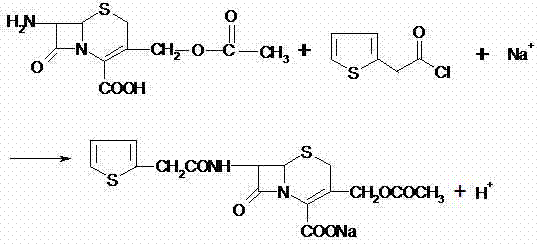
Impurity profiles are tightly managed through the specific sequence of decolorization and crystallization steps designed into this aqueous process. The addition of activated carbon in the aqueous phase effectively adsorbs colored by-products and polymeric impurities formed during the acylation, ensuring a visually clean filtrate. Because the product is isolated via precipitation and centrifugation rather than evaporation, thermal degradation is minimized, preserving the potency of the intermediate. The washing steps with small amounts of organic solvent, such as ethyl acetate, are sufficient to remove unreacted thiophene acetic acid derivatives without redissolving the product, optimizing solvent usage. This selective solubility behavior in the aqueous-organic interface allows for a highly efficient purification process that removes side products effectively. The final vacuum drying at moderate temperatures, specifically around 40-44°C, ensures that the moisture content is reduced to ≤0.5% without compromising the chemical stability of the cephalothin acid, resulting in a specification-ready intermediate suitable for downstream antibiotic synthesis.
How to Synthesize Cephalothin Acid Efficiently
To implement this efficient synthesis route, operators must strictly adhere to the defined parameters for pH control and temperature management to ensure reproducibility. The process begins with the dissolution of solid 7-ACA in deionized water under neutral or alkaline conditions, followed by the controlled addition of the acylating agent to manage the exotherm. Detailed standard operating procedures regarding the specific molar ratios, addition rates, and stirring speeds are essential to replicate the high yields reported in the patent embodiments successfully. The following guide outlines the critical operational milestones required to achieve consistent batch-to-batch quality and maximize the economic benefits of the water-phase method. For the complete, step-by-step standardized synthesis protocol, please refer to the detailed technical guide below.
- Dissolve solid 7-amino-cephalo-alkanoic acid in an aqueous phase solvent under neutrality or alkaline conditions.
- Perform condensation reaction with 2-thiophene acetyl reagent, then add a reaction terminating agent to induce precipitation.
- Filter the product, refine and decolorize in water, adjust pH for crystallization, and vacuum dry to obtain the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
This innovative manufacturing process offers substantial strategic advantages for procurement and supply chain stakeholders by fundamentally altering the cost structure and risk profile of production. By transitioning to a water-based system, the process removes the dependency on volatile and expensive organic solvents, which are subject to market price fluctuations and strict transportation regulations. This shift not only stabilizes raw material costs but also simplifies the logistics of inbound material handling, as water is universally available and non-hazardous. Furthermore, the reduction in process complexity leads to shorter cycle times and higher throughput, enabling suppliers to respond more agilely to market demand spikes. The inherent safety of the aqueous method reduces insurance premiums and regulatory compliance costs, passing significant savings down the supply chain to the end buyer.
- Cost Reduction in Manufacturing: The most immediate financial benefit stems from the drastic reduction in organic solvent consumption, which the patent explicitly states can be saved by over 70 percent compared to traditional methods. This translates directly into lower raw material procurement costs and reduced waste disposal fees, as there is significantly less hazardous waste to treat. Eliminating the need for large-scale solvent recovery systems further decreases capital expenditure and energy consumption associated with distillation. Additionally, the simplified workflow reduces labor costs by minimizing the time required for batching, discharging, and equipment cleaning. These cumulative efficiencies result in a significantly lower cost of goods sold (COGS), making the final API intermediate more competitive in the global market.
- Enhanced Supply Chain Reliability: Operational simplicity enhances supply chain reliability by reducing the number of potential failure points in the manufacturing process. The process requires less specialized equipment, such as extensive explosion-proofing, allowing for production in a wider range of facilities without major infrastructure upgrades. Reduced labor intensity means fewer bottlenecks in manual operations like batching and discharging, leading to more consistent production schedules. The use of water as a primary solvent also mitigates the risk of supply disruptions caused by shortages of specific organic solvents, ensuring business continuity. This robustness makes the supplier a more dependable partner for long-term contracts and just-in-time delivery models.
- Scalability and Environmental Compliance: The aqueous nature of the reaction makes scale-up inherently safer and more predictable, facilitating the transition from pilot plant to commercial tonnage production. Heat transfer is more efficient in water, reducing the risk of thermal runaways during exothermic acylation, which allows for larger batch sizes without proportional increases in risk. This safety margin supports the commercial scale-up of complex pharmaceutical intermediates while maintaining strict adherence to environmental regulations. The significant reduction in VOC emissions aligns with increasingly stringent global environmental standards, future-proofing the production facility against tighter regulations. Consequently, this method supports sustainable growth and ensures uninterrupted supply even as regulatory landscapes evolve.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous phase synthesis technology. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation to provide clarity on process capabilities. Understanding these details is crucial for evaluating the feasibility of integrating this intermediate into your existing supply chain. We encourage technical teams to review these points to assess the alignment with their specific quality and volume requirements.
Q: How does the aqueous phase method reduce production costs compared to solvent methods?
A: The aqueous phase method saves over 70% of organic solvents, eliminating the high costs associated with purchasing, recovering, and disposing of large volumes of flammable organic reagents like acetone and ethyl acetate.
Q: What are the safety advantages of using water as a reaction medium for cephalothin acid?
A: Using water eliminates the fire and explosion hazards associated with volatile organic solvents, significantly lowering the safety requirements for the production facility and reducing environmental pollution risks.
Q: Is the purity of cephalothin acid produced via this method suitable for pharmaceutical use?
A: Yes, the process includes specific refining, decolorizing, and crystallization steps that yield a product with moisture content ≤0.5%, meeting stringent quality specifications for antibiotic intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cephalothin Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to maintain competitiveness in the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the aqueous phase method can be seamlessly transferred to industrial manufacturing. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise allows us to navigate the complexities of process optimization, ensuring that the theoretical benefits of patents like CN102040614A are fully realized in commercial output.
We invite you to collaborate with us to leverage these technological advancements for your specific product lines. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our optimized production methods can enhance your supply chain efficiency and reduce overall manufacturing costs.
