Optimizing 18F-FDG Precursor Supply: Advanced Purification of Mannose Intermediates
The escalating global demand for Positron Emission Tomography (PET) imaging agents has placed immense pressure on the supply chain of critical precursors, particularly for the widely used tracer 18F-FDG. At the heart of this supply chain lies the synthesis of mannose triflate, a key intermediate whose quality directly dictates the efficiency of the final radiopharmaceutical. Patent CN103739635A addresses a longstanding bottleneck in this sector by introducing a refined purification methodology for 1,3,4,6-tetra-O-acetyl-beta-D-mannopyranose. This specific intermediate serves as the foundational building block for mannose triflate, and historically, its production has been plagued by inconsistent yields and purity issues that ripple through the entire manufacturing process. By leveraging a novel solvent engineering approach, this technology offers a pathway to stabilize the supply of high-grade diagnostic reagents, ensuring that pharmaceutical manufacturers can meet the rigorous quality standards required for clinical applications without compromising on production throughput.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing 1,3,4,6-tetra-O-acetyl-beta-D-mannopyranose have relied heavily on standard recrystallization techniques using solvents such as ethanol or diethyl ether. While these methods are chemically straightforward, they suffer from significant thermodynamic limitations that result in suboptimal recovery rates. In conventional processes, the solubility profiles of the target beta-anomer and its closely related impurities, such as the alpha-anomer and acetyl-migrated byproducts, are often too similar in single-solvent systems. This similarity prevents effective separation during the crystallization phase, leading to product losses where the target compound remains dissolved in the mother liquor or co-crystallizes with impurities. Consequently, historical data indicates that yields often stagnate around 40% to 50%, forcing manufacturers to process larger volumes of raw materials to achieve the same output, which drastically inflates operational costs and waste generation.
The Novel Approach
The innovation detailed in the patent data introduces a sophisticated binary solvent system comprising n-butyl alcohol and 2-methoxyethanol to overcome these solubility constraints. By carefully tuning the volume ratio of these two solvents, typically between 4:1 and 8:1, the process creates a unique polarity environment that selectively favors the precipitation of the desired beta-anomer. This approach fundamentally alters the crystallization kinetics, allowing the target molecule to form a stable crystal lattice while keeping the problematic impurities in solution. The result is a dramatic enhancement in both isolation efficiency and product specification, transforming a previously loss-heavy step into a high-yield operation. This shift not only maximizes the utility of the expensive upstream intermediates but also simplifies the downstream processing requirements by delivering a product that is ready for the subsequent triflation reaction with minimal additional refinement.
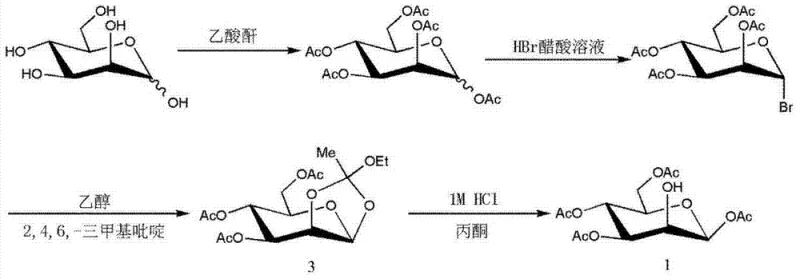
Mechanistic Insights into Solvent-Mediated Crystallization and Impurity Control
The efficacy of this purification strategy is rooted in the precise manipulation of intermolecular forces and solvation shells surrounding the carbohydrate molecules. During the hydrolysis step of the precursor synthesis, the reaction mixture inevitably generates a complex equilibrium of anomers and regioisomers due to acetyl group migration. Specifically, the formation of 2,3,4,6-tetra-acetyl-beta-D-mannose and its alpha-counterpart poses a significant challenge, as these structures are stereoisomers with very similar physical properties. The introduction of 2-methoxyethanol, an ether-alcohol with distinct hydrogen-bonding capabilities, alongside the longer-chain n-butyl alcohol, disrupts the solvation of the target beta-anomer more effectively than it does for the impurities. This differential solvation energy lowers the saturation point of the target compound specifically, driving it out of the solution phase as pure crystals while the impurities remain thermodynamically stable in the liquid phase.
Furthermore, the process conditions, including controlled cooling to temperatures between 8°C and 15°C, are critical for managing the mutarotation equilibrium. Rapid or uncontrolled temperature changes could trigger further acetyl migration or anomerization, reintroducing impurities that were previously removed. The patented method mitigates this risk by maintaining a gentle thermal gradient that allows for the growth of large, well-defined crystals. These larger crystals possess a lower surface-area-to-volume ratio, which inherently reduces the adsorption of mother liquor containing impurities onto the crystal surface. This mechanistic control ensures that the final product not only meets high chemical purity standards but also exhibits superior physical characteristics, such as flowability and stability, which are essential for automated synthesis modules used in radiopharmacy.
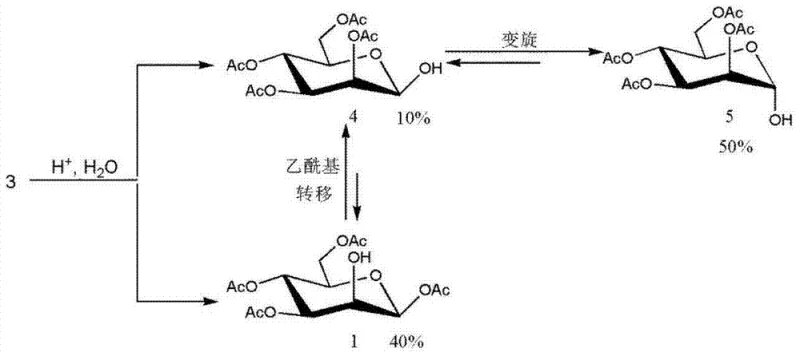
How to Synthesize 1,3,4,6-tetra-O-acetyl-beta-D-mannopyranose Efficiently
Implementing this purification protocol requires strict adherence to solvent ratios and thermal profiles to replicate the high yields observed in the patent examples. The process begins with the dissolution of the crude material in the heated binary solvent mixture, ensuring complete solvation before initiating the crystallization phase. Operators must monitor the cooling rate carefully to avoid oiling out or premature precipitation, which can trap impurities within the crystal matrix. Following filtration, a specific washing regimen using n-butyl alcohol is employed to displace any residual mother liquor from the crystal surface without redissolving the product. Finally, vacuum drying at moderate temperatures removes the solvent traces, yielding a stable, free-flowing powder suitable for long-term storage or immediate conversion to mannose triflate.
- Dissolve the crude 1,3,4,6-tetraacetyl-beta-D-mannopyranose in a heated mixture of n-butyl alcohol and 2-methoxyethanol (volume ratio 4-8: 1).
- Cool the solution gradually to 8-15°C to induce complete crystallization of the target beta-anomer.
- Filter the crystals, wash repeatedly with n-butyl alcohol, and vacuum dry at 45-55°C to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this advanced purification technology translates directly into enhanced operational resilience and cost efficiency. The primary economic driver is the substantial increase in process yield, which effectively reduces the consumption of upstream raw materials such as D-mannose and acetic anhydride per kilogram of final product. By minimizing material waste, manufacturers can significantly lower the variable costs associated with production, creating a more competitive pricing structure for the final intermediate. Additionally, the robustness of the solvent system reduces the sensitivity of the process to minor fluctuations in reaction conditions, leading to more consistent batch-to-batch quality. This consistency is vital for supply chain planning, as it reduces the frequency of failed batches and the need for costly re-processing or disposal of off-spec material.
- Cost Reduction in Manufacturing: The shift from low-yield ethanol crystallization to the high-efficiency binary solvent system eliminates the need for excessive over-processing to meet purity specifications. By recovering a significantly larger fraction of the target molecule from the crude mixture, the effective cost per unit of pure intermediate is drastically reduced. This efficiency gain allows for better margin management in the face of fluctuating raw material prices and provides a buffer against supply shocks. Furthermore, the reduced volume of waste solvent and solid byproducts lowers the environmental compliance costs associated with waste treatment and disposal, contributing to a leaner and more sustainable manufacturing footprint.
- Enhanced Supply Chain Reliability: High-yield processes inherently offer greater supply security because they maximize output from existing reactor capacity. In a market where demand for PET tracers is growing, the ability to produce more intermediate from the same equipment footprint ensures that suppliers can meet surging orders without immediate capital expenditure on new facilities. The improved purity profile also shortens the lead time for quality control testing, as fewer iterations of analysis are needed to verify compliance with pharmacopeial standards. This acceleration facilitates faster release of goods to customers, reducing inventory holding times and improving cash flow dynamics for all stakeholders in the value chain.
- Scalability and Environmental Compliance: The solvents utilized in this process, n-butyl alcohol and 2-methoxyethanol, are industrially available and manageable within standard chemical handling protocols, facilitating easy scale-up from pilot to commercial production. The process design minimizes the use of highly volatile or hazardous ethers that often pose safety risks in large-scale operations. By optimizing the solvent recovery potential through distillation due to the distinct boiling points involved, the overall environmental impact is mitigated. This alignment with green chemistry principles not only satisfies regulatory requirements but also appeals to end-users who are increasingly prioritizing suppliers with strong environmental, social, and governance (ESG) credentials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing the long-term viability of the supply source. The answers are derived directly from the experimental data and mechanistic explanations provided in the patent literature, ensuring accuracy and relevance to industrial applications.
Q: Why is the purity of 1,3,4,6-tetra-O-acetyl-beta-D-mannopyranose critical for 18F-FDG production?
A: High purity is essential because impurities such as alpha-anomers or acetyl-migrated byproducts can interfere with the subsequent triflation reaction, reducing the radiochemical yield of the final PET tracer and complicating quality control for clinical use.
Q: How does the n-butyl alcohol and 2-methoxyethanol solvent system improve yield compared to ethanol?
A: This specific binary solvent system optimizes the solubility differential between the target beta-anomer and its impurities, allowing for superior crystal lattice formation and significantly higher recovery rates compared to traditional single-solvent recrystallization.
Q: What are the main impurities removed during this purification process?
A: The process effectively removes 2,3,4,6-tetra-acetyl-beta-D-mannose and 2,3,4,6-tetra-acetyl-alpha-D-mannose, which are formed via acetyl group migration and mutarotation during the preceding hydrolysis steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,3,4,6-tetra-O-acetyl-beta-D-mannopyranose Supplier
As the demand for high-quality diagnostic intermediates continues to rise, partnering with a manufacturer that possesses deep technical expertise in purification technologies is essential for securing a stable supply. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced process chemistry to deliver intermediates that meet the most stringent global standards. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs whether you are in the clinical trial phase or full-scale commercial manufacturing. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify every batch against the highest benchmarks for pharmaceutical intermediates.
We invite you to collaborate with us to optimize your supply chain for PET tracer precursors. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. By engaging with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate how our optimized purification processes can enhance your overall manufacturing efficiency. Contact us today to discuss how we can support your mission of delivering life-saving diagnostic tools to patients worldwide.
