Advanced Synthesis of Water-Soluble Beta-Elemene Glycoside Derivatives for Oncology Applications
The pharmaceutical industry continuously seeks to optimize the pharmacokinetic profiles of potent natural products, and patent CN101538234A presents a significant breakthrough in this domain by detailing the synthesis of beta-elemene glycoside derivatives. Beta-elemene, a sesquiterpene extracted from Curcuma wenyujin, is well-known for its broad-spectrum antitumor activity and safety profile; however, its clinical utility is severely hampered by low polarity and poor water solubility. This technical disclosure addresses these limitations by chemically modifying the beta-elemene scaffold through glycosylation, effectively introducing polar carbohydrate moieties that enhance aqueous solubility without compromising the core terpenoid structure. The patent outlines a robust, multi-step synthetic pathway that transforms the lipophilic parent compound into highly functionalized intermediates and final glycoside products, offering a viable route for developing next-generation anticancer agents with improved bioavailability.
For research and development teams focusing on oncology drug discovery, this methodology provides a critical toolkit for structure-activity relationship (SAR) studies. By enabling the attachment of various monosaccharides and disaccharides such as glucose, galactose, and lactose, the process allows for the fine-tuning of molecular properties to maximize therapeutic efficacy. The strategic introduction of hydrophilic groups not only facilitates formulation development but also potentially leverages active transport mechanisms associated with sugar uptake in tumor cells. As a reliable pharmaceutical intermediates supplier, understanding these synthetic nuances is essential for scaling production while maintaining the stringent purity required for preclinical and clinical evaluation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to utilizing beta-elemene have largely relied on emulsion formulations to overcome its inherent hydrophobicity, yet these physical mixtures often suffer from stability issues and inconsistent absorption rates in vivo. Direct extraction yields a compound that is difficult to formulate for intravenous administration without complex surfactant systems, which can introduce additional toxicity or immunogenicity concerns. Furthermore, simple physical blending does not alter the intrinsic chemical nature of the molecule, meaning the fundamental barrier of membrane permeability and solubility remains a bottleneck for high-dose therapies. Previous chemical modifications often lacked regioselectivity or resulted in unstable linkages that degraded prematurely in physiological conditions, failing to deliver the active moiety to the target site effectively.
The Novel Approach
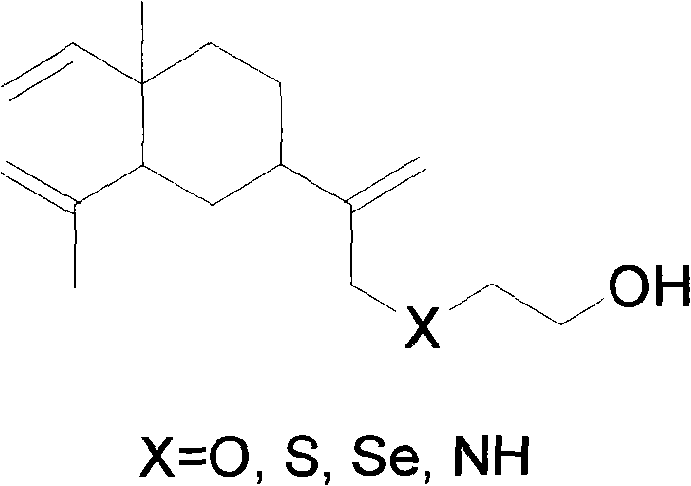
The novel approach described in the patent utilizes a covalent glycosylation strategy that fundamentally alters the physicochemical properties of the beta-elemene skeleton. By first converting beta-elemene into a reactive chloride intermediate and subsequently linking it to ethanolamines or mercaptoethanol, the process creates a stable spacer arm capable of supporting glycosidic bond formation. This modular design allows for the precise attachment of fully acetylated sugars via robust O-glycosidic or S-glycosidic linkages, ensuring metabolic stability during circulation. The resulting beta-elemene substituted ethyl glycosides exhibit markedly improved water solubility, addressing the primary limitation of the parent compound while retaining the pharmacophore responsible for antitumor activity. This chemical evolution represents a paradigm shift from formulation-based solutions to structure-based optimization.
Mechanistic Insights into BF3-Catalyzed Glycosylation
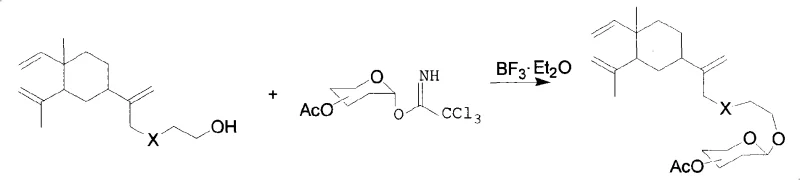
The core of this synthetic innovation lies in the glycosylation step, specifically the trichloroacetimidate method which offers excellent stereocontrol and yield. In this mechanism, a fully acetylated sugar trichloroacetimidate acts as the glycosyl donor, activated by a Lewis acid catalyst such as boron trifluoride diethyl etherate (BF3·Et2O). The reaction proceeds through the formation of an oxocarbenium ion intermediate, which is then attacked by the hydroxyl group of the beta-elemene substituted ethanol acceptor. The use of 4A molecular sieves is critical in this step to scavenge trace moisture, preventing the hydrolysis of the sensitive imidate donor and ensuring high conversion rates. This method is particularly advantageous for high-purity pharmaceutical intermediates as it typically proceeds under mild conditions, minimizing side reactions such as elimination or rearrangement of the terpenoid backbone.
Impurity control is meticulously managed through the choice of protecting groups and reaction conditions. The acetyl groups on the sugar moiety serve a dual purpose: they enhance the solubility of the intermediate in organic solvents like dichloromethane and prevent unwanted side reactions at other hydroxyl positions. Following the glycosylation, the reaction mixture is neutralized with triethylamine to quench the acidic catalyst, preventing acid-catalyzed degradation of the product. Subsequent purification via flash silica gel column chromatography effectively removes unreacted starting materials and any orthoester byproducts that may form. This rigorous control over the reaction environment ensures that the final beta-elemene glycoside derivatives meet the stringent quality standards required for biological testing and potential clinical translation.
How to Synthesize Beta-Elemene Glycoside Derivatives Efficiently
The synthesis of these valuable derivatives follows a logical three-stage progression designed for scalability and reproducibility. Initially, beta-elemene is functionalized to create a reactive handle, followed by the coupling of the sugar unit, and finally, the removal of protecting groups to reveal the bioactive polar structure. This streamlined workflow minimizes unit operations and maximizes overall throughput, making it suitable for both laboratory-scale SAR studies and larger pilot production runs. The detailed standardized synthesis steps for implementing this protocol are outlined in the guide below.
- Preparation of Beta-Elemene Substituted Ethanol intermediates via chlorination and subsequent reaction with mercaptoethanol or aminoethanol.
- Glycosylation reaction using either the trichloroacetimidate method with BF3·Et2O or the bromosugar method with Ag2CO3 promoter.
- Final deprotection of acetyl groups using sodium methoxide in methanol to yield the target beta-elemene glycoside.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthetic route offers substantial benefits regarding raw material availability and process robustness. The starting material, beta-elemene, is derived from abundant natural sources, ensuring a stable supply chain foundation that is less susceptible to the volatility associated with totally synthetic complex scaffolds. Furthermore, the reagents employed in the glycosylation steps, such as silver carbonate and boron trifluoride, are commodity chemicals available from multiple global vendors, reducing the risk of single-source dependency. The ability to utilize various sugars allows for flexibility in sourcing, enabling procurement teams to optimize costs based on market fluctuations of specific carbohydrate feedstocks without altering the core process flow.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive transition metal catalysts often found in cross-coupling reactions, relying instead on more economical Lewis acids and promoters. The operational simplicity, characterized by room temperature reactions for the substitution step and straightforward workup procedures involving filtration and concentration, significantly reduces energy consumption and labor costs. Additionally, the high yields reported in the deprotection step minimize material loss, ensuring that the overall cost of goods sold (COGS) remains competitive for large-scale pharmaceutical intermediates manufacturing.
- Enhanced Supply Chain Reliability: The synthetic pathway is designed with resilience in mind, utilizing stable intermediates that can be stored and transported if necessary. The separation techniques described, such as silica gel chromatography and liquid-liquid extraction, are standard unit operations in any GMP facility, ensuring that technology transfer to contract manufacturing organizations (CMOs) is seamless. This compatibility with existing infrastructure reduces lead times for scale-up and ensures a continuous supply of critical intermediates for downstream drug development programs.
- Scalability and Environmental Compliance: The methods described avoid the use of highly toxic heavy metals, simplifying waste treatment and disposal protocols in compliance with increasingly strict environmental regulations. The solvent systems, primarily consisting of dichloromethane, ethanol, and petroleum ether, are well-understood and can be efficiently recovered and recycled in modern chemical plants. This focus on greener chemistry principles not only lowers the environmental footprint but also mitigates regulatory risks associated with residual solvent limits in final drug substances.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these compounds, derived directly from the patent specifications. Understanding these details is crucial for partners evaluating the feasibility of integrating these derivatives into their development pipelines. The answers reflect the specific experimental conditions and outcomes documented in the intellectual property.
Q: What is the primary advantage of synthesizing beta-elemene glycoside derivatives over native beta-elemene?
A: The primary advantage is significantly improved water solubility. Native beta-elemene has low polarity and poor water solubility, which limits its absorption and application range. Glycosylation introduces polar sugar moieties, enhancing bioavailability and potential biological activity.
Q: Which glycosylation methods are described in the patent for these derivatives?
A: The patent details two main methods: the trichloroacetimidate ester method utilizing BF3·Et2O as a catalyst at low temperatures, and the bromosugar method employing Ag2CO3-diatomaceous earth as a promoter under reflux conditions.
Q: What types of sugars can be conjugated to the beta-elemene scaffold?
A: The synthesis is versatile and supports various monosaccharides and disaccharides, including glucose, mannose, galactose, xylose, lactose, and maltose, allowing for a diverse library of derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Elemene Glycoside Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of beta-elemene glycoside derivatives in modern oncology therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to clinical supply is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee the quality of every batch, adhering to the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with us to leverage this innovative synthetic technology for your drug development needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your project milestones and accelerate your time to market.
