Advanced Industrial Synthesis of 3,4-Difluorobenzonitrile for Global Agrochemical Supply Chains
Introduction to Next-Generation Fluorination Technology
The global demand for fluorinated intermediates, particularly within the agrochemical and pharmaceutical sectors, necessitates robust and scalable manufacturing protocols. Patent CN102690214A introduces a transformative industrial preparation process for 3,4-difluorobenzonitrile, a critical building block for selectivity herbicides and advanced liquid crystal materials. This technology addresses the longstanding challenges of traditional halogen exchange reactions by employing a specialized catalytic system comprising anhydrous potassium fluoride and a unique ammonium salt catalyst in a 1,3-dimethyl-2-imidazolidinone (DMI) solvent matrix. Unlike conventional methods that suffer from harsh conditions and poor atom economy, this novel approach operates under nitrogen protection at controlled temperatures between 200°C and 230°C, achieving superior conversion rates. For R&D directors and procurement specialists seeking a reliable agrochemical intermediate supplier, understanding the mechanistic advantages of this patent is crucial for securing a stable supply of high-purity precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3,4-difluorobenzonitrile has been plagued by convoluted multi-step pathways that impose significant burdens on production costs and environmental compliance. Traditional routes often involve the acylation of difluorobenzene followed by oxidation, chlorination, amidation, and finally dehydration, creating a lengthy operational sequence with cumulative yield losses at every stage. As illustrated in the reaction scheme below, these legacy processes require hazardous reagents and generate substantial quantities of toxic waste, complicating downstream purification and waste treatment protocols.
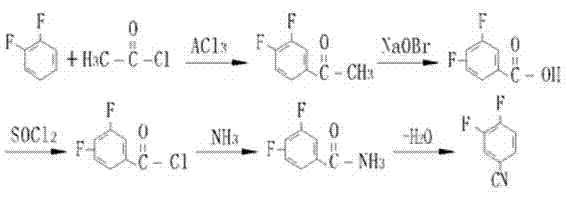
Furthermore, alternative pathways utilizing bromination and copper-cyanide substitution, while chemically viable, introduce heavy metal contamination risks and require expensive catalysts that are difficult to remove to pharmaceutical grades. The reliance on high-pressure autoclaves and extreme temperatures in these older methodologies often leads to safety incidents and equipment corrosion, resulting in unpredictable downtime for manufacturing facilities. Consequently, the industry has urgently required a streamlined alternative that eliminates these bottlenecks while maintaining the structural integrity of the fluorine substituents.
The Novel Approach
The methodology disclosed in CN102690214A represents a paradigm shift by utilizing a direct nucleophilic aromatic substitution (SnAr) on 3,4-dichlorobenzonitrile. This single-step fluorination strategy bypasses the need for constructing the carbon skeleton from scratch, instead leveraging the readily available dichloro-precursor. The integration of reactive distillation allows for the continuous removal of the product as it forms, effectively driving the equilibrium towards completion and preventing the reverse reaction or thermal decomposition of the sensitive nitrile group. This innovation not only simplifies the process flow but also drastically reduces the reaction cycle time, making it highly suitable for the commercial scale-up of complex agrochemical intermediates.
Mechanistic Insights into Catalytic Halogen Exchange
The core of this technological breakthrough lies in the synergistic interaction between the solvent, the fluoride source, and the phase-transfer catalyst. The use of 1,3-dimethyl-2-imidazolidinone (DMI) provides a highly polar aprotic environment that effectively solvates the potassium cation, thereby "naked" the fluoride anion and enhancing its nucleophilicity. However, the true innovation is the addition of N-bis(dimethylamino)methylene-1,3-dimethyl imidazoline-2-ammonium chloride salt. This specific catalyst exhibits exceptional thermal stability, remaining active at the elevated temperatures of 200°C to 230°C required to overcome the activation energy of the chlorine-fluorine exchange on the electron-deficient aromatic ring.
From an impurity control perspective, the mechanism inherently suppresses the formation of poly-fluorinated byproducts or hydrolysis products. By maintaining a strict nitrogen atmosphere and utilizing vacuum-assisted dehydration prior to reactant addition, the system minimizes the presence of moisture which could otherwise lead to the hydrolysis of the nitrile group into carboxylic acids. The continuous distillation of the product further ensures that the 3,4-difluorobenzonitrile spends minimal time in the high-temperature reaction zone, preserving its chemical purity and ensuring that the final distillate meets stringent specifications without requiring extensive recrystallization steps.
How to Synthesize 3,4-Difluorobenzonitrile Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and thermal gradients to maximize the efficiency of the halogen exchange. The patent outlines a detailed protocol where the molar ratio of 3,4-dichlorobenzonitrile to potassium fluoride is optimized between 1:2.2 and 1:4.0, ensuring an excess of fluoride to drive the reaction to completion without excessive waste. The catalyst loading is carefully tuned to 5% to 10% molar equivalent relative to the substrate, balancing cost against catalytic activity. Operators must strictly adhere to the temperature ramping profile, initiating the reaction only after thorough dehydration of the solvent system to prevent catalyst deactivation.
- Charge the fluorination reactor with 1,3-dimethyl-2-imidazolidinone (DMI) solvent and toluene, followed by the addition of anhydrous potassium fluoride and the ammonium salt catalyst under nitrogen protection.
- Heat the mixture to 200-230°C while applying vacuum to remove water, then introduce 3,4-dichlorobenzonitrile gradually while maintaining reactive distillation to collect the crude product.
- Separate the solid byproducts via centrifugation, recover the solvent for recycling, and purify the crude 3,4-difluorobenzonitrile through fractional distillation to achieve >98.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates directly into enhanced operational resilience and cost competitiveness. The elimination of multi-step synthetic sequences reduces the number of unit operations required, thereby lowering capital expenditure on equipment and minimizing the footprint of the manufacturing facility. The ability to recycle both the solvent and the unreacted solid materials back into the process creates a closed-loop system that significantly mitigates the volatility of raw material pricing. This circular economy approach ensures that the cost reduction in agrochemical intermediate manufacturing is sustainable over the long term, shielding buyers from market fluctuations associated with disposable reagents.
- Cost Reduction in Manufacturing: The most significant economic driver of this technology is the dramatic improvement in reaction yield. While traditional catalysts such as crown ethers or organophosphorus compounds typically achieve yields around 30%, the novel ammonium salt catalyst described in the patent pushes yields to approximately 80%. This near-tripling of efficiency means that for every ton of raw material purchased, nearly three times more product is generated compared to legacy methods. Furthermore, the recovery and reuse of the DMI solvent eliminate the recurring cost of purchasing fresh solvent for every batch, compounding the savings across thousands of tons of annual production volume.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of sourcing specialized reagents for multi-step syntheses. By simplifying the route to a direct fluorination of a commodity chemical (3,4-dichlorobenzonitrile), the supply chain becomes far more robust. The starting materials are widely available from bulk chemical suppliers, reducing the risk of bottlenecks associated with niche intermediates. Additionally, the shortened reaction cycle time allows manufacturers to respond more agilely to spikes in demand, reducing lead time for high-purity 3,4-difluorobenzonitrile and ensuring that downstream herbicide production lines remain fully stocked without interruption.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces unforeseen safety and environmental hurdles, particularly when dealing with exothermic reactions or toxic byproducts. This process is designed with industrial scalability in mind, utilizing standard stainless steel reactors equipped with distillation columns rather than exotic high-pressure vessels. The reduction in hazardous waste generation, achieved through solvent recycling and the avoidance of heavy metal cyanide reagents, simplifies regulatory compliance. This ease of waste management facilitates faster permitting for capacity expansion, allowing suppliers to scale up from pilot plants to 100 MT annual production capacities with minimal environmental friction.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this fluorination technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the method's capabilities and limitations for potential partners.
Q: What represents the primary advantage of the catalytic system in patent CN102690214A?
A: The primary advantage is the use of a specific N-bis(dimethylamino)methylene ammonium salt catalyst which tolerates high temperatures (200-230°C) and significantly boosts yield to approximately 80%, compared to only 30% with traditional crown ethers or organophosphorus catalysts.
Q: How does the reactive distillation process improve the synthesis efficiency?
A: By combining reaction and rectification, the process continuously removes the product (3,4-difluorobenzonitrile) from the reaction zone. This shifts the chemical equilibrium forward according to Le Chatelier's principle, accelerates the reaction rate, and minimizes thermal degradation or defluorination side reactions.
Q: Is the solvent system sustainable for large-scale manufacturing?
A: Yes, the process utilizes 1,3-dimethyl-2-imidazolidinone (DMI) which is recovered and recycled efficiently. Both the solid and liquid outputs from the separation step can be reused in subsequent batches, drastically reducing three-waste discharge and raw material consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Difluorobenzonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires deep technical expertise and rigorous quality assurance. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN102690214A are fully realized in practice. Our facilities are equipped with state-of-the-art reactive distillation units and stringent purity specifications are enforced through our rigorous QC labs, guaranteeing that every batch of 3,4-difluorobenzonitrile meets the exacting standards required for global agrochemical registration.
We invite international partners to collaborate with us to leverage this advanced synthesis route for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic impact of switching to this high-yield process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical foundation available in the market.
