Advanced Manufacturing of 5-Lipoxygenase Inhibitor Intermediates via Solid-Base Catalysis
Introduction to Next-Generation 5-Lipoxygenase Inhibitor Synthesis
The development of potent anti-inflammatory agents relies heavily on the efficient synthesis of complex heterocyclic intermediates. Patent CN1291609A discloses a groundbreaking methodology for preparing compounds of formula (1.0.0), which serve as critical inhibitors of 5-lipoxygenase activity. These compounds are pivotal in treating inflammation, allergy, and cardiovascular diseases by blocking the arachidonic acid cascade. The patent outlines a sophisticated route that overcomes historical limitations in yield and purity, utilizing advanced catalytic systems and solid-state reagents to achieve superior results. This technical insight explores the transformative potential of this chemistry for modern pharmaceutical supply chains.
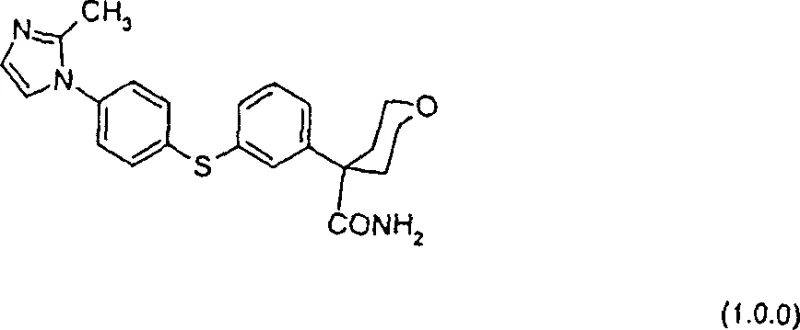
At the core of this innovation is the ability to construct the diaryl sulfide linkage and the N-heterocyclic attachment with exceptional precision. Traditional methods often struggled with harsh conditions that degraded sensitive functional groups or required tedious purification steps. The disclosed process introduces a streamlined approach that integrates palladium-catalyzed cross-coupling with a novel solid-base mediated nucleophilic substitution. This dual-strategy ensures that the final active pharmaceutical ingredient (API) intermediate meets stringent quality standards while maintaining economic viability for large-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of diaryl sulfides and N-aryl heterocycles relied heavily on aqueous basic conditions or expensive stoichiometric reagents. Conventional nucleophilic aromatic substitutions often required strong electron-withdrawing groups on the aromatic ring to proceed, limiting substrate scope. Furthermore, the use of aqueous bases frequently led to hydrolysis of sensitive nitrile or amide functionalities, resulting in significant yield losses and complex impurity profiles. Purification was often cumbersome, involving multiple extraction steps that generated substantial chemical waste and increased processing time.
The Novel Approach
The methodology described in CN1291609A revolutionizes this landscape by employing solid sodium hydroxide or potassium hydroxide in aprotic solvents like DMSO. This shift eliminates bulk water from the reaction environment, drastically reducing hydrolytic degradation. The process leverages phase transfer catalysts, such as tetra-n-butylammonium chloride (TBAC), to facilitate the reaction between the solid base and organic substrates. This allows for milder yet effective conditions, typically between 100°C and 130°C, ensuring high conversion rates without compromising structural integrity.
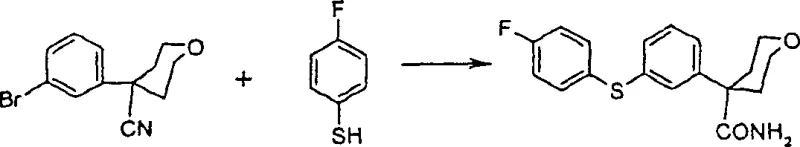
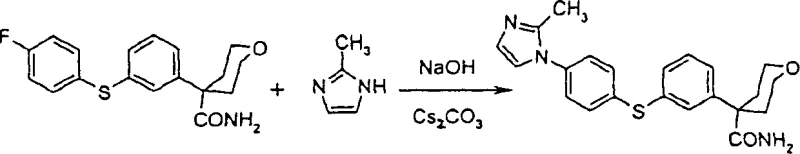
Additionally, the integration of palladium-catalyzed thioetherification enables the direct coupling of aryl halides with thiophenols. As illustrated in the reaction scheme, tetrahydro-4-(3-bromo-phenyl)-2H-pyran-4-carbonitrile reacts efficiently with 4-fluorothiophenol using tetrakis(triphenylphosphine)palladium(0). This step is crucial for establishing the core scaffold. The subsequent N-arylation step, depicted below, utilizes the solid-base protocol to attach the imidazole or pyrazole ring, completing the molecular architecture with remarkable efficiency.
Mechanistic Insights into Solid-Base Mediated N-Arylation
The mechanistic elegance of this process lies in the interplay between the solid base and the phase transfer catalyst. In the absence of bulk water, the solid sodium hydroxide provides a high concentration of hydroxide ions at the solid-liquid interface. The phase transfer catalyst, typically a quaternary ammonium salt, forms lipophilic ion pairs with the deprotonated heterocycle. These ion pairs are soluble in the organic phase (DMSO), allowing the nucleophile to attack the electron-deficient aryl fluoride effectively. This mechanism bypasses the need for activating groups on the aromatic ring, expanding the scope of accessible derivatives.
Impurity control is inherently built into this design. By avoiding aqueous workups until the final quenching stage, the formation of hydrolysis byproducts is minimized. The use of activated carbon during the recrystallization of intermediates further scavenges trace metal impurities, particularly residual palladium from the earlier coupling step. This rigorous attention to purification ensures that the final methanesulfonate salt exhibits high chemical purity, a critical requirement for regulatory approval in pharmaceutical manufacturing.
How to Synthesize Tetrahydro-4-phenyl-2H-pyran-4-carboxamide Efficiently
The synthesis pathway detailed in the patent offers a robust framework for producing these valuable intermediates. It begins with the palladium-catalyzed coupling of a bromo-nitrile precursor with a fluorinated thiophenol, followed by hydrolysis to the carboxamide. The final assembly involves the displacement of the fluoride atom with a nitrogen heterocycle under solid-base conditions. This sequence is optimized for both yield and operational simplicity, making it ideal for process chemistry teams looking to scale up production.
- Perform palladium-catalyzed coupling of tetrahydro-4-(3-bromo-phenyl)-2H-pyran-4-carbonitrile with 4-fluorothiophenol in alcohol solvent with strong base.
- Hydrolyze the nitrile group to a carboxamide either concurrently or in a separate step using aqueous base and heat.
- Execute nucleophilic aromatic substitution using solid sodium hydroxide and a phase transfer catalyst in DMSO to attach the heterocyclic ring.
- Purify the final product via recrystallization and convert to the methanesulfonate salt for improved stability and handling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible strategic benefits. The shift towards solid-base chemistry significantly reduces the volume of aqueous waste generated during production. This aligns with modern environmental compliance standards and lowers the costs associated with waste treatment and disposal. Furthermore, the use of commodity chemicals like sodium hydroxide and DMSO ensures a stable and cost-effective supply of raw materials, mitigating risks associated with specialized reagent shortages.
- Cost Reduction in Manufacturing: The elimination of complex aqueous extraction steps simplifies the manufacturing workflow. By using solid bases and phase transfer catalysts, the process reduces energy consumption associated with heating large volumes of water. Additionally, the high yields reported in the patent examples mean less raw material is wasted, directly lowering the cost of goods sold (COGS) for the final API intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely available catalysts like tetrakis(triphenylphosphine)palladium(0) and common solvents ensures supply continuity. Unlike processes dependent on exotic ligands or cryogenic conditions, this method can be implemented in standard glass-lined or stainless steel reactors. This flexibility allows for multi-vendor sourcing strategies, reducing dependency on single suppliers and enhancing overall supply chain resilience.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated to work effectively with varying batch sizes. The reduced solvent usage and simplified workup procedures contribute to a smaller environmental footprint. This sustainability advantage is increasingly important for pharmaceutical companies aiming to meet corporate social responsibility goals and adhere to strict green chemistry principles.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for successful implementation. The following questions address common concerns regarding reaction conditions, catalyst selection, and purification strategies. These insights are derived directly from the experimental data provided in the patent, offering practical guidance for chemists and engineers evaluating this technology for commercial adoption.
Q: What are the advantages of using solid sodium hydroxide in the N-arylation step?
A: Using solid sodium hydroxide minimizes water content in the reaction mixture, which significantly reduces hydrolysis side reactions and simplifies downstream workup. This approach, often combined with phase transfer catalysts like TBAC, allows for higher reaction temperatures (100-130°C) and improved yields compared to traditional aqueous base methods.
Q: How is residual palladium managed in this synthesis route?
A: Residual palladium from the initial thioetherification step is effectively removed through activated carbon treatment during the recrystallization of the intermediate. The subsequent methanesulfonate salt formation step further ensures high purity by precipitating the product while leaving metal impurities in the mother liquor.
Q: Can this process be scaled for commercial API production?
A: Yes, the process is designed for scalability. The use of common solvents like isopropanol and DMSO, along with robust catalysts like tetrakis(triphenylphosphine)palladium(0), supports large-scale manufacturing. The solid-base protocol eliminates the need for complex aqueous extractions, streamlining the workflow for industrial applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydro-4-phenyl-2H-pyran-4-carboxamide Supplier
At NINGBO INNO PHARMCHEM, we specialize in translating complex patent methodologies into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from the lab to the market. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-lipoxygenase inhibitor intermediate meets the highest industry standards.
We invite you to collaborate with us to optimize your supply chain for these critical pharmaceutical building blocks. Contact our technical procurement team today to request a Customized Cost-Saving Analysis. We are ready to provide specific COA data and route feasibility assessments tailored to your unique production requirements, helping you secure a competitive edge in the global marketplace.
