Advanced Synthesis of Stable 25-Hydroxyvitamin D2 Side Chain Intermediates for Commercial Scale-Up
Introduction to Next-Generation Vitamin D Intermediate Technology
The pharmaceutical landscape for treating secondary hyperparathyroidism and osteoporosis relies heavily on the efficient production of 25-hydroxyvitamin D2 analogues, such as Paricalcitol and 25-hydroxy Vitamin D2. A pivotal breakthrough in this domain is detailed in patent CN101955498A, which introduces a novel class of side chain intermediates characterized by a phosphine oxide structure. Unlike traditional reagents that have plagued the industry with stability issues and purification nightmares, these new compounds, represented generally by Formula 1, exhibit exceptional chemical stability and are amenable to long-term storage without significant degradation. This technological leap is not merely an academic exercise but a critical enabler for reliable pharmaceutical intermediate supplier networks aiming to secure the global supply of life-saving renal medications. The patent explicitly highlights that these intermediates serve as the universal building blocks for a series of bioactive molecules, streamlining the manufacturing process for diverse analogues including Zemplar and 1-alpha,25-dihydroxyvitamin D2.
The strategic importance of this invention cannot be overstated for procurement and R&D teams alike. By shifting the paradigm from unstable sulfone or ylide reagents to robust phosphine oxide derivatives, the industry can achieve substantial cost savings in vitamin D manufacturing through reduced waste and simplified logistics. The structural versatility of Formula 1 allows for various protecting groups (R) such as silyl ethers, acyl groups, or alkoxyalkyl chains, providing chemists with the flexibility to optimize downstream coupling reactions. This adaptability ensures that the synthesis can be tailored to specific facility capabilities while maintaining high purity standards essential for regulatory compliance. As we delve deeper into the technical specifics, it becomes clear that this innovation addresses the core bottlenecks that have historically limited the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
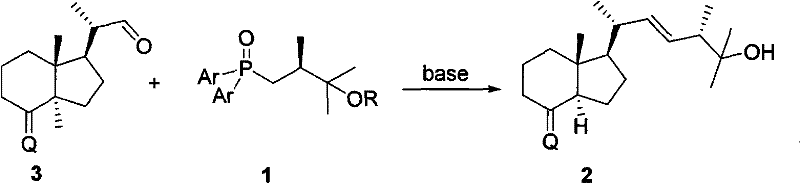
Historically, the construction of the C25-hydroxylated side chain for Vitamin D2 medicines has been fraught with significant technical hurdles, primarily revolving around the instability and reactivity of the coupling reagents. As illustrated in the prior art, the industry has relied heavily on sulfone derivatives (Compound 4) and phosphonium ylide salts (Compound 5) to forge the critical carbon-carbon bond. However, the utilization of sulfone 4 is notoriously inefficient, often resulting in very low yields that render the process economically unviable for large-scale production. The low conversion rates necessitate extensive recycling of starting materials or result in massive material loss, driving up the cost of goods sold and creating supply chain vulnerabilities. Furthermore, the purification of reaction mixtures containing sulfone byproducts is often cumbersome, requiring multiple chromatographic steps that are difficult to translate from the laboratory to multi-ton manufacturing suites.
Similarly, the alternative approach using phosphonium ylide salt 5 presents its own set of formidable challenges. While ylides are powerful reagents, they are inherently unstable and typically must be generated in situ under strictly anhydrous and often cryogenic conditions. This requirement for immediate consumption precludes the possibility of stockpiling the reagent, forcing manufacturers to operate in a Just-In-Time mode that is highly susceptible to operational disruptions. Moreover, the synthesis and purification of the ylide precursor itself are difficult, often leading to impurities that carry over into the final drug substance. The sensitivity of these ylides to moisture and oxygen also imposes severe constraints on the choice of solvents and equipment, increasing the capital expenditure required for specialized reactors. These cumulative limitations create a fragile supply chain for high-purity vitamin D intermediates, where any deviation in process parameters can lead to batch failures.
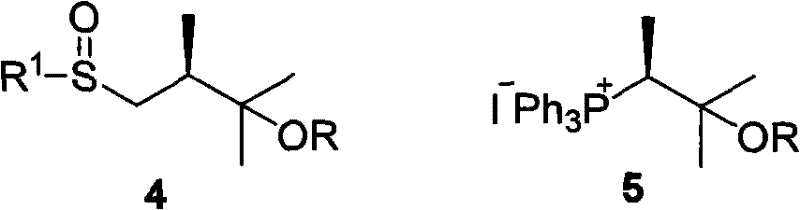
The Novel Approach
In stark contrast to these legacy methods, the novel approach disclosed in CN101955498A utilizes a phosphine oxide intermediate (Compound 1) that fundamentally alters the risk profile of the synthesis. This new reagent is chemically robust, allowing it to be synthesized, isolated, purified, and stored for extended periods without loss of potency. This stability is a game-changer for supply chain reliability, as it decouples the production of the side chain from the final coupling step, allowing for independent quality control and inventory management. The coupling reaction, depicted in the transformation of Compound 1 and Compound 3 to Compound 2, proceeds with high efficiency under basic conditions, delivering the desired Vitamin D analogue with superior stereocontrol. The use of phosphine oxides eliminates the need for the harsh conditions associated with ylide generation, thereby reducing the energy footprint and safety risks of the manufacturing process.
Furthermore, the novel approach offers a streamlined pathway that significantly reduces the number of unit operations required to reach the final intermediate. The ease of purification associated with phosphine oxide byproducts means that crystallization or simple extraction techniques can often replace complex column chromatography, facilitating the transition to continuous manufacturing or large-batch processing. This simplicity directly translates to cost reduction in vitamin D manufacturing by minimizing solvent consumption and labor hours. The versatility of the R group in Formula 1 allows manufacturers to select protecting groups that are optimal for their specific downstream deprotection strategies, whether that involves acid labile silyl groups or base labile esters. This flexibility ensures that the technology can be integrated into existing production lines with minimal retrofitting, accelerating the time to market for generic versions of blockbuster renal drugs.
Mechanistic Insights into Phosphine Oxide Side Chain Synthesis
The synthesis of the target phosphine oxide Compound 1 is a masterpiece of modern organic synthesis, designed to maximize atom economy while preserving the critical chirality at the C20 position. The route commences with a readily available chiral starting material, (R)-2-methyl-3-hydroxymethyl propionate (Compound 6), which serves as the foundation for the stereocenter. The first critical transformation involves the addition of a methyl metal reagent, such as methylmagnesium bromide or methyllithium, to the ester functionality. This Grignard-type addition converts the ester into a tertiary alcohol (Compound 7), effectively installing the gem-dimethyl group characteristic of the Vitamin D side chain. Careful control of temperature and stoichiometry during this exothermic step is paramount to prevent epimerization of the adjacent chiral center, ensuring that the optical purity of the final drug substance remains intact. The resulting diol contains both a primary and a tertiary hydroxyl group, setting the stage for selective functionalization.
Following the formation of the diol, the synthesis proceeds through a sequence of selective protection and activation steps. The primary alcohol is selectively converted into a leaving group, such as a halide (iodide or bromide) or a sulfonate ester (tosylate or mesylate), to yield Compound 8 or 9. This activation is crucial for the subsequent carbon-phosphorus bond formation. Simultaneously, the more hindered tertiary hydroxyl group is protected using a variety of masking groups, including silyl ethers (e.g., triethylsilyl), acyl groups (e.g., benzoyl), or alkoxyalkyl ethers (e.g., MOM or THP). This orthogonal protection strategy prevents the tertiary alcohol from interfering with the nucleophilic substitution at the primary carbon. The final and most defining step involves the reaction of the activated alkyl halide with a diaryl phosphine or a phosphite ester. This Arbuzov-type or nucleophilic substitution reaction installs the phosphorus moiety, which is subsequently oxidized to the phosphine oxide state. The result is a highly stable, crystalline or oily intermediate that is ready for the final coupling with the Vitamin D A-ring fragment.
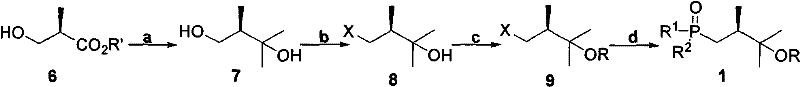
Impurity control in this synthesis is achieved through the rigorous selection of reagents and the inherent selectivity of the reaction steps. The use of chiral pool starting materials ensures that the stereochemistry is established early and maintained throughout the sequence. The protection of the tertiary alcohol not only prevents side reactions but also facilitates purification, as the protected intermediates often have distinct polarity profiles compared to unreacted starting materials or over-alkylated byproducts. Additionally, the phosphine oxide functionality itself acts as a polar handle that aids in the removal of non-polar hydrocarbon impurities during workup. The robustness of the phosphine oxide bond means that the intermediate can withstand the rigors of isolation and drying without decomposition, a stark contrast to the fragile nature of ylide precursors. This mechanistic robustness is the key to achieving the high purity specifications required for pharmaceutical applications, minimizing the burden on downstream purification processes.
How to Synthesize 25-Hydroxyvitamin D2 Side Chain Efficiently
The practical implementation of this synthesis route requires a deep understanding of organometallic chemistry and protection group strategies. The process begins with the careful addition of methyl Grignard reagents to chiral esters at controlled low temperatures to manage exotherms and preserve stereochemistry. Following the formation of the diol, selective activation of the primary alcohol is performed using sulfonyl chlorides or halogenating agents, followed by the installation of a robust protecting group on the tertiary alcohol. The final coupling with phosphorus reagents is conducted under anhydrous conditions to ensure high conversion to the phosphine oxide. For a detailed, step-by-step protocol including specific molar ratios, solvent choices, and workup procedures, please refer to the standardized guide below.
- Grignard addition to chiral hydroxy esters to form diol intermediates, followed by selective halogenation or sulfonylation of the primary alcohol.
- Protection of the tertiary hydroxyl group using silyl, acyl, or alkoxyalkyl protecting groups to prevent side reactions.
- Formation of the carbon-phosphorus bond via reaction with diaryl phosphine or phosphite, followed by oxidation to yield the target phosphine oxide.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of this novel phosphine oxide technology offers profound commercial advantages that extend far beyond the laboratory bench, directly impacting the bottom line and operational resilience of pharmaceutical manufacturers. For procurement managers, the shift away from unstable ylides and low-yielding sulfones represents a significant opportunity for cost optimization. The ability to purchase or manufacture the side chain intermediate in bulk and store it safely eliminates the need for complex, on-demand reagent generation systems. This decoupling of supply and demand allows for strategic sourcing, where intermediates can be procured during periods of low raw material costs and held in inventory, buffering the production schedule against market volatility. Furthermore, the high yield and simplified purification reduce the consumption of expensive solvents and chromatography media, driving down the variable costs associated with each kilogram of produced API.
From a supply chain perspective, the enhanced stability of the phosphine oxide intermediate drastically improves supply continuity. Traditional ylide-based processes are prone to batch failures due to moisture ingress or reagent degradation, leading to unpredictable lead times and potential drug shortages. In contrast, the robust nature of Compound 1 ensures consistent quality and availability, enabling manufacturers to commit to stricter delivery schedules with their customers. The shortened synthesis route also means fewer processing steps, which reduces the overall cycle time from raw material intake to finished intermediate. This efficiency gain allows for faster response to surges in demand, such as those seen during flu seasons or changes in clinical guidelines for renal disease. Additionally, the reduced environmental footprint, stemming from higher atom economy and less waste generation, aligns with increasingly stringent global regulations on pharmaceutical manufacturing emissions.
- Cost Reduction in Manufacturing: The elimination of cryogenic ylide generation and the replacement of low-yielding sulfone couplings with high-efficiency phosphine oxide reactions leads to substantial operational savings. By removing the need for specialized low-temperature reactors and reducing the volume of waste solvents requiring disposal, the overall cost of goods is significantly lowered. The high purity of the crude product minimizes the need for resource-intensive recrystallization or chromatography steps, further enhancing economic efficiency.
- Enhanced Supply Chain Reliability: The chemical stability of the phosphine oxide intermediate allows for the creation of safety stocks, mitigating the risk of production stoppages due to reagent failure. This reliability ensures a steady flow of materials to the final API synthesis stage, preventing bottlenecks that could delay the release of finished drugs to the market. The use of common, commercially available starting materials also reduces dependency on niche suppliers, diversifying the supply base and enhancing resilience.
- Scalability and Environmental Compliance: The streamlined synthetic route is inherently more scalable, as it avoids the heat transfer and mixing challenges associated with cryogenic ylide formations. This facilitates the transition from pilot plant to commercial scale without the need for major process re-engineering. Moreover, the reduction in hazardous waste and solvent usage supports sustainability goals, making the process more compliant with green chemistry principles and environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel side chain technology. These answers are derived directly from the patent data and practical experience in scaling similar phosphorus-mediated coupling reactions. Understanding these nuances is critical for R&D directors evaluating the feasibility of technology transfer and for procurement teams assessing the long-term viability of the supply source.
Q: Why are phosphine oxide intermediates preferred over sulfones for Vitamin D synthesis?
A: Phosphine oxide intermediates offer significantly higher stability during storage and handling compared to traditional sulfone reagents. Furthermore, the coupling reactions using phosphine oxides typically proceed with higher yields and generate byproducts that are much easier to remove during purification, reducing overall processing time.
Q: What represents the key challenge in synthesizing the side chain for Paricalcitol?
A: The primary challenge lies in maintaining the stereochemical integrity of the chiral centers while introducing the phosphorus functionality. Conventional methods often suffer from racemization or low yields during the formation of the carbon-phosphorus bond, which this novel route addresses through optimized protection strategies.
Q: How does this new synthesis route impact commercial scalability?
A: The new route utilizes readily available starting materials and avoids the use of highly unstable ylides that require cryogenic conditions and immediate use. This stability allows for batch production and inventory stocking, drastically simplifying supply chain logistics and enabling seamless scale-up from pilot to commercial tonnage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 25-Hydroxyvitamin D2 Side Chain Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of essential medicines. Our team of expert chemists has extensively evaluated the technology disclosed in CN101955498A and possesses the capability to execute this synthesis with precision and scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications for every batch of phosphine oxide intermediate we produce. We understand that in the pharmaceutical industry, quality is non-negotiable, and our commitment to excellence is embedded in every step of our manufacturing process.
We invite you to collaborate with us to leverage this advanced technology for your Vitamin D projects. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable supply chain partner dedicated to driving innovation and efficiency in the production of life-saving pharmaceutical intermediates.
