Revolutionizing Agrochemical Intermediate Production with Low-Cost Epoxidation Technology
Revolutionizing Agrochemical Intermediate Production with Low-Cost Epoxidation Technology
The global demand for high-efficacy agricultural agents continues to drive innovation in the synthesis of complex heterocyclic compounds, particularly azole derivatives which serve as critical fungicides and plant growth regulators. Patent CN116171273A introduces a groundbreaking manufacturing method that addresses the longstanding economic and operational bottlenecks associated with producing these valuable intermediates. By fundamentally re-engineering the epoxidation and precursor synthesis steps, this technology eliminates the reliance on prohibitively expensive reagents such as trimethylsulfoxonium bromide (TMSOB) and elemental iodine. For R&D directors and procurement strategists, this represents a pivotal shift towards more sustainable and cost-efficient supply chains. The patent outlines a robust pathway that not only reduces raw material expenditures but also enhances process safety by operating under alkaline conditions with readily available sulfur-based reagents. This technical advancement positions manufacturers to offer high-purity agrochemical intermediates at a fraction of the traditional cost, ensuring competitiveness in a volatile market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for azole derivative intermediates have long been plagued by the necessity of using high-cost specialty reagents that strain manufacturing budgets and complicate supply logistics. Specifically, prior art methods, such as those disclosed in International Publication No. 2019/093522, rely heavily on trimethylsulfoxonium bromide (TMSOB) for the critical epoxidation step, alongside iodine and methyl iodide for functional group transformations. These reagents are not only expensive to procure but often require specialized handling and storage due to their reactivity and potential hazards. Furthermore, the conventional bromination and esterification processes frequently suffer from the formation of insoluble white deposits within reaction vessels, leading to equipment clogging and necessitating frequent, labor-intensive cleaning cycles. This operational inefficiency translates directly into increased downtime and higher overhead costs, making the conventional approach less viable for large-scale commercial production where margin compression is a constant threat.
The Novel Approach
The innovative methodology presented in this patent circumvents these historical constraints by substituting costly reagents with economically superior alternatives that maintain high reaction efficiency. Instead of TMSOB, the process utilizes a combination of dimethyl sulfide and dimethyl sulfoxide (DMSO) in the presence of an inorganic base to achieve the necessary epoxidation, effectively generating the active sulfonium species in situ. Additionally, the synthesis of the ketoester precursor replaces iodine with bromine and utilizes simple alcohols for esterification rather than methyl iodide. This strategic substitution drastically lowers the bill of materials while simplifying the reaction workflow. By integrating these changes, the novel approach not only achieves significant cost reduction in agrochemical manufacturing but also improves the overall safety profile of the operation by avoiding the handling of volatile iodine species. The result is a streamlined, scalable process that delivers the target epoxy derivatives with improved yield and purity profiles.
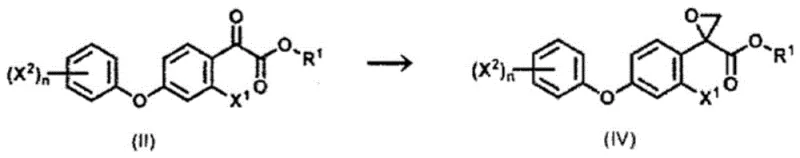
Mechanistic Insights into In-Situ Sulfonium Salt Formation and Bromination
The core chemical innovation lies in the simultaneous generation of the sulfonium salt and the subsequent epoxidation reaction within a single reaction vessel, eliminating the need for isolating unstable intermediates. In this mechanism, dimethyl sulfide or dimethyl sulfoxide reacts with a methylating agent, such as dimethyl sulfate (methyl-LG), in the presence of an inorganic base like potassium carbonate. This interaction generates the active sulfonium ylide species directly in the reaction mixture, which then immediately attacks the carbonyl group of the ketoester derivative to form the epoxide ring. This telescoped process minimizes material loss and exposure to air or moisture, which are common causes of yield degradation in multi-step syntheses. The use of an inorganic base ensures that the reaction environment remains sufficiently alkaline to drive the deprotonation required for ylide formation, while the choice of solvent, typically dichloroethane, provides optimal solubility for both organic substrates and inorganic salts. This mechanistic elegance allows for precise control over reaction kinetics, ensuring high conversion rates without the need for cryogenic conditions often associated with sensitive organometallic reagents.
Furthermore, the patent details a sophisticated approach to impurity control during the precursor synthesis stage, specifically addressing the issue of reactor fouling. During the bromination of the methyl ketone derivative in DMSO, side reactions can lead to the precipitation of white solid deposits that adhere to reactor walls and agitators. The invention identifies that the addition of specific compounds, most notably urea, effectively suppresses the formation of these deposits. Mechanistically, urea likely interacts with the brominating species or the byproduct acids to stabilize the reaction medium, preventing the polymerization or precipitation of insoluble materials. This additive strategy is crucial for maintaining heat transfer efficiency and mixing homogeneity throughout the batch cycle. By preventing physical blockages, the process ensures consistent reaction temperatures and reduces the risk of hot spots that could lead to decomposition. This level of process robustness is essential for maintaining stringent purity specifications required for downstream pharmaceutical or agrochemical applications.
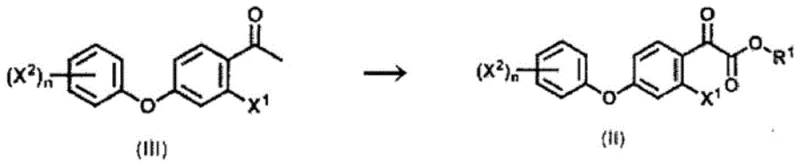
How to Synthesize Epoxy Ester Derivatives Efficiently
The synthesis of these high-value epoxy ester derivatives follows a logical sequence designed to maximize atom economy and minimize waste generation. The process begins with the conversion of a substituted acetophenone derivative into a ketoester via bromination and subsequent esterification, followed by the critical epoxidation step using the novel sulfur-based reagent system. Detailed operational parameters, including temperature gradients, reagent addition rates, and workup procedures, are critical for achieving the reported yields of over 90% in optimized examples. The protocol emphasizes the importance of controlled addition of reagents to manage exotherms and ensure complete conversion. For a comprehensive understanding of the specific molar ratios, solvent volumes, and purification techniques required to replicate this success in a pilot or production setting, operators should refer to the standardized synthesis guidelines provided below.
- Convert methyl ketone derivatives to ketoester derivatives using bromine and dimethyl sulfoxide in the presence of urea to prevent clogging.
- Perform epoxidation on the ketoester derivative using dimethyl sulfide, dimethyl sulfoxide, and a methylating agent like dimethyl sulfate with an inorganic base.
- React the resulting epoxy derivative with 1,2,4-triazole and an inorganic base to form the final azole derivative intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented technology offers tangible benefits that extend far beyond simple raw material substitution. The primary advantage is the drastic reduction in the cost of goods sold (COGS) achieved by replacing premium-priced reagents like TMSOB and iodine with commodity chemicals such as dimethyl sulfide, DMSO, and bromine. These commodities are produced on a massive global scale, ensuring stable pricing and reliable availability even during periods of market volatility. This shift mitigates the risk of supply disruptions that often plague specialized chemical markets, allowing for more accurate long-term forecasting and inventory planning. Moreover, the elimination of iodine-based chemistry reduces the environmental burden associated with heavy metal waste disposal, aligning production practices with increasingly strict global environmental regulations and sustainability goals.
- Cost Reduction in Manufacturing: The replacement of expensive proprietary reagents with bulk commodity chemicals fundamentally alters the cost structure of the manufacturing process. By utilizing dimethyl sulfide and dimethyl sulfoxide instead of TMSOB, manufacturers avoid the high licensing fees and synthesis costs associated with the latter. Additionally, the use of bromine instead of iodine for the halogenation step leverages the significant price differential between these two halogens, resulting in substantial savings on the bill of materials. The process also reduces the consumption of auxiliary reagents through optimized addition protocols, further driving down variable costs. These cumulative savings allow for more competitive pricing strategies in the final agrochemical market while preserving healthy profit margins for the manufacturer.
- Enhanced Supply Chain Reliability: Relying on widely available industrial solvents and reagents significantly de-risks the supply chain compared to depending on niche specialty chemicals. Dimethyl sulfoxide and dichloroethane are standard solvents with multiple qualified suppliers globally, ensuring that production schedules are not held hostage by single-source bottlenecks. The simplified reagent profile also reduces the complexity of hazardous material logistics and storage requirements. This reliability is crucial for meeting just-in-time delivery commitments to downstream formulators and ensures business continuity even when specific supply lanes face temporary constraints. The robust nature of the chemistry means that production can be easily transferred between different manufacturing sites without significant requalification efforts.
- Scalability and Environmental Compliance: The suppression of reactor fouling through the use of urea additives directly translates to improved scalability and operational uptime. In large-scale reactors, heat transfer is critical, and the accumulation of solid deposits can severely compromise cooling efficiency, posing safety risks and limiting batch sizes. By preventing this fouling, the process enables larger batch sizes and longer campaign runs without the need for frequent shutdowns for cleaning. This operational efficiency reduces labor costs and increases overall plant throughput. Furthermore, the avoidance of iodine and the use of alkaline conditions simplify wastewater treatment protocols, reducing the cost and complexity of environmental compliance and waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this technology into their existing production portfolios. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a factual basis for decision-making. These insights cover aspects ranging from reagent selection to impurity management, ensuring a holistic view of the process capabilities.
Q: How does this new method reduce production costs compared to conventional routes?
A: The method replaces expensive reagents like trimethylsulfoxonium bromide (TMSOB), iodine, and methyl iodide with cheaper alternatives such as dimethyl sulfide, dimethyl sulfoxide, and bromine, significantly lowering raw material expenses.
Q: What specific technical challenge does the addition of urea address in the synthesis?
A: Urea is added during the bromination step to suppress the formation of white deposits that typically clog reaction vessels, thereby improving operational efficiency and reducing maintenance downtime.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common industrial solvents like dichloroethane and avoids hazardous or hard-to-source reagents, making it highly scalable and safer for commercial production environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azole Derivative Intermediate Supplier
As the agrochemical industry evolves, the ability to produce complex intermediates efficiently and sustainably becomes a key differentiator for market leadership. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies like the one described in CN116171273A to deliver superior value to our global partners. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of azole derivative intermediate meets the highest international standards. Our commitment to quality and consistency makes us the preferred partner for companies seeking to optimize their supply chains without compromising on product performance.
We invite you to explore how our optimized manufacturing processes can enhance your product portfolio and improve your bottom line. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our expertise in cost-effective synthesis can provide you with a competitive edge in the global agrochemical market, ensuring a reliable supply of high-quality intermediates for your future growth.
