Advanced Imidazolium Fluoride Fluorinating Agents for Scalable Pharmaceutical Manufacturing
The landscape of organofluorine chemistry is undergoing a significant transformation driven by the urgent need for safer, more efficient, and environmentally benign fluorinating reagents. Patent CN1898187A, titled "Fluorinating agent and method for producing fluorine-containing compound using same," represents a pivotal advancement in this domain, introducing a novel class of alkyl-substituted imidazolium salts containing fluoride ions. This technology addresses the critical bottlenecks associated with traditional fluorination methods, particularly the reliance on hazardous hydrogen fluoride (HF) or poorly soluble inorganic fluorides like potassium fluoride (KF). For R&D directors and process chemists in the pharmaceutical and electronic materials sectors, this patent offers a robust pathway to synthesize high-purity fluorine-containing organic compounds, which are essential building blocks for modern APIs and advanced functional materials. The core innovation lies in the tunable nature of the imidazolium cation, which not only solubilizes the fluoride anion effectively but also allows for the modulation of physical properties such as melting point through anion mixing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of fluorine atoms into organic molecules has been fraught with significant operational and safety challenges. Traditional nucleophilic fluorination often relies on alkali metal fluorides such as potassium fluoride (KF) or cesium fluoride (CsF). While inexpensive, these reagents suffer from poor solubility in organic solvents, necessitating the use of phase transfer catalysts or high-boiling polar aprotic solvents like DMF and DMSO, which complicate downstream purification and waste treatment. Furthermore, alternative methods utilizing hydrogen fluoride (HF) or its adducts, while effective, pose severe safety risks due to the extreme corrosivity and toxicity of HF, requiring specialized reactor materials and rigorous safety protocols that drastically increase capital expenditure. Additionally, the generation of stoichiometric amounts of inorganic salt waste from these processes creates substantial environmental burdens, conflicting with the growing industry mandate for greener chemistry practices.
The Novel Approach
The methodology disclosed in CN1898187A circumvents these issues by employing imidazolium-based ionic liquids as the fluorinating medium. By replacing the inorganic cation with an organic imidazolium cation, the fluoride ion becomes highly soluble in a wide range of organic media, enhancing reaction kinetics and selectivity without the need for auxiliary phase transfer agents. A key feature of this approach is the ability to create mixed-anion systems where the fluoride ion coexists with other anions like chloride or bromide in a defined ratio (0 < x ≤ 1). This structural flexibility allows chemists to tailor the melting point of the reagent, potentially creating liquids at room temperature that are easy to pump and dose in continuous flow reactors. This shift from solid, hygroscopic inorganic salts to tunable ionic liquids represents a paradigm shift in process design, enabling cleaner reaction profiles and simplified work-up procedures.
Mechanistic Insights into Imidazolium-Mediated Nucleophilic Fluorination
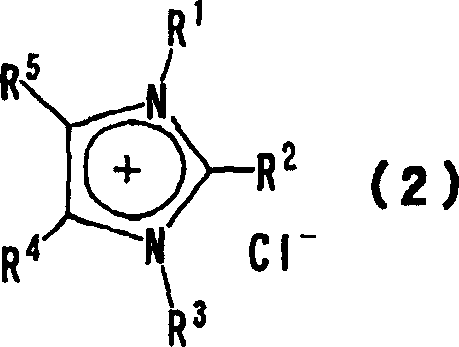
The efficacy of the imidazolium fluoride reagent stems from the unique interaction between the bulky, charge-delocalized imidazolium cation and the small, hard fluoride anion. In conventional inorganic salts, the strong lattice energy holds the fluoride tightly, rendering it less nucleophilic until high energies are applied. In the imidazolium system, the lattice energy is significantly disrupted, especially when mixed anions are present, effectively "naked" the fluoride ion and increasing its nucleophilicity. The reaction proceeds via a classic SN2 mechanism where the fluoride ion attacks the electrophilic carbon center of the substrate (Formula 6), displacing a leaving group such as a halide or sulfonate. The imidazolium cation acts as a spectator ion that stabilizes the transition state through electrostatic interactions without participating directly in bond breaking, thereby minimizing side reactions such as elimination which are common with basic inorganic fluorides.
Furthermore, the patent details a sophisticated control mechanism over the reagent's physical state through the variable 'x' in Formula 1. When x is close to 1, the material behaves more like a pure salt with a higher melting point. However, by intentionally maintaining x in the range of 0.4 to 0.9, the crystalline lattice is disrupted by the presence of competing anions (e.g., chloride), resulting in a eutectic-like mixture that remains liquid at lower temperatures. This is crucial for industrial applications where pumping viscous solids is impractical. The synthesis of these reagents can be achieved through salt metathesis, typically reacting an imidazolium chloride precursor with silver fluoride (AgF) or potassium fluoride (KF). The use of AgF drives the reaction to completion via the precipitation of insoluble silver chloride, ensuring high purity of the resulting fluorinating agent, while the KF route offers a more cost-effective alternative for large-scale production where trace silver contamination is less critical.
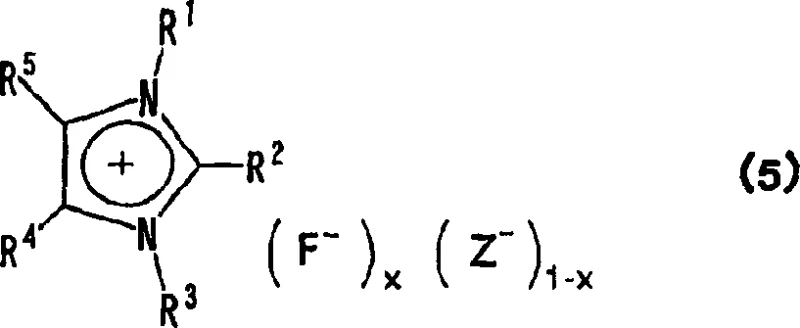
How to Synthesize Imidazolium Fluoride Efficiently
The preparation of these advanced fluorinating agents is designed to be operationally simple, leveraging standard unit operations familiar to any chemical manufacturing facility. The process generally involves dissolving the imidazolium chloride precursor in a suitable solvent such as water or methanol, followed by the addition of the fluoride source. The reaction conditions are mild, typically proceeding at temperatures between -20°C and 200°C, allowing for flexibility depending on the thermal stability of the specific imidazolium substituents. Following the ion exchange, the insoluble byproduct (silver chloride or potassium chloride) is removed via filtration, and the filtrate is concentrated to yield the active fluorinating agent.
- Dissolve alkyl-substituted imidazolium chloride in water or methanol solvent.
- Add silver fluoride (AgF) or potassium fluoride (KF) to the solution and stir at temperatures between -20°C and 200°C.
- Filter off precipitated silver chloride or potassium salts, then concentrate the filtrate to obtain the fluorinating agent.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology translates into tangible risk mitigation and cost optimization strategies. The primary advantage lies in the drastic reduction of hazard management costs associated with handling toxic fluorinating agents. By eliminating the need for hydrogen fluoride, facilities can avoid the expensive infrastructure required for HF containment, neutralization, and emergency response, leading to substantial capital and operational expenditure savings. Moreover, the improved solubility and reactivity of the imidazolium fluoride reagents often result in higher reaction yields and fewer byproducts, which directly reduces the cost of raw materials per kilogram of final product. The ability to recycle the imidazolium cation further enhances the economic viability, as the expensive organic component can be recovered from the reaction waste stream and regenerated, creating a closed-loop system that minimizes waste disposal fees.
- Cost Reduction in Manufacturing: The transition to imidazolium-based fluorination eliminates the need for expensive phase transfer catalysts and reduces solvent consumption due to higher reagent solubility. The process operates under milder conditions compared to traditional high-temperature KF fluorinations, resulting in lower energy consumption for heating and cooling. Additionally, the simplified work-up procedure, often requiring only extraction and concentration rather than complex chromatographic purifications, significantly reduces downstream processing time and labor costs.
- Enhanced Supply Chain Reliability: Unlike hydrogen fluoride, which is subject to strict transportation regulations and limited supplier availability due to its hazardous nature, the precursors for imidazolium fluorides (imidazolium chlorides and simple fluoride salts) are commodity chemicals with robust global supply chains. This ensures consistent availability and reduces the risk of production stoppages caused by regulatory delays in raw material shipping. The stability of the reagent also allows for longer shelf-life and safer storage, reducing inventory write-offs.
- Scalability and Environmental Compliance: The liquid nature of the optimized reagents (when x is tuned) makes them ideal for scale-up in continuous flow reactors, which offer superior heat and mass transfer compared to batch processing of solid slurries. From an environmental perspective, the process generates significantly less hazardous waste, as the byproduct is typically a benign inorganic salt (like KCl or AgCl) rather than acidic fluoride waste streams. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, facilitating easier permitting for new manufacturing lines.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this fluorination technology. These insights are derived directly from the experimental data and embodiments provided in the patent documentation, ensuring accuracy and relevance for process development teams.
Q: What are the safety advantages of imidazolium fluoride over hydrogen fluoride?
A: Unlike highly corrosive and toxic hydrogen fluoride (HF), imidazolium fluoride salts are ionic liquids that can be handled with significantly reduced risk, eliminating the need for specialized HF-resistant equipment.
Q: Can the melting point of the fluorinating agent be adjusted?
A: Yes, by adjusting the ratio of fluoride ions to other anions (represented by variable x in Formula 1), the melting point can be tuned to be below room temperature, facilitating easier handling and pumping.
Q: Is the imidazolium cation recoverable after the reaction?
A: Yes, the patent describes methods to recover the mixed alkyl-substituted imidazolium salt from the reaction mixture and regenerate it into the active fluorinating agent for reuse.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazolium Fluoride Supplier
As the demand for fluorinated intermediates continues to surge across the pharmaceutical and agrochemical industries, partnering with a technically proficient manufacturer is essential for maintaining competitive advantage. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our stringent purity specifications and rigorous QC labs guarantee that every batch of fluorinating agent meets the exacting standards required for GMP-compliant API synthesis, minimizing the risk of batch failures due to reagent impurities.
We invite you to leverage our technical expertise to optimize your fluorination processes. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific synthetic route. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our imidazolium fluoride solutions can enhance your production efficiency and safety profile.
