Revolutionizing Alkynone Production: A Dual-Catalyst Strategy for Commercial Scale-Up
The landscape of organic synthesis is constantly evolving, driven by the demand for more efficient and sustainable pathways to complex molecular architectures. A pivotal advancement in this domain is documented in patent CN108752174B, which discloses a sophisticated catalytic synthesis method for alkynone compounds. These unsaturated ketones serve as critical building blocks in the construction of heterocyclic systems and bioactive natural products, holding immense value for the pharmaceutical and agrochemical industries. The core innovation lies in the deployment of a unique dual-component composite catalyst system, comprising an organic palladium compound and ferrocene, which operates in synergy with specific nitrogen-containing ligands and oxidants. This technical breakthrough allows for the direct oxidative coupling of benzylamine derivatives with aryl alkynes, bypassing the multi-step sequences often required in traditional methodologies. By leveraging this robust catalytic cycle, manufacturers can achieve exceptional conversion rates under relatively mild thermal conditions, marking a significant leap forward in process chemistry efficiency.
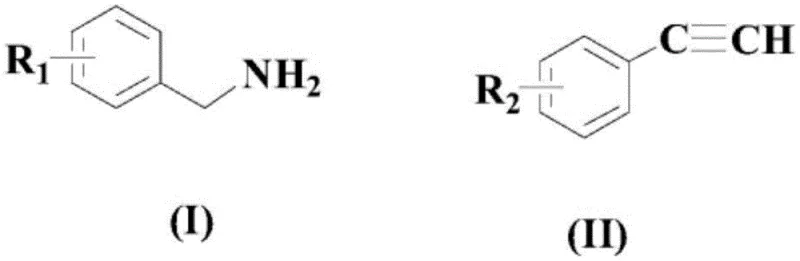
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
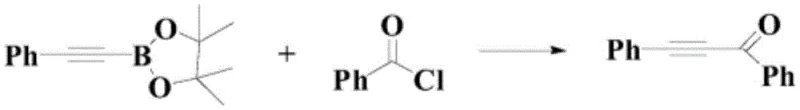
Historically, the synthesis of conjugated alkynones has been fraught with challenges that hinder industrial scalability and economic viability. Prior art methods, such as those reported by Yasuuchi Nishihara et al., often rely on the cross-coupling of alkyl boronates with acid chlorides, necessitating the use of stoichiometric amounts of copper salts and strictly anhydrous conditions. As illustrated in earlier literature, these pathways frequently involve unstable reagents that pose significant safety hazards during storage and handling. Furthermore, alternative approaches utilizing Lewis acid catalysis with potassium alkynyltrifluoroborates often suffer from limited functional group tolerance, leading to side reactions that complicate downstream purification. The reliance on harsh reaction environments not only increases energy consumption but also generates substantial chemical waste, creating a burden on environmental compliance protocols. Additionally, many conventional routes require pre-functionalized starting materials, adding extra synthetic steps that erode overall atom economy and drive up the cost of goods sold.
The Novel Approach
In stark contrast to these legacy techniques, the methodology outlined in the present invention introduces a streamlined, one-pot oxidative coupling strategy that fundamentally reshapes the production landscape. By utilizing readily accessible benzylamines and terminal aryl alkynes as direct substrates, the process eliminates the need for pre-activated organometallic reagents. The introduction of the palladium-ferrocene dual catalyst system creates a highly active species capable of facilitating C-C bond formation with remarkable precision. This novel approach operates effectively in polar aprotic solvents like DMSO, which are inexpensive and widely available in bulk quantities. The integration of a specific acidic promoter further accelerates the reaction kinetics, allowing the transformation to proceed to completion within a practical timeframe of 5 to 10 hours. This shift from multi-step, hazardous protocols to a direct catalytic oxidation represents a paradigm shift, offering a cleaner, safer, and more economically attractive route for generating high-value alkynone intermediates.
Mechanistic Insights into Pd-Ferrocene Dual Catalysis
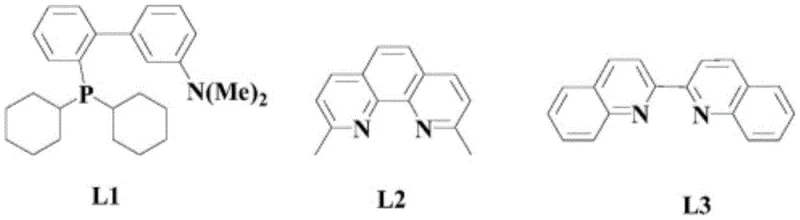
The efficacy of this synthesis hinges on the intricate interplay between the palladium center and the ferrocene co-catalyst, which together orchestrate a complex oxidative cycle. The organic palladium compound, preferably PdCl2(cod), acts as the primary activation site for the alkyne substrate, while ferrocene serves as a redox mediator that facilitates the regeneration of the active palladium species. This synergistic relationship prevents the premature deactivation of the catalyst, a common pitfall in single-metal systems, thereby sustaining high turnover numbers throughout the reaction duration. The presence of the nitrogen-containing ligand, specifically the bidentate structure designated as L1, plays a crucial role in stabilizing the transition state and enforcing regioselectivity. Without this specific ligand architecture, the reaction efficiency drops precipitously, underscoring the importance of steric and electronic tuning in the catalyst design. The mechanism likely proceeds through a palladium-acetylide intermediate, which undergoes nucleophilic attack by the amine-derived species, followed by oxidative elimination to release the final alkynone product.
Beyond the primary catalytic cycle, the control of impurities is meticulously managed through the selection of the oxidizing agent and acidic environment. The use of m-chloroperoxybenzoic acid (m-CPBA) ensures a controlled oxidation potential that minimizes over-oxidation of the sensitive alkyne moiety or the aromatic rings. The acidic promoter, optimally trifluoroacetic acid, protonates intermediate species to facilitate leaving group departure without causing degradation of the acid-sensitive functional groups on the substrate. This delicate balance allows for the tolerance of diverse substituents, including halogens, cyano groups, and alkyl chains, which might otherwise be incompatible with stronger acidic or basic conditions. Consequently, the resulting crude reaction mixture exhibits a simplified impurity profile, significantly reducing the burden on purification units. This mechanistic robustness translates directly into higher isolated yields and consistent batch-to-batch reproducibility, which are critical metrics for regulatory approval in pharmaceutical manufacturing.
How to Synthesize Alkynone Compounds Efficiently
Implementing this advanced synthesis protocol requires precise adherence to the optimized reaction parameters established in the patent data to ensure maximum yield and purity. The process begins with the careful weighing of the benzylamine and aryl alkyne substrates, maintaining a molar ratio that favors the alkyne to drive the equilibrium forward. The dual-catalyst system must be prepared fresh or handled under inert atmosphere to prevent premature oxidation before the reaction commences. Operators should monitor the temperature closely, as the exothermic nature of the oxidation step requires efficient heat dissipation to maintain the optimal 40-60°C window. Detailed standard operating procedures regarding the addition sequence of the oxidant and acid promoter are essential to prevent localized hot spots that could degrade the product. For a comprehensive, step-by-step technical guide on executing this synthesis, please refer to the standardized protocol below.
- Prepare the reaction mixture by combining benzylamine derivatives and aryl alkyne compounds in DMSO solvent with a precise molar ratio.
- Introduce the dual-component catalyst system consisting of an organic palladium compound and ferrocene, along with a nitrogen-containing ligand.
- Add the oxidizing agent and acidic promoter, then heat the mixture to 40-60°C for 5-10 hours to complete the oxidative coupling.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this catalytic technology offers profound advantages that extend well beyond simple yield improvements. The ability to utilize commodity chemicals like benzylamines and phenylacetylene derivatives as starting materials drastically simplifies the raw material sourcing strategy. Unlike specialized organoboron or organosilicon reagents required in older methods, these substrates are produced on a massive global scale, ensuring a stable and resilient supply chain that is less susceptible to market volatility. The elimination of expensive and toxic heavy metal scavengers, often needed to remove residual copper or nickel from traditional couplings, further streamlines the downstream processing workflow. This reduction in unit operations translates directly into lower capital expenditure for equipment and reduced operational costs associated with waste disposal and solvent recovery. Furthermore, the mild reaction conditions reduce the energy load on manufacturing facilities, aligning with modern sustainability goals and carbon footprint reduction initiatives.
- Cost Reduction in Manufacturing: The implementation of this dual-catalyst system fundamentally alters the cost structure of alkynone production by removing the need for costly pre-functionalized reagents. By avoiding the synthesis of unstable intermediates like acid chlorides or boronates, manufacturers can bypass entire production stages, leading to substantial savings in labor and material costs. The high atom economy of the direct oxidative coupling means that a greater proportion of the input mass is converted into valuable product rather than waste byproducts. Additionally, the catalyst loading is remarkably low, and the potential for catalyst recycling or recovery further enhances the economic viability of the process. These factors combine to create a highly competitive cost position for producers adopting this technology, allowing for better margin management in price-sensitive markets.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on widely available feedstock chemicals that are not subject to the same geopolitical or logistical constraints as exotic reagents. The robustness of the reaction against moisture and air, relative to sensitive organometallic methods, reduces the risk of batch failures due to environmental excursions during transport or storage. This reliability ensures consistent delivery schedules to downstream customers, minimizing the risk of production stoppages in their own facilities. The simplified purification process also shortens the overall manufacturing lead time, enabling faster response to fluctuating market demands. Consequently, suppliers utilizing this method can offer more reliable service level agreements and maintain healthier inventory levels without the fear of rapid reagent degradation.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the use of standard stainless steel reactors and common solvents like DMSO, which do not require specialized glass-lined or Hastelloy equipment. The absence of highly pyrophoric reagents simplifies safety protocols and lowers insurance premiums associated with chemical manufacturing. From an environmental standpoint, the generation of less hazardous waste streams simplifies effluent treatment and helps facilities meet increasingly stringent regulatory standards. The high selectivity of the reaction minimizes the formation of isomeric impurities, reducing the solvent volume required for chromatographic purification. This green chemistry profile not only appeals to environmentally conscious partners but also future-proofs the manufacturing asset against tightening global environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this alkynone synthesis technology. These insights are derived directly from the experimental data and optimization studies presented in the patent documentation. Understanding these nuances is vital for R&D teams evaluating the feasibility of integrating this route into their existing portfolios. The answers provided reflect the specific advantages of the Pd-Ferrocene system over alternative methodologies, highlighting its suitability for diverse applications.
Q: What represents the primary advantage of the dual-catalyst system over traditional methods?
A: The dual-catalyst system utilizing palladium and ferrocene offers a synergistic effect that significantly enhances reaction yields and functional group tolerance compared to single-component catalysts or harsh Lewis acid methods found in prior art.
Q: Which oxidizing agents are compatible with this synthesis protocol?
A: While various oxidants like hydrogen peroxide and DDQ were tested, m-chloroperoxybenzoic acid (m-CPBA) demonstrated the highest efficiency and yield consistency for this specific oxidative coupling transformation.
Q: Is this process suitable for large-scale manufacturing of pharmaceutical intermediates?
A: Yes, the process operates at mild temperatures (40-60°C) and utilizes readily available starting materials, making it highly scalable and cost-effective for industrial production of complex alkynone intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkynone Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this dual-catalyst synthesis method for the production of high-purity alkynone intermediates. As a premier CDMO partner, we possess the technical expertise and infrastructure to seamlessly translate this patented laboratory protocol into robust commercial manufacturing processes. Our engineering teams have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the nuances of the Pd-Ferrocene catalytic cycle are perfectly preserved at scale. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the absence of residual metals and organic impurities. Our commitment to quality assurance guarantees that every batch of alkynone compound meets the exacting standards required for pharmaceutical and fine chemical applications.
We invite potential partners to engage with our technical sales team to discuss how this innovative synthesis route can optimize your supply chain and reduce overall manufacturing costs. By leveraging our capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments for your target molecules. Together, we can accelerate your development timelines and bring high-value products to market faster and more efficiently.
