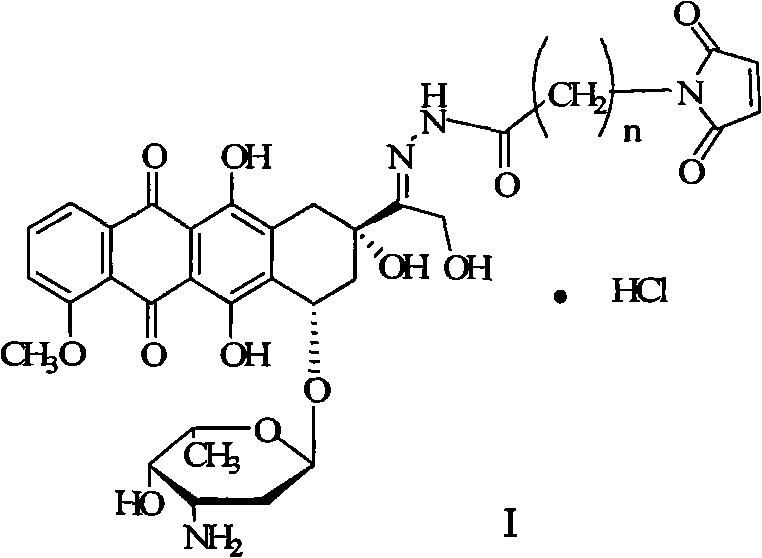
Scalable Synthesis of Doxorubicin 13-Position Hydrazone Derivatives via Maleic Chloride Acylation
Introduction to Advanced Doxorubicin Derivative Synthesis
The pharmaceutical industry continuously seeks robust synthetic pathways for anticancer agents that balance high purity with economic viability. A pivotal advancement in this domain is detailed in patent CN102675385A, which discloses a novel preparation method for Doxorubicin 13-position hydrazone derivatives. This specific class of compounds is critical for the development of antibody-drug conjugates (ADCs) and targeted cancer therapies, where the stability and linkage chemistry determine therapeutic efficacy. The disclosed innovation fundamentally shifts the synthetic paradigm by utilizing maleic chloride and aminoalkyl carboxylic acids as primary starting materials, bypassing the inefficient cyclization steps associated with traditional maleic anhydride routes. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediates supplier options, this methodology represents a significant leap forward in process chemistry, offering a streamlined approach that enhances overall yield while minimizing environmental footprint through reduced solvent consumption.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Zorubicin (Doxorubicin) 13-position hydrazone derivatives has been plagued by inefficiencies that hinder commercial scalability. As illustrated in the background art, conventional routes typically initiate with the reaction of maleic anhydride and 6-aminocaproic acid in large volumes of acetic acid to form a diacid intermediate. This is followed by a cyclization step using agents such as diacetyl oxide or chlorotrimethylsilane to generate the maleimide ring. This specific cyclization step is a major bottleneck, often resulting in poor yields ranging merely from 30% to 40%. Furthermore, the subsequent protection and deprotection sequences require substantial quantities of organic solvents, including tetrahydrofuran (THF) and trifluoroacetic acid (TFA). The cumulative effect of these inefficiencies is a total recovery rate of less than 20%, coupled with an exorbitant solvent-to-oil ratio where every gram of product consumes approximately 70mL of acetic acid and 300mL of THF. Additionally, the necessity for column chromatography to purify intermediates like 2-(6-maleimido) hexanoyl tert-butyl carbazate introduces significant operational complexity and cost, making the traditional process economically unsustainable for large-scale manufacturing.
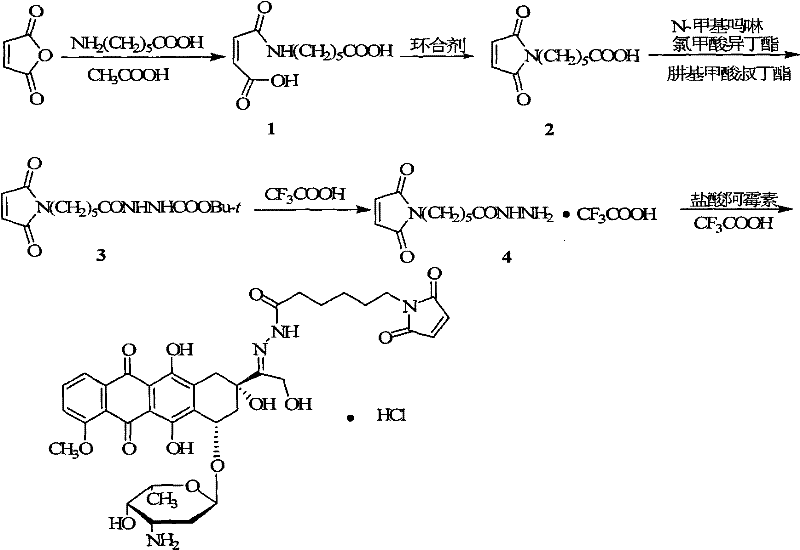
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel approach described in the patent utilizes a direct acylation strategy starting from maleic chloride. This strategic change allows for the direct formation of the maleimide aminoalkyl carboxylic acid intermediate through a condensation reaction under alkaline catalysis, effectively merging the ring formation and acylation into a more efficient sequence. By eliminating the discrete, low-yield cyclization step, the new method achieves a dramatic improvement in the yield of the key maleimide intermediate, reaching up to 90% in optimized embodiments. The downstream processing is equally refined; the conversion to the hydrazide and subsequent deprotection are managed with precise stoichiometric control, avoiding the excessive solvent loads of the past. Crucially, the final condensation with Doxorubicin hydrochloride is conducted under hydrogen chloride catalysis without the introduction of trifluoroacetic acid, which historically contaminated the final product. This results in a simplified purification protocol that relies on recrystallization and washing rather than chromatography, boosting the overall yield to approximately 50%—a more than twofold increase over conventional techniques.
Mechanistic Insights into Maleic Chloride Acylation and Cyclization
The core chemical innovation lies in the reactivity of maleic chloride (fumaryl chloride isomer context) towards nucleophilic attack by aminoalkyl carboxylic acids. In the presence of a base such as potassium carbonate or triethylamine, the amino group of the alkyl carboxylic acid attacks one of the acid chloride moieties of the maleic chloride. This initial nucleophilic acyl substitution forms an amide bond. Unlike the anhydride route which requires a separate dehydration step to close the imide ring, the acid chloride pathway facilitates a spontaneous or base-promoted intramolecular cyclization. The second acid chloride group reacts with the carboxylic acid moiety (often activated in situ or via the specific reaction conditions) or the amide nitrogen undergoes cyclization driven by the release of HCl, which is scavenged by the base. This mechanism ensures the rapid formation of the stable five-membered maleimide ring directly attached to the alkyl chain. The use of maleic chloride essentially pre-activates the dicarboxylic acid equivalent, removing the thermodynamic barrier associated with dehydrating maleic acid derivatives in the traditional route.
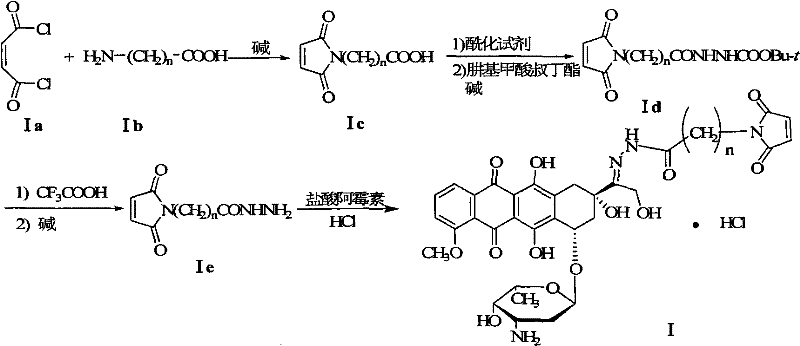
Furthermore, the mechanistic pathway for the hydrazide formation leverages standard peptide coupling logic but optimized for this specific scaffold. The maleimide aminoalkyl carboxylic acid is activated using thionyl chloride to form the corresponding acid chloride in situ. This highly reactive species then undergoes condensation with tert-butyl carbazate. The choice of thionyl chloride is critical as it generates gaseous byproducts (SO2 and HCl), driving the reaction to completion and simplifying the workup by avoiding non-volatile coupling reagents that are difficult to remove. The subsequent deprotection of the tert-butyl group using trifluoroacetic acid proceeds via a standard acid-catalyzed cleavage mechanism, generating the free hydrazine ready for conjugation. By isolating the trifluoroacetic acid salt and neutralizing it prior to the final step, the process ensures that no residual TFA carries over into the final Doxorubicin conjugation, thereby solving a persistent impurity issue found in prior art methods.
How to Synthesize Doxorubicin Hydrazone Derivative Efficiently
The implementation of this synthesis requires careful attention to stoichiometry and moisture control, particularly during the acid chloride formation steps. The process is designed to be telescoped where possible to minimize isolation losses, although the patent describes distinct isolation points for quality control. The initial reaction between maleic chloride and the amino acid must be kept anhydrous to prevent hydrolysis of the acid chloride, typically employing dry chloroform as the solvent. Following the formation of the maleimide intermediate, the activation with thionyl chloride requires reflux conditions to ensure complete conversion. The final conjugation with Doxorubicin hydrochloride is sensitive to light and pH, necessitating an argon shield and lucifuge conditions to prevent degradation of the anthracycline core. For a detailed breakdown of the specific molar ratios, temperature profiles, and workup procedures required to replicate this high-yield process, please refer to the standardized synthesis guide below.
- Condense maleic chloride with aminoalkyl carboxylic acid in the presence of an alkali catalyst to form maleimide aminoalkyl carboxylic acid.
- Activate the carboxylic acid with an acylating reagent like thionyl chloride, then condense with tert-butyl carbazate to form the protected hydrazide.
- Deprotect the hydrazide using trifluoroacetic acid followed by neutralization, then condense with doxorubicin hydrochloride under HCl catalysis to yield the final derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this maleic chloride-based route offers compelling economic and logistical benefits that extend beyond simple yield metrics. The most immediate impact is seen in the drastic reduction of raw material costs and waste disposal fees. By eliminating the need for expensive cyclizing agents like diacetyl oxide and reducing the reliance on vast quantities of acetic acid and THF, the variable cost per kilogram of the intermediate is significantly lowered. The simplification of the purification process is another major cost driver; replacing column chromatography with recrystallization and aqueous washing not only reduces solvent purchase costs but also decreases the time and labor required for production batches. This efficiency gain translates directly into improved throughput capacity, allowing manufacturers to meet high-volume demands without proportional increases in facility footprint or equipment investment.
- Cost Reduction in Manufacturing: The elimination of low-yield steps and expensive reagents drives down the Cost of Goods Sold (COGS). The doubling of the overall yield from under 20% to approximately 50% means that nearly half the starting material is converted to valuable product compared to the quarter or less in old methods. This efficiency inherently reduces the cost burden associated with raw material procurement and waste treatment, providing a substantial margin improvement for the final API or conjugate manufacturer.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as maleic chloride, thionyl chloride, and common amino acids ensures a robust and stable supply chain. Unlike specialized cyclizing agents that may have limited suppliers, these reagents are widely available globally, reducing the risk of supply disruptions. Furthermore, the simplified process flow reduces the number of critical control points, minimizing the risk of batch failures and ensuring consistent on-time delivery of high-purity intermediates to downstream partners.
- Scalability and Environmental Compliance: The significant reduction in solvent usage aligns with increasingly stringent environmental regulations regarding volatile organic compound (VOC) emissions. The process generates less hazardous waste, simplifying compliance and lowering disposal costs. The operational simplicity, characterized by fewer unit operations and the absence of complex chromatography, makes this route highly amenable to scale-up from pilot plant to multi-ton commercial production, ensuring long-term supply continuity for global pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Doxorubicin hydrazone derivatives using this patented methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, aimed at clarifying the feasibility and advantages for potential partners. Understanding these nuances is essential for R&D teams evaluating technology transfer and procurement teams assessing vendor capabilities.
Q: What is the primary advantage of using maleic chloride over maleic anhydride in this synthesis?
A: Using maleic chloride eliminates the need for a separate cyclization step with harsh agents like diacetyl oxide, directly forming the maleimide ring with significantly higher yields (up to 90% vs 30-40%) and reducing solvent consumption.
Q: How does this new method improve the purity profile of the final Doxorubicin derivative?
A: The novel route avoids the introduction of trifluoroacetic acid in the final condensation step, which was a persistent impurity source in conventional methods, thereby simplifying purification and enhancing final product quality.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process replaces complex column chromatography with simple recrystallization and washing steps, drastically reducing solvent usage and operational complexity, making it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Doxorubicin Hydrazone Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality linkers and intermediates play in the success of next-generation oncology therapeutics. Our technical team has extensively analyzed the pathway described in CN102675385A and possesses the expertise to execute this synthesis with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to industrial manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, ensuring that every batch of Doxorubicin hydrazone derivative meets the exacting standards required for clinical and commercial applications.
We invite you to collaborate with us to optimize your supply chain for anticancer drug conjugates. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this advanced synthetic route can reduce your overall project costs. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us partner with you to accelerate the development of life-saving therapies through superior chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
