Optimizing Vitamin B6 Production: A Technical Analysis of Biphasic Aromatization for Commercial Scale-Up
Optimizing Vitamin B6 Production: A Technical Analysis of Biphasic Aromatization for Commercial Scale-Up
The global demand for high-purity vitamins remains a cornerstone of the pharmaceutical and nutritional sectors, with Pyridoxine Hydrochloride (Vitamin B6) serving as a critical active ingredient. A significant technological advancement in this domain is detailed in patent CN102584692A, which introduces a robust preparation method capable of achieving yields exceeding 87 percent. This innovation addresses long-standing inefficiencies in the aromatization step, traditionally a bottleneck in Vitamin B6 synthesis. By shifting from a homogeneous acidic environment to a sophisticated water-insoluble solvent and water biphasic system, the process significantly mitigates the degradation of sensitive intermediates. For R&D directors and supply chain leaders, this represents a pivotal opportunity to enhance process reliability and reduce waste in the manufacturing of essential pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
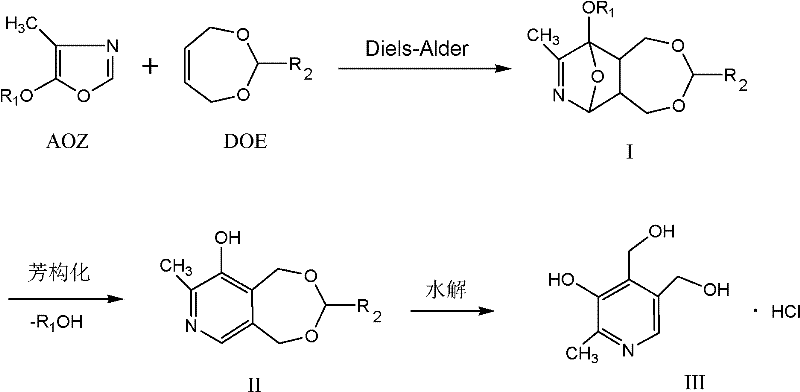
Historically, the industrial synthesis of Pyridoxine Hydrochloride has relied heavily on methods utilizing ethanol, water, and hydrochloric acid as the aromatization medium. While these conditions are relatively mild, they suffer from inherent chemical drawbacks that compromise overall efficiency. In a homogeneous system, the strong acidity of the catalyst can aggressively attack the bicyclic intermediate (Formula I) before it successfully converts to the aromatized product (Formula II). Furthermore, the presence of alcohol in the system can inhibit complete aromatization, leading to the formation of stubborn by-products. Perhaps most critically, the conventional requirement to distill off the solvent for product isolation exposes the thermally sensitive aromatized intermediate to high temperatures, causing partial decomposition and significantly lowering the final yield. These factors collectively result in a process that is not only chemically inefficient but also operationally burdensome due to complex purification requirements.
The Novel Approach
The methodology disclosed in CN102584692A fundamentally reengineers the reaction environment by introducing a biphasic system comprising a water-insoluble organic solvent, water, and an acid catalyst. This strategic shift creates a unique interfacial reaction zone where the aromatization of the bicyclic intermediate occurs under moderated conditions. As the aromatized product (Formula II) forms, its low solubility in the biphasic mixture promotes immediate precipitation or phase separation, effectively pulling the reaction equilibrium towards completion according to Le Chatelier's principle. Crucially, this approach eliminates the need for high-temperature distillation to remove the organic solvent; instead, the immiscible layers can be separated mechanically, preserving the integrity of the product. This results in a dramatic improvement in conversion rates and simplifies the downstream processing workflow, offering a clear path to higher purity and reduced operational costs.
Mechanistic Insights into Biphasic Acid-Catalyzed Aromatization
The core of this synthetic route lies in the precise control of the aromatization transformation from the Diels-Alder adduct (Formula I) to the pyridine derivative (Formula II). The reaction initiates with the contact of Formula I, where R1 is a C1-C4 alkyl group, with the biphasic solvent system. The acid catalyst, which can range from organic acids like acetic acid to mineral acids like hydrochloric acid, facilitates the elimination of the alkoxy group and the subsequent rearomatization of the ring system. The use of water-insoluble solvents such as petroleum ether, toluene, or 1,2-dichloroethane ensures that the organic phase acts as a reservoir for the reactants while the aqueous phase manages the proton transfer. This segregation prevents the localized high acid concentrations that typically lead to polymerization or degradation in single-phase systems. The reaction is typically conducted in two stages: an initial phase at 10-40°C to gently initiate the transformation, followed by a heating phase at 50-80°C to drive the reaction to completion, ensuring that the conversion ratio is maximized before proceeding to hydrolysis.
Following the successful formation of the aromatized intermediate, the process transitions to a hydrolysis step to yield the final Pyridoxine Hydrochloride (Formula III). This step involves adjusting the pH of the mixture to between 0 and 2 using concentrated hydrochloric acid and heating the aqueous layer to 60-90°C. Under these conditions, the remaining protecting groups are cleaved, and the molecule assumes its final vitamin structure. The mechanistic elegance of this route is evident in the impurity profile; by avoiding the harsh thermal stress of solvent distillation prior to hydrolysis, the formation of tar-like by-products is minimized. The resulting crude product exhibits a much cleaner profile, which simplifies the final crystallization step. This level of control over the reaction trajectory is essential for meeting the stringent purity specifications required for pharmaceutical-grade Vitamin B6, ensuring that the final API intermediate is free from genotoxic impurities often associated with aggressive acid treatments.
How to Synthesize Pyridoxine Hydrochloride Efficiently
The synthesis protocol outlined in the patent offers a reproducible framework for manufacturing high-quality Vitamin B6. The process begins with the preparation of the bicyclic intermediate via a Diels-Alder reaction between 4-methyl-5-alkoxyoxazole and a dihydro-dioxepin derivative. Once this precursor is obtained, it is dissolved in a water-insoluble solvent such as petroleum ether or toluene. Water and a catalytic amount of acid are added, and the mixture is stirred at controlled temperatures to effect aromatization. After the reaction is confirmed complete via HPLC, the layers are allowed to separate. The organic layer is recovered for reuse, while the aqueous layer containing the product is treated with additional hydrochloric acid and heated to induce hydrolysis. The final product is isolated through evaporative crystallization using ethanol, yielding Pyridoxine Hydrochloride that meets pharmacopoeial standards. Detailed standardized synthesis steps follow below.
- Conduct aromatization by contacting the bicyclic intermediate (Formula I) with a water-insoluble solvent, water, and an acid catalyst at 10-80°C to form the aromatized product (Formula II).
- Separate the phases and add hydrochloric acid to the aqueous layer to induce hydrolysis at 60-90°C, converting Formula II into Pyridoxine Hydrochloride (Formula III).
- Isolate the final product through crystallization using ethanol, avoiding high-temperature distillation of the reaction solvent to prevent product degradation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this biphasic synthesis route offers tangible strategic benefits beyond mere chemical yield. The primary advantage lies in the drastic simplification of the unit operations involved. By replacing energy-intensive distillation steps with simple phase separation, the process reduces the thermal load on the equipment and minimizes the risk of batch failure due to overheating. This operational robustness translates directly into enhanced supply chain reliability, as the process is less susceptible to the variability that often plagues complex multi-step syntheses. Furthermore, the ability to recover and reuse the water-insoluble organic solvent without degradation means that raw material consumption is optimized, contributing to a more sustainable and cost-effective manufacturing model.
- Cost Reduction in Manufacturing: The elimination of solvent distillation prior to product isolation represents a significant reduction in energy consumption and processing time. In traditional methods, the need to strip ethanol or other solvents under vacuum often requires specialized equipment and extended cycle times. By utilizing a biphasic system where the solvent is immiscible and easily separated, the process bypasses these costly steps entirely. Additionally, the higher yield (>87 percent) means that less raw material is required to produce the same amount of finished goods, effectively lowering the cost of goods sold (COGS) without compromising on quality. The reduction in by-product formation also lowers the cost associated with waste treatment and disposal.
- Enhanced Supply Chain Reliability: The use of commodity chemicals such as petroleum ether, toluene, and hydrochloric acid ensures that the supply chain is not dependent on exotic or scarce reagents. This availability mitigates the risk of production stoppages due to raw material shortages. Moreover, the robustness of the biphasic reaction conditions allows for greater flexibility in scaling operations. Whether producing in 100 kg batches or multi-ton campaigns, the phase separation mechanics remain consistent, ensuring that lead times for high-purity pharmaceutical intermediates can be reliably met even during periods of high market demand.
- Scalability and Environmental Compliance: From an environmental perspective, this method aligns well with green chemistry principles by reducing solvent waste and energy usage. The closed-loop potential for the organic solvent reduces the volume of volatile organic compounds (VOCs) released into the atmosphere. For facilities operating under strict environmental regulations, this process offers a compliant pathway to scale up production. The simplified workup also reduces the aqueous waste load, as there is less need for extensive washing to remove residual solvents or degradation products, thereby streamlining the effluent treatment process and reducing the overall environmental footprint of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and process descriptions found in CN102584692A, providing a factual basis for evaluating the technology's fit within your existing manufacturing infrastructure. Understanding these nuances is critical for making informed decisions about process adoption and supplier qualification.
Q: How does the biphasic system improve yield compared to traditional ethanol-based methods?
A: Traditional homogeneous systems using ethanol and strong acid often degrade the sensitive intermediate before aromatization completes. The biphasic system moderates acidity at the interface and allows the product to precipitate, driving the equilibrium forward without thermal degradation during solvent removal.
Q: What solvents are compatible with this aromatization process?
A: The process utilizes water-insoluble organic solvents such as C6-C10 alkanes (e.g., n-hexane, petroleum ether), toluene, or chlorinated solvents like 1,2-dichloroethane, which facilitate easy phase separation and recycling.
Q: Can this method be scaled for industrial production of Vitamin B6?
A: Yes, the method is designed for scalability. It eliminates complex distillation steps for solvent recovery, uses readily available raw materials, and achieves yields exceeding 87%, making it highly suitable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyridoxine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and scalable infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields demonstrated in patent literature can be replicated consistently on an industrial scale. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Pyridoxine Hydrochloride meets the exacting standards required by the global pharmaceutical and nutraceutical industries. Our commitment to quality assurance ensures that your supply of this critical vitamin intermediate remains uninterrupted and compliant with international regulations.
We invite you to engage with our technical procurement team to discuss how this advanced biphasic synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic benefits specific to your operation. We encourage you to reach out for specific COA data and route feasibility assessments to validate the performance of this method against your current benchmarks. Let us collaborate to optimize your Vitamin B6 sourcing strategy with a solution that balances technical excellence with commercial viability.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
