Scalable Synthesis of Carbazole-Benzimidazole Hybrids for Advanced Pharmaceutical Applications
Scalable Synthesis of Carbazole-Benzimidazole Hybrids for Advanced Pharmaceutical Applications
The development of novel heterocyclic compounds with extended conjugated systems remains a cornerstone of modern medicinal chemistry and materials science. Patent CN111303127A introduces a significant advancement in the preparation of (E)2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole derivatives, a class of molecules exhibiting potent antibacterial properties and promising optoelectronic characteristics. As a leading reliable pharmaceutical intermediates supplier, we recognize that the integration of carbazole and benzimidazole moieties creates a large conjugated system with superior thermal and optical stability. This technical insight report analyzes the proprietary synthesis route detailed in the patent, highlighting its potential for cost reduction in fine chemical manufacturing and its suitability for commercial scale-up of complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for constructing benzimidazole-fused carbazole architectures often suffer from significant operational inefficiencies that hinder industrial adoption. Conventional methodologies frequently rely on multi-step sequences involving harsh reaction conditions, such as strong acidic environments at elevated temperatures for prolonged periods, which can lead to the degradation of sensitive functional groups on the carbazole backbone. Furthermore, standard amide coupling strategies to link the carboxylic acid precursor with o-phenylenediamine typically necessitate expensive coupling reagents like EDC or HOBt, drastically inflating the raw material costs. These legacy processes often generate substantial amounts of organic waste and require tedious purification steps, such as column chromatography, to remove urea byproducts and unreacted starting materials, thereby reducing the overall atom economy and throughput capacity of the manufacturing line.
The Novel Approach
The innovative methodology disclosed in the patent circumvents these bottlenecks by employing a direct cyclodehydration strategy using phosphorus oxychloride (POCl3) as both a solvent and a catalyst. This approach streamlines the synthesis into a single pot operation where N-alkyl-3-carbazole acrylic acid reacts directly with o-phenylenediamine. 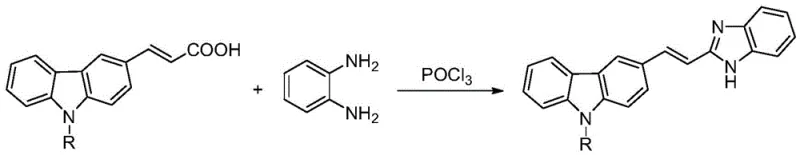 By utilizing POCl3, the reaction activates the carboxylic acid group in situ, facilitating rapid nucleophilic attack by the diamine and subsequent ring closure without the need for pre-activation or protecting groups. The process operates effectively within a moderate temperature range of 80°C to 105°C, significantly shortening the reaction time compared to traditional thermal cyclizations. This simplification not only enhances the reaction efficiency but also drastically simplifies the post-processing workflow, making it an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates.
By utilizing POCl3, the reaction activates the carboxylic acid group in situ, facilitating rapid nucleophilic attack by the diamine and subsequent ring closure without the need for pre-activation or protecting groups. The process operates effectively within a moderate temperature range of 80°C to 105°C, significantly shortening the reaction time compared to traditional thermal cyclizations. This simplification not only enhances the reaction efficiency but also drastically simplifies the post-processing workflow, making it an ideal candidate for reducing lead time for high-purity pharmaceutical intermediates.
Mechanistic Insights into POCl3-Mediated Cyclodehydration
The core of this synthetic breakthrough lies in the dual role of phosphorus oxychloride as a dehydrating agent and a reaction medium. Mechanistically, the oxygen atom of the carboxylic acid group on the carbazole acrylic acid precursor coordinates with the phosphorus center of POCl3, forming a highly reactive acyl phosphate intermediate. This activation lowers the energy barrier for the nucleophilic attack by one of the amino groups of o-phenylenediamine. Following the initial amide bond formation, the second amino group attacks the vinyl position or facilitates intramolecular cyclization, driven by the strong dehydrating capability of the phosphorus species which removes water molecules generated during the ring closure. This cascade results in the formation of the stable benzimidazole ring fused to the vinyl-carbazole system. The use of concentrated hydrochloric acid as an alternative catalyst in some embodiments further demonstrates the robustness of the acid-catalyzed cyclization mechanism, although POCl3 generally provides superior yields due to its water-scavenging properties.
Impurity control is meticulously managed through a physical-chemical workup sequence designed to maximize purity without chromatography. Post-reaction, the mixture is treated with activated carbon at elevated temperatures (80°C-105°C) to adsorb colored impurities and polymeric tars that often form during acid-catalyzed condensations. The hot filtration step ensures that the desired product remains in solution while insoluble particulates are removed. Subsequent neutralization of the acidic filtrate with ammonia water in an ice bath induces rapid precipitation of the target molecule. This pH-swing crystallization technique leverages the basicity of the benzimidazole nitrogen, ensuring that the final solid is free from acidic residues and unreacted diamines, thus meeting stringent purity specifications required for downstream biological applications.
How to Synthesize (E)2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole Efficiently
The synthesis protocol described offers a robust framework for producing these valuable intermediates with high consistency. The process begins with the preparation of the key precursor, N-alkyl-3-carbazole acrylic acid, which can itself be synthesized via a green chemistry approach using deep eutectic solvents (DES) like choline chloride-urea. 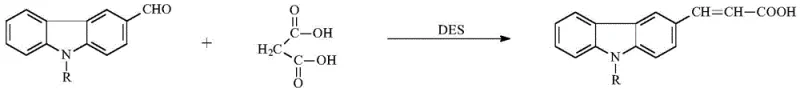 This upstream efficiency contributes to the overall sustainability of the supply chain. For the final cyclization step, precise stoichiometric control is maintained, typically using a slight excess of the acrylic acid derivative relative to o-phenylenediamine to drive the reaction to completion. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles for various alkyl chain lengths, are outlined in the guide below to ensure reproducible results in a GMP-compliant environment.
This upstream efficiency contributes to the overall sustainability of the supply chain. For the final cyclization step, precise stoichiometric control is maintained, typically using a slight excess of the acrylic acid derivative relative to o-phenylenediamine to drive the reaction to completion. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles for various alkyl chain lengths, are outlined in the guide below to ensure reproducible results in a GMP-compliant environment.
- Charge N-alkyl-3-carbazole acrylic acid and o-phenylenediamine into a dry reactor with phosphorus oxychloride.
- Heat the mixture to 80°C-105°C and monitor reaction progress via TLC until completion.
- Cool the reaction, treat with activated carbon, filter hot, and precipitate the product by adjusting pH to alkaline with ammonia water.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented technology offers transformative advantages that directly impact the bottom line and operational reliability. The elimination of expensive coupling reagents and the substitution of volatile organic solvents with POCl3 (which can be recovered) or DES in the precursor step represents a significant shift towards cost-effective manufacturing. The simplicity of the workup—relying on filtration and precipitation rather than energy-intensive distillation or chromatography—reduces utility consumption and labor hours per batch. This operational simplicity translates into substantial cost savings and enhances the economic viability of producing these complex molecules at a commercial scale.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the replacement of precious metal catalysts or expensive peptide coupling agents with commodity chemicals like phosphorus oxychloride and o-phenylenediamine. By avoiding the use of transition metals, the downstream burden of heavy metal removal testing and validation is completely eliminated, which is a major cost center in API intermediate production. Furthermore, the high yields reported (exceeding 90% in optimized examples) minimize raw material waste, ensuring that the cost per kilogram of the final active intermediate is significantly lower than that achieved through conventional multi-step syntheses.
- Enhanced Supply Chain Reliability: The raw materials required for this synthesis, including substituted carbazoles, malonic acid, and phenylenediamines, are widely available commodity chemicals with stable global supply chains. This abundance mitigates the risk of supply disruptions that often plague processes relying on specialized or custom-synthesized reagents. The robustness of the reaction conditions, which tolerate a wide range of alkyl substituents from methyl to hexadecyl chains, allows for flexible production scheduling. Manufacturers can easily switch between different derivatives based on market demand without requiring extensive re-validation of the core process parameters, thereby ensuring consistent delivery timelines.
- Scalability and Environmental Compliance: The process is inherently scalable due to its homogeneous nature and the absence of difficult-to-handle intermediates. The use of activated carbon for decolorization is a standard unit operation easily implemented in large-scale reactors. Additionally, the potential to utilize deep eutectic solvents in the precursor synthesis aligns with modern green chemistry principles, reducing the environmental footprint of the manufacturing process. The straightforward isolation of the product via precipitation minimizes solvent usage in the workup phase, simplifying waste treatment and ensuring compliance with increasingly stringent environmental regulations regarding VOC emissions and hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and quality attributes of this synthesis route. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this intermediate into their drug discovery pipelines. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a transparent view of the technology's capabilities and limitations.
Q: What is the typical yield for this cyclization process?
A: According to patent CN111303127A, the process achieves high yields ranging from 66.1% to 93.1%, depending on the specific alkyl chain length and reaction temperature optimization.
Q: Does this method require column chromatography for purification?
A: No, the method utilizes a robust precipitation and filtration workup. The crude product is purified by hot filtration with activated carbon followed by pH-adjusted precipitation, eliminating the need for costly chromatographic separation.
Q: Can this synthesis be adapted for long-chain alkyl derivatives?
A: Yes, the protocol is highly versatile and has been successfully demonstrated for alkyl chains ranging from methyl (-CH3) to hexadecyl (-C16H33-n), maintaining structural integrity and reasonable yields across the series.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E)2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this advanced chemistry to your supply chain. Our state-of-the-art facilities are equipped to handle the specific safety requirements of phosphorus oxychloride reactions, ensuring both operator safety and product integrity. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of carbazole-benzimidazole derivative meets the exacting standards required for pharmaceutical and agrochemical applications. Our commitment to quality assurance ensures that the impurity profiles are tightly controlled, facilitating smoother regulatory filings for your final products.
We invite you to collaborate with us to optimize this synthesis for your specific needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can accelerate your project timelines and reduce your overall cost of goods sold.
