Advanced Semisynthesis of Staurosporine Analogs for Oncology Drug Development
Introduction to Next-Generation Staurosporine Analogs
The landscape of oncology drug development is constantly evolving, driven by the need for more selective kinase inhibitors with reduced toxicity profiles. Patent CN102924479A introduces a pivotal breakthrough in the semisynthetic modification of staurosporine, a potent but non-selective protein kinase C (PKC) inhibitor. This intellectual property outlines a robust methodology for transforming fermented staurosporine products into novel derivatives that retain antitumor activity while potentially offering improved therapeutic indices. The core innovation lies in a controlled chemical modification of the indolo[2,3-a]carbazole scaffold, specifically targeting the sugar moiety and adjacent nitrogen functionalities to create new chemical entities.
For pharmaceutical developers, accessing these complex scaffolds reliably is critical. The disclosed method leverages fermentation-derived starting materials, bridging the gap between natural product isolation and fully synthetic routes. By utilizing a combination of tungsten-catalyzed oxidation and titanium-mediated reduction, the process achieves high purity and yield without the need for exotic catalysts. This approach not only validates the chemical feasibility of these analogs but also establishes a foundation for scalable manufacturing of high-value API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the modification of staurosporine has been fraught with challenges related to regioselectivity and functional group tolerance. Traditional total synthesis routes are often prohibitively long and expensive, involving dozens of steps with low overall yields, making them unsuitable for commercial supply chains. Furthermore, direct chemical modification of the native fermentation product often suffers from poor control over reaction sites, leading to complex mixtures of byproducts that are difficult to separate. Conventional oxidation methods frequently employ harsh reagents that can degrade the sensitive carbazole core or cause unwanted cleavage of the glycosidic bond, resulting in significant material loss and increased waste generation.
The Novel Approach
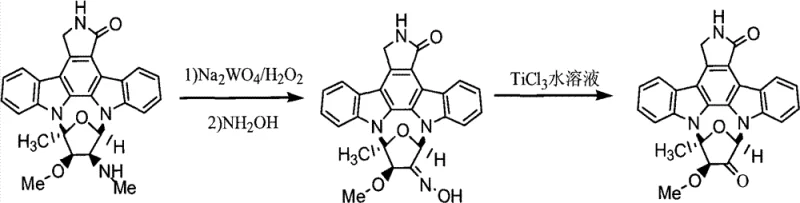
The methodology presented in CN102924479A offers a streamlined alternative that circumvents these historical bottlenecks. By employing a specific sequence of sodium tungstate dihydrate and hydrogen peroxide followed by hydroxylamine treatment, the process achieves selective functionalization under mild conditions. This is followed by a crucial reduction step using aqueous titanium trichloride, which operates effectively at room temperature. This two-stage strategy minimizes side reactions and preserves the integrity of the complex molecular architecture. The result is a process that delivers Derivative 1 with yields exceeding 75% and the final Derivative 2 with yields surpassing 91%, representing a substantial improvement in material efficiency compared to legacy methods.
Mechanistic Insights into Tungsten-Catalyzed Oxidation and TiCl3 Reduction
The chemical elegance of this synthesis lies in its dual-catalyst system. The initial step utilizes sodium tungstate as a catalyst for hydrogen peroxide oxidation. In this mechanism, the tungsten species likely forms a peroxo-complex that acts as an electrophilic oxidant, selectively activating specific C-H or N-H bonds on the staurosporine scaffold. Subsequent treatment with hydroxylamine hydrochloride facilitates the formation of an oxime or hydroxylamine intermediate (Derivative 1). This transformation is critical as it installs a handle for further diversification while maintaining the stereochemical integrity of the molecule. The use of pyridine as a base ensures the reaction proceeds smoothly without acid-catalyzed degradation of the starting material.
The second phase involves the use of titanium(III) chloride, a single-electron transfer reagent known for its ability to reduce N-O bonds and facilitate deoxygenation. In the context of this synthesis, TiCl3 likely reduces the newly formed hydroxylamine or oxime functionality, potentially triggering a cascade of intramolecular rearrangements or simply restoring the amine functionality with high fidelity. The reaction is conducted in DMF, a polar aprotic solvent that stabilizes the charged intermediates and ensures solubility of the polar titanium species. This mechanistic pathway allows for precise control over the oxidation state of the nitrogen atoms, which is essential for tuning the biological activity of the final kinase inhibitor.
How to Synthesize Staurosporine Derivatives Efficiently
Executing this semisynthetic route requires careful attention to solvent quality and stoichiometry to maximize the reported yields. The process begins with the dissolution of the fermented raw material in a methanol and dichloromethane mixture, followed by the controlled addition of the oxidant system. After the formation of the intermediate, the subsequent reduction step must be performed under an inert nitrogen atmosphere to prevent the re-oxidation of the titanium species by atmospheric oxygen. Detailed operational parameters, including specific molar ratios and workup procedures, are essential for reproducing the high purity levels described in the patent literature.
- Oxidation and Oxime Formation: Dissolve fermented staurosporine in methanol/dichloromethane, treat with sodium tungstate and hydrogen peroxide, followed by hydroxylamine hydrochloride to yield Derivative 1.
- Titanium-Mediated Reduction: Dissolve Derivative 1 in DMF and treat with aqueous TiCl3 under nitrogen protection to effect reduction and cyclization modifications.
- Purification: Perform solvent extraction, silica gel column chromatography, and recrystallization to obtain high-purity Staurosporine Analog Derivative 2.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers compelling advantages rooted in the availability and cost of raw materials. The reliance on fermentation products as the starting point leverages established biotechnological supply chains, ensuring a consistent source of the complex carbazole core without the need for expensive de novo synthesis. Furthermore, the reagents employed—sodium tungstate, hydrogen peroxide, and titanium trichloride—are commodity chemicals available in bulk quantities at competitive prices. This contrasts sharply with processes requiring precious metal catalysts like palladium or rhodium, which introduce significant cost volatility and supply risk.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of ambient temperature reactions significantly lower the operational expenditure (OPEX) associated with this synthesis. By avoiding cryogenic cooling or high-pressure hydrogenation equipment, the capital expenditure (CAPEX) for setting up production lines is also minimized. The high yields reported in the patent directly translate to lower cost of goods sold (COGS), as less raw material is wasted in purification steps, making the final API intermediate more economically viable for generic drug manufacturers.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply chain. Since the process does not depend on specialized reagents with long lead times, procurement managers can secure materials from multiple global suppliers, reducing the risk of single-source dependency. The stability of the intermediates allows for flexible batch scheduling, enabling manufacturers to respond quickly to fluctuations in market demand for oncology therapeutics without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: The use of standard organic solvents like methanol, dichloromethane, and ethyl acetate simplifies solvent recovery and recycling processes, aligning with modern green chemistry initiatives. The aqueous workup steps facilitate the removal of inorganic salts and metal residues, ensuring the final product meets stringent regulatory limits for heavy metals. This environmental compatibility reduces the burden on waste treatment facilities and streamlines the regulatory approval process for new drug applications relying on this intermediate.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into their R&D pipelines. The following questions address common inquiries regarding the scalability, purity, and specific chemical transformations involved in producing these staurosporine analogs. These insights are derived directly from the experimental data and claims presented in the underlying patent documentation.
Q: What is the overall yield of the semisynthetic staurosporine derivative process?
A: The process described in patent CN102924479A demonstrates high efficiency, with the first intermediate (Derivative 1) achieving yields around 75.3% and the final product (Derivative 2) reaching yields up to 91.5% after purification.
Q: Why is TiCl3 used in the second step of the synthesis?
A: Titanium(III) chloride acts as a mild yet effective reducing agent capable of selectively reducing N-O bonds or facilitating specific deoxygenation steps without damaging the sensitive indolo[2,3-a]carbazole core structure.
Q: Is this synthesis scalable for commercial API production?
A: Yes, the method utilizes commercially available reagents like sodium tungstate and operates at ambient temperatures, avoiding cryogenic conditions or high-pressure equipment, which significantly enhances its feasibility for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Staurosporine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the race to develop next-generation cancer therapies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of staurosporine derivative meets the exacting standards required for clinical and commercial pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for these critical oncology intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing expertise can accelerate your drug development timeline while reducing overall project costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
