Advanced Synthesis Of Thiouracil-Containing Triazolopyrimidines For Antibacterial Drug Development
Introduction To Novel Antibacterial Intermediates
The escalating crisis of bacterial resistance, driven by the overuse of traditional antibiotics, necessitates the urgent development of novel therapeutic agents with unique mechanisms of action. Patent CN111518104A discloses a groundbreaking class of thiouracil-containing 1,2,4-triazolo[1,5-a]pyrimidine compounds designed to address this global health challenge. These molecules represent a strategic fusion of two potent pharmacophores: the thiouracil moiety, known for inhibiting the bacterial SecA protein, and the rigid 1,2,4-triazolo[1,5-a]pyrimidine scaffold, which enhances binding affinity through hydrogen bonding interactions. This dual-action design offers a promising pathway for creating next-generation antibacterial drugs capable of combating superbugs that are insensitive to current treatments.
![General chemical structure of thiouracil-containing 1,2,4-triazolo[1,5-a]pyrimidine compounds showing variable substituents R1 and R2](/insights/img/thiouracil-triazolopyrimidine-synthesis-pharma-supplier-20260309044613-01.png)
From a process chemistry perspective, the patent outlines a robust and scalable synthetic methodology that avoids the pitfalls of complex multi-step sequences often associated with fused heterocyclic systems. The described route utilizes readily available starting materials such as 3-amino-1,2,4-triazole and aromatic aldehydes, ensuring a stable supply chain for raw materials. By employing a convergent synthesis strategy, the process allows for the parallel preparation of key intermediates, significantly optimizing production timelines. For pharmaceutical companies seeking a reliable pharmaceutical intermediate supplier, this technology offers a viable platform for developing high-purity active ingredients with strong biological profiles against pathogens like Staphylococcus aureus and Bacillus subtilis.
The Limitations Of Conventional Methods Vs. The Novel Approach
The Limitations Of Conventional Methods
Traditional approaches to synthesizing complex nitrogen-rich heterocycles often suffer from significant drawbacks that hinder commercial viability and environmental compliance. Many legacy routes rely heavily on the use of toxic heavy metal catalysts or harsh reagents that generate substantial amounts of hazardous waste, complicating disposal and increasing operational costs. Furthermore, conventional linear syntheses frequently require multiple protection and deprotection steps to manage the reactivity of various functional groups, leading to lower overall yields and extended production cycles. The purification of intermediates in these older methods often demands energy-intensive chromatographic separations, which are difficult to scale up for industrial manufacturing. These inefficiencies result in higher cost of goods sold (COGS) and longer lead times, posing challenges for procurement managers aiming for cost reduction in API manufacturing.
The Novel Approach
In contrast, the methodology presented in CN111518104A introduces a streamlined convergent strategy that dramatically simplifies the construction of the target molecular architecture. The process is divided into two distinct branches: the formation of the chloromethyl-triazolopyrimidine core and the synthesis of the thiouracil derivative, which are subsequently coupled. This approach minimizes the number of sequential operations and eliminates the need for sensitive protecting groups. The reaction conditions are relatively mild, utilizing common solvents like glacial acetic acid and acetonitrile, which are easy to recover and recycle. By avoiding transition metals and focusing on simple acid-base workups and precipitation techniques, the novel approach ensures a cleaner impurity profile. This efficiency translates directly into substantial cost savings and enhanced supply chain reliability for partners looking to scale up complex pharmaceutical intermediates.
Mechanistic Insights Into Convergent Heterocyclic Assembly
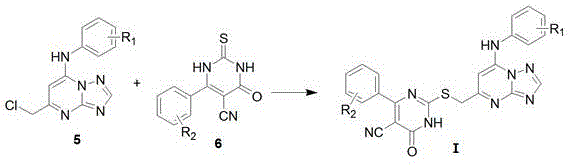
The core of this synthetic innovation lies in the efficient construction of the triazolopyrimidine ring system and its subsequent functionalization. The initial cyclization involves the condensation of 3-amino-1,2,4-triazole with ethyl 4-chloroacetoacetate in glacial acetic acid under reflux. This step effectively builds the fused bicyclic skeleton while installing a reactive chloromethyl handle. Following this, the hydroxyl group is activated via chlorination using phosphorus oxychloride (POCl3), creating a highly electrophilic center susceptible to nucleophilic attack. The introduction of the aniline moiety occurs through a nucleophilic aromatic substitution, where the electron-rich amine displaces the chloride atom. This sequence is critical for establishing the structural diversity of the final library, as various substituted anilines can be employed to tune the electronic properties of the molecule.
The second branch of the synthesis focuses on generating the thiouracil component through a multicomponent condensation reaction. Aromatic aldehydes react with ethyl cyanoacetate and thiourea in the presence of piperidine, a secondary amine catalyst that facilitates the Knoevenagel condensation and subsequent cyclization. The final convergence step involves the nucleophilic substitution of the chloromethyl group on the triazolopyrimidine intermediate by the sulfur atom of the thiouracil derivative. This thioether linkage is formed under basic conditions using potassium carbonate in acetonitrile. The choice of base and solvent is crucial for minimizing side reactions such as hydrolysis of the nitrile group. This mechanistic pathway ensures high regioselectivity and yield, providing a robust method for producing high-purity OLED material precursors or pharmaceutical intermediates with consistent quality.
How To Synthesize Thiouracil-Containing Triazolopyrimidines Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize yield and purity while maintaining safety standards. The process begins with the preparation of the two key intermediates separately, allowing for quality control checks before the final coupling. The chlorination step with POCl3 must be handled with care due to the evolution of HCl gas, requiring appropriate scrubbing systems. Similarly, the final coupling reaction benefits from anhydrous conditions to prevent the degradation of the reactive chloromethyl species. The workup procedures described, involving pH adjustments and selective precipitation, are designed to remove unreacted starting materials and byproducts efficiently without the need for column chromatography on a large scale. Detailed standardized operating procedures for these steps are essential for technology transfer.
- Synthesize the triazolopyrimidine core by refluxing 3-amino-1,2,4-triazole with ethyl 4-chloroacetoacetate in glacial acetic acid, followed by chlorination with POCl3 and amination with substituted aniline.
- Prepare the thiouracil intermediate via condensation of substituted benzaldehyde, ethyl cyanoacetate, and thiourea catalyzed by piperidine in ethanol, followed by acidification to precipitate the product.
- Couple the two intermediates in acetonitrile using potassium carbonate as a base under reflux conditions to form the final thioether-linked target compound.
Commercial Advantages For Procurement And Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical novelty. The reliance on commodity chemicals such as acetic acid, ethanol, and acetonitrile means that raw material sourcing is straightforward and less susceptible to market volatility compared to processes requiring specialized reagents. The elimination of precious metal catalysts removes a significant cost driver and simplifies the regulatory burden associated with residual metal limits in final drug substances. Furthermore, the solid-state isolation of intermediates via precipitation facilitates easier handling, storage, and transportation, reducing the risks associated with liquid intermediates. These factors collectively contribute to a more resilient and cost-effective supply chain for the production of antibacterial agents.
- Cost Reduction In Manufacturing: The process achieves significant economic advantages by utilizing inexpensive, bulk-available starting materials and avoiding costly transition metal catalysts. The simplified purification strategy, which relies on crystallization and filtration rather than chromatography, drastically reduces solvent consumption and processing time. This lean manufacturing approach lowers the overall cost of goods, making the final active pharmaceutical ingredient more competitive in the marketplace. Additionally, the high atom economy of the condensation reactions minimizes waste generation, further reducing disposal costs and enhancing the sustainability profile of the production facility.
- Enhanced Supply Chain Reliability: By decoupling the synthesis into two parallel branches, the manufacturing timeline is compressed, allowing for faster response to market demand. The robustness of the reaction conditions ensures consistent batch-to-batch reproducibility, which is critical for maintaining long-term supply contracts. The use of stable intermediates that can be stockpiled provides a buffer against potential disruptions in the supply of final coupling partners. This flexibility enables manufacturers to optimize inventory levels and reduce lead times for high-purity pharmaceutical intermediates, ensuring uninterrupted production schedules for downstream API synthesis.
- Scalability And Environmental Compliance: The synthetic route is inherently scalable, utilizing unit operations such as reflux, distillation, and filtration that are standard in industrial chemical plants. The absence of hazardous reagents like hydrazine or azides enhances operational safety and simplifies regulatory compliance. Waste streams are primarily composed of aqueous salts and recoverable organic solvents, which can be treated using conventional wastewater management systems. This alignment with green chemistry principles not only meets stringent environmental regulations but also improves the corporate social responsibility standing of the manufacturing organization, appealing to eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel compounds. Understanding the nuances of the synthesis and the biological potential of the molecules is essential for stakeholders evaluating this technology for integration into their R&D pipelines. The answers provided are derived directly from the technical specifications and experimental data disclosed in the patent literature, ensuring accuracy and relevance for decision-makers.
Q: What is the primary mechanism of action for these thiouracil-triazolopyrimidine compounds?
A: These compounds function by inhibiting the SecA protein in bacteria, a novel target for antibacterial drugs that helps overcome resistance mechanisms found in traditional antibiotics.
Q: Are precious metal catalysts required for this synthesis?
A: No, the process utilizes organic bases like piperidine and inorganic bases like potassium carbonate, eliminating the need for expensive transition metal catalysts and simplifying purification.
Q: How is the purity of the final intermediate controlled?
A: Purity is managed through specific precipitation steps involving pH adjustment and recrystallization, ensuring high-quality material suitable for downstream API processing without extensive chromatography.
Partnering With NINGBO INNO PHARMCHEM: Your Reliable Thiouracil-Containing Triazolopyrimidine Supplier
As the demand for novel antibacterial agents continues to rise, partnering with an experienced CDMO is crucial for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from pilot plant to full-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific reaction conditions required for heterocyclic synthesis, including corrosion-resistant reactors for acid-mediated steps and advanced filtration systems for solid isolation. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest international standards for pharmaceutical intermediates.
We invite you to collaborate with our technical team to explore the full potential of this thiouracil-triazolopyrimidine platform. By leveraging our process optimization expertise, we can help you achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for your upcoming projects. Together, we can accelerate the development of life-saving antibacterial therapies and secure a sustainable supply of high-quality chemical intermediates for the global healthcare market.
