Scalable Nickel-Catalyzed Asymmetric Hydroamination for High-Purity Chiral Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for constructing chiral amine motifs, which serve as critical scaffolds in countless bioactive molecules. Patent CN110734378B introduces a transformative approach to this challenge by detailing a highly chemoselective and stereoselective method for preparing chiral allylamine compounds. This technology leverages a cost-effective Nickel-based catalytic system, specifically utilizing bis(1,5-cyclooctadiene)nickel [Ni(COD)2] in conjunction with chiral bisphosphine ligands and Brønsted acid additives. Unlike traditional methods that often rely on expensive noble metals or harsh reaction conditions, this innovation operates under remarkably mild parameters, typically at room temperature (25 °C), while delivering exceptional enantiomeric excess (ee) values reaching up to 99%. For R&D directors and process chemists, this represents a significant opportunity to streamline synthetic routes for complex intermediates, ensuring high purity and structural integrity without the burden of extreme thermal or pressure requirements.
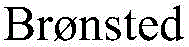
The core of this technological breakthrough lies in its ability to facilitate the direct addition of N-H bonds across unsaturated carbon-carbon double bonds with precise stereocontrol. The reaction system is designed to be atom-economical and aligns with green chemistry principles by minimizing waste and energy consumption. By dissolving 1,3-conjugated dienes and various amines in common organic solvents such as toluene, the process generates valuable chiral allylamine products after simple column chromatography purification. The patent highlights the method's versatility across a broad spectrum of substrates, proving its utility not just for simple model compounds but also for the derivatization of complex drug molecules. This capability positions the technology as a vital tool for the reliable pharmaceutical intermediate supplier looking to diversify their catalog with high-value chiral building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric hydroamination of alkenes and dienes has been dominated by catalysts based on precious metals such as Rhodium, Iridium, or Palladium. While effective, these systems present substantial economic and logistical challenges for large-scale manufacturing. The high cost of noble metal precursors directly impacts the cost reduction in chiral amine manufacturing, making the final API intermediates prohibitively expensive for generic drug production. Furthermore, many conventional protocols require stringent reaction conditions, including elevated temperatures, high pressures, or the use of hazardous reagents to drive conversion, which complicates the commercial scale-up of complex intermediates. Another persistent issue is regioselectivity; traditional methods often struggle to distinguish between multiple nucleophilic sites on amine substrates, leading to mixtures of regioisomers that are difficult and costly to separate. These limitations create bottlenecks in the supply chain, extending lead times and increasing the environmental footprint due to extensive purification steps and solvent usage.
The Novel Approach
The methodology disclosed in CN110734378B offers a compelling alternative by shifting the paradigm to base-metal catalysis. By employing Nickel, an earth-abundant and inexpensive metal, the process inherently lowers the raw material cost baseline. The introduction of a Brønsted acid additive acts as a crucial cocatalyst, enhancing the reactivity of the Nickel center and improving the protonation steps necessary for the hydroamination cycle. This synergistic effect allows the reaction to proceed efficiently at ambient temperature (25 °C), eliminating the need for energy-intensive heating or cooling systems. Moreover, the system demonstrates remarkable chemoselectivity, effectively identifying and reacting at specific nucleophilic sites even in amines containing multiple potential reaction centers. This precision reduces the formation of byproducts and simplifies downstream processing, thereby enhancing the overall efficiency of high-purity OLED material or pharmaceutical intermediate production lines.
Mechanistic Insights into Ni-Catalyzed Asymmetric Hydroamination
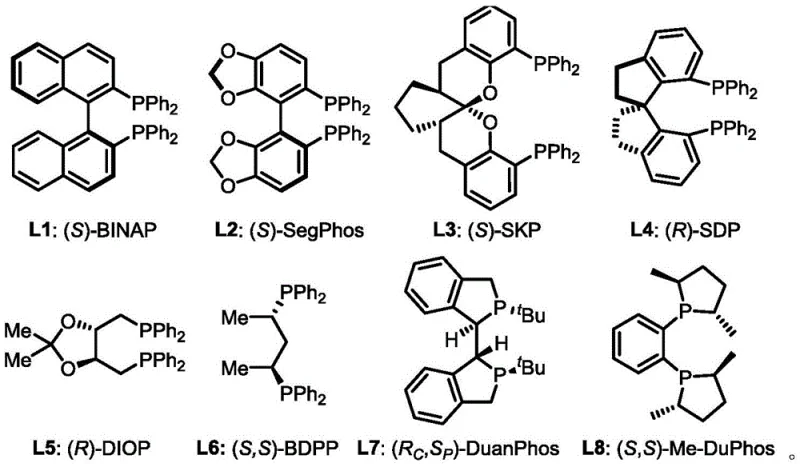
The success of this transformation hinges on the intricate interplay between the Nickel precursor, the chiral ligand environment, and the acid additive. The catalytic cycle likely initiates with the coordination of the chiral bisphosphine ligand to the Ni(0) center, generating a sterically defined active species. Ligands such as (S,S)-Me-DuPhos, (R)-BINAP, or (S)-SegPhos create a chiral pocket that dictates the facial selectivity of the diene coordination. The presence of the acid additive, such as phthalic acid or benzoic acid, is believed to facilitate the protonolysis of the organometallic intermediate, regenerating the active catalyst and releasing the product with high stereochemical fidelity. This mechanism ensures that the newly formed C-N bond is established with the desired configuration, consistently achieving ee values greater than 99% in optimized examples. The robustness of this catalytic system is evidenced by its tolerance to various functional groups, allowing for the synthesis of diverse derivatives without the need for extensive protecting group strategies.
Impurity control is another critical aspect where this mechanism excels. The high regioselectivity prevents the formation of branched isomers or over-hydrogenated side products that often plague transition metal-catalyzed aminations. The mild reaction conditions further mitigate the risk of thermal degradation or polymerization of the sensitive 1,3-diene substrates. For quality assurance teams, this means a cleaner crude reaction profile, which translates to higher recovery rates during purification and a more consistent impurity profile in the final bulk drug substance. The ability to tune the electronic and steric properties of the ligand allows process chemists to optimize the reaction for specific substrate classes, ensuring that the synthesis remains robust even when scaling from milligram to kilogram quantities. This level of control is essential for meeting the stringent purity specifications required by global regulatory bodies.
How to Synthesize Chiral Allylamine Compounds Efficiently
The practical implementation of this synthesis involves a straightforward protocol suitable for both laboratory discovery and pilot plant operations. The process begins with the preparation of the catalyst solution under an inert atmosphere, ensuring the stability of the Ni(0) species. Substrates are then introduced along with the acid promoter, and the mixture is allowed to stir at room temperature, typically completing within 24 hours. This operational simplicity reduces the technical barrier for adoption, allowing manufacturing teams to integrate this chemistry into existing workflows with minimal retooling. For detailed procedural specifics regarding stoichiometry, solvent choices, and workup procedures, please refer to the standardized synthesis guide below.
- Prepare the catalyst solution by mixing Ni(COD)2 and a chiral diphosphine ligand (e.g., (S,S)-Me-DuPhos) in dry organic solvent under inert gas.
- Add the 1,3-conjugated diene substrate, amine nucleophile, and a Brønsted acid additive (e.g., phthalic acid) to the catalyst mixture.
- Stir the reaction at room temperature (25°C) for 24 hours, then concentrate and purify via column chromatography to isolate the chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this Nickel-catalyzed technology offers profound strategic benefits. The shift from precious metals to Nickel represents a direct mitigation of supply risk associated with volatile noble metal markets. By securing a synthesis route that relies on abundant base metals, organizations can stabilize their cost structures and protect against sudden price spikes in raw materials. Furthermore, the mild reaction conditions reduce the energy load on manufacturing facilities, contributing to lower operational expenditures and a smaller carbon footprint. The high selectivity of the process minimizes solvent consumption and waste generation, aligning with increasingly strict environmental regulations and sustainability goals. These factors collectively enhance the resilience of the supply chain, ensuring a continuous and reliable flow of critical intermediates.
- Cost Reduction in Manufacturing: The substitution of expensive noble metal catalysts with Nickel significantly lowers the direct material cost per kilogram of product. Additionally, the ability to reduce catalyst loading to below 1 mol% in scaled reactions further amplifies these savings. The elimination of harsh conditions reduces energy costs and extends the lifespan of reactor equipment, while the high selectivity minimizes the loss of valuable starting materials to side reactions, optimizing the overall mass balance and yield efficiency.
- Enhanced Supply Chain Reliability: Nickel and the associated ligands are commercially available from multiple global suppliers, reducing the risk of single-source dependency. The robustness of the reaction across a wide substrate scope means that a single platform technology can be applied to synthesize a diverse library of intermediates, simplifying inventory management. The mild conditions also allow for safer transport and storage of reagents, reducing logistical complexities and ensuring that production schedules are met without interruption due to safety incidents or equipment failures.
- Scalability and Environmental Compliance: The patent explicitly demonstrates successful gram-scale amplification, indicating a clear path to ton-scale production. The use of common organic solvents like toluene facilitates solvent recovery and recycling, supporting circular economy initiatives. The reduction in hazardous waste generation simplifies effluent treatment processes, ensuring compliance with environmental discharge standards. This scalability ensures that the technology can meet the growing demand for chiral amines in the pharmaceutical and agrochemical sectors without compromising on quality or sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Nickel-catalyzed hydroamination technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is crucial for project managers evaluating the feasibility of integrating this method into their current production pipelines.
Q: What are the advantages of using Nickel over precious metals for chiral amine synthesis?
A: Nickel catalysts like Ni(COD)2 are significantly more abundant and cost-effective than traditional noble metals such as Rhodium or Palladium. This patent demonstrates that Nickel, when paired with specific chiral ligands and acid additives, achieves comparable or superior enantioselectivity (up to >99% ee) under much milder conditions, drastically reducing raw material costs.
Q: Can this hydroamination process be scaled for industrial production?
A: Yes, the patent explicitly validates gram-scale amplification with catalyst loading reduced to below 1 mol%. The reaction proceeds efficiently at room temperature (25°C) without requiring high pressure or cryogenic conditions, which simplifies reactor requirements and enhances safety for large-scale commercial manufacturing.
Q: What is the substrate scope for this asymmetric hydroamination method?
A: The method exhibits exceptional versatility, accommodating a wide range of 1,3-conjugated dienes including aryl, heteroaryl, and alkyl-substituted variants. It is also highly compatible with diverse amine nucleophiles, including primary amines, secondary amines, aromatic amines, and even complex drug molecules like desloratadine derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Allylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the one described in CN110734378B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into industrial reality. Our state-of-the-art facilities are equipped to handle air-sensitive Nickel catalysis with rigorous safety protocols, and our stringent purity specifications guarantee that every batch meets the highest international standards. With our rigorous QC labs and dedicated process development teams, we are committed to delivering high-purity chiral intermediates that accelerate your drug development timelines.
We invite you to collaborate with us to leverage this cost-effective and scalable synthesis route for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this Nickel-catalyzed approach can optimize your budget. Please contact us today to request specific COA data for similar chiral amine structures and to discuss route feasibility assessments for your target compounds. Let us be your partner in driving efficiency and innovation in your supply chain.
