Revolutionizing 2-Cyano-4-Nitroaniline Production: A Technical Deep Dive into Patent CN114213281A
Revolutionizing 2-Cyano-4-Nitroaniline Production: A Technical Deep Dive into Patent CN114213281A
The chemical industry is constantly seeking more efficient pathways for producing critical dye intermediates, and the recent disclosure in patent CN114213281A presents a transformative approach to synthesizing 2-cyano-4-nitroaniline. This compound serves as a vital diazo component for high-performance disperse dyes, yet its traditional manufacturing has long been plagued by excessive acid consumption and purity challenges. The patented methodology introduces a sophisticated organic solvent-mediated nitration followed by a unique anhydrous ammonolysis strategy. By shifting the reaction paradigm from bulk acid systems to controlled organic phases, this technology addresses fundamental bottlenecks in mass transfer and byproduct formation. For R&D leaders and procurement strategists alike, understanding this shift is crucial for evaluating future supply chain resilience and cost structures in the fine chemicals sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 2-cyano-4-nitroaniline has relied heavily on a brute-force nitration process where o-chlorobenzonitrile is dripped into a massive excess of concentrated sulfuric and nitric acids. In these legacy systems, sulfuric acid acts not merely as a catalyst but as the primary solvent to ensure the substrate dissolves, leading to staggering volumes of waste acid that require costly neutralization and disposal. Furthermore, the subsequent ammonolysis step often utilizes aqueous ammonia or chlorobenzene solvents, which introduce significant risks of cyano group hydrolysis, resulting in carboxylic acid impurities that degrade the final dye quality. The isolation of the intermediate typically involves water precipitation, filtration, and drying, creating a discontinuous process flow that increases operational complexity and energy consumption while exposing the sensitive nitrile group to moisture.
The Novel Approach
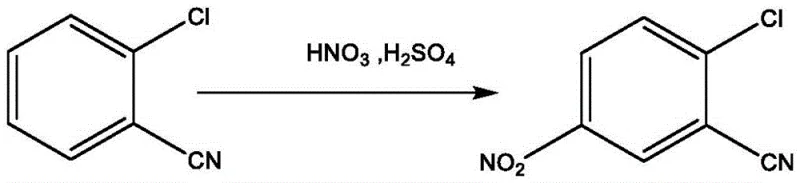
In stark contrast, the methodology outlined in CN114213281A leverages a biphasic system where o-chlorobenzonitrile is dissolved in a recoverable organic solvent such as 1,2-dichloroethane prior to nitration. This strategic modification drastically reduces the requirement for sulfuric acid, as the organic medium handles the solvation burden, allowing the acid to function primarily as a nitrating agent. Following the reaction, the mixture is treated with water to adjust acidity and induce phase separation, yielding an organic layer rich in 2-chloro-5-nitrobenzonitrile that is essentially free of bulk water. This organic phase is then transferred directly to the ammonolysis reactor, bypassing the isolation and drying steps entirely. This seamless integration not only streamlines the workflow but also creates an inherently anhydrous environment for the ammonia substitution, effectively suppressing hydrolysis side reactions.
Mechanistic Insights into Organic Phase Nitration and Anhydrous Ammonolysis
The core innovation lies in the precise control of the reaction microenvironment during the nitration and subsequent substitution phases. By utilizing halogenated organic solvents like 1,2-dichloroethane or 1,3-dichloropropane, the system achieves superior solubility for the aromatic substrate at much lower acid concentrations compared to traditional homogenous acid systems. The nitration proceeds efficiently at mild temperatures between 0°C and 20°C, minimizing thermal degradation. Crucially, the post-nitration workup involves a controlled mixing step where water is added to adjust the acidity value to a specific range of 5% to 25%. This precise adjustment ensures that the nitrated product remains fully dissolved in the denser organic下层 (lower layer) while inorganic salts and spent acids partition into the upper aqueous phase. This physical separation mechanism is far more efficient than the precipitation-filtration cycles of the past, ensuring high recovery of the intermediate without mechanical losses.
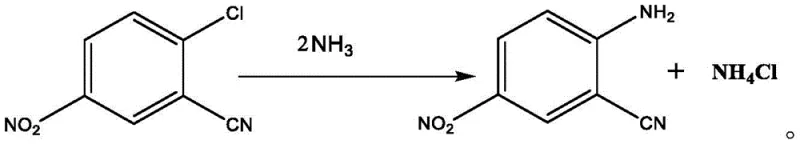
Furthermore, the ammonolysis mechanism benefits profoundly from the absence of water in the reaction vessel. In conventional aqueous ammonia processes, the nucleophilic attack by ammonia on the chloro-substituted ring competes with the hydrolysis of the adjacent cyano group, especially under the high temperature and pressure conditions required for substitution. By introducing liquid ammonia or ammonia gas directly into the dried organic phase containing the intermediate, the patent ensures that the nucleophile concentration is high while water activity is negligible. This kinetic favorability drives the substitution of the chlorine atom with the amino group to form 2-cyano-4-nitroaniline with exceptional selectivity. The result is a product profile with significantly reduced hydrolysis peaks, as evidenced by HPLC purities exceeding 98% in the provided examples, which is critical for downstream dye synthesis where impurity carryover can alter shade and fastness properties.
How to Synthesize 2-Cyano-4-Nitroaniline Efficiently
The implementation of this synthesis route requires careful attention to solvent selection and phase management to maximize the benefits of the patented process. Operators must ensure that the organic solvent chosen has a density greater than water to facilitate the specific layering described in the separation step, with 1,2-dichloroethane being a preferred candidate due to its boiling point and solvation properties. The transition from the nitration kettle to the ammonolysis autoclave must be managed to prevent moisture ingress, preserving the anhydrous advantage of the organic phase. Detailed standard operating procedures regarding temperature ramping during ammonolysis and pressure control are essential to maintain safety and yield consistency.
- Dissolve o-chlorobenzonitrile in an organic solvent like 1,2-dichloroethane and perform nitration with mixed acid at 0-20°C.
- Mix the reaction product with water to adjust acidity, allow phase separation, and isolate the organic layer containing the nitro-intermediate.
- Subject the organic phase directly to ammonolysis with liquid ammonia at 140-160°C to yield high-purity 2-cyano-4-nitroaniline.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible operational efficiencies and risk mitigation strategies. The most immediate impact is the drastic reduction in raw material costs associated with sulfuric acid; by replacing the bulk acid solvent role with a recyclable organic solvent, the process significantly lowers the volume of hazardous waste generated per kilogram of product. This reduction in waste acid volume directly correlates to lower disposal costs and reduced environmental compliance burdens, making the manufacturing footprint cleaner and more sustainable. Additionally, the ability to recover and reuse the organic solvent and unreacted ammonia creates a circular material flow that insulates the production cost from volatile raw material price fluctuations, offering a more predictable cost base for long-term contracting.
- Cost Reduction in Manufacturing: The elimination of extensive filtration, washing, and drying steps for the intermediate represents a substantial decrease in energy consumption and labor hours. By processing the intermediate in solution rather than as a solid cake, the facility saves on utility costs for heating and vacuum drying, while also reducing the wear and tear on solid-handling equipment. The qualitative shift from a batch-solid process to a continuous-liquid flow for the intermediate handling enhances overall plant throughput without requiring capital expansion, effectively lowering the unit cost of production through improved asset utilization.
- Enhanced Supply Chain Reliability: The simplified process flow reduces the number of unit operations required to convert raw materials into the final API intermediate, thereby shortening the overall manufacturing cycle time. Fewer processing steps mean fewer potential points of failure or bottlenecking, leading to more reliable delivery schedules for downstream dye manufacturers. Furthermore, the robustness of the organic solvent system against hydrolysis ensures consistent batch-to-batch quality, reducing the likelihood of off-spec material that could disrupt the customer's production lines and necessitate urgent replacement shipments.
- Scalability and Environmental Compliance: From an environmental perspective, the minimization of waste acid generation aligns perfectly with increasingly stringent global regulations on industrial effluent. The process design inherently supports greener chemistry principles by maximizing atom economy in the nitration step and minimizing solvent loss through efficient recovery loops. This environmental stewardship not only future-proofs the supply chain against regulatory tightening but also enhances the brand value of the final dye products in markets that prioritize sustainably sourced ingredients, providing a competitive edge in eco-conscious sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this method for commercial scale-up.
Q: How does this new method prevent cyano group hydrolysis?
A: The process isolates the nitration intermediate in an organic phase and subjects it directly to anhydrous ammonolysis using liquid ammonia, eliminating the water exposure that typically causes hydrolysis in traditional aqueous ammonia methods.
Q: What are the primary cost-saving mechanisms in this patent?
A: Significant cost reductions are achieved by drastically lowering sulfuric acid consumption through the use of organic solvents as the reaction medium, and by enabling the recovery and reuse of both the organic solvent and unreacted ammonia.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method simplifies the workflow by omitting filtration and drying steps for the intermediate, making it highly scalable and operationally efficient for commercial manufacturing of dye intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyano-4-Nitroaniline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the value chain of specialty dyes and pigments. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes like the one described in CN114213281A can be successfully translated into robust industrial reality. Our facilities are equipped with rigorous QC labs and stringent purity specifications that guarantee every batch of 2-cyano-4-nitroaniline meets the exacting standards required for high-performance textile applications. We understand that consistency is key, and our engineering teams are adept at optimizing reaction parameters to maintain the delicate balance between yield and purity.
We invite you to collaborate with us to leverage these technological advancements for your specific supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this optimized route can improve your bottom line. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to process innovation can secure your supply of high-quality dye intermediates for the future.
