Advanced Fluorine-Substituted Benzoxazole Liquid Crystals for High-Performance Display Manufacturing
Advanced Fluorine-Substituted Benzoxazole Liquid Crystals for High-Performance Display Manufacturing
The rapid evolution of the display industry demands materials that offer superior optical performance, specifically regarding response speed and phase modulation capabilities. Patent CN110437846B introduces a groundbreaking class of fluorine-substituted benzoxazole liquid crystal compounds containing acetylene bonds, designed to address the critical need for large birefringence (Δn) in modern optoelectronic devices. These compounds are engineered with a rigid heterocyclic skeleton that enhances molecular ordering while maintaining low viscosity, a crucial factor for reducing the driving voltage and power consumption in high-definition screens. The structural versatility allows for variations in the alkyl chain length (C5~C12) and substituents (H, CH3, NO2), providing a tunable platform for specific device requirements. By integrating these advanced materials into the supply chain, manufacturers can achieve significant improvements in the performance of tablet PCs, mobile phones, and adaptive optics systems. 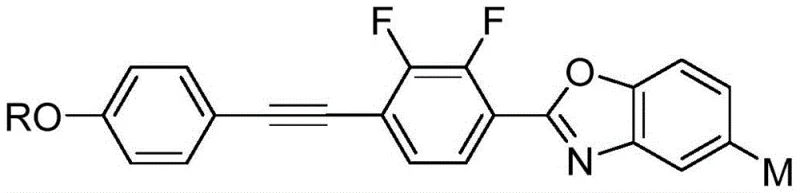
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional liquid crystal materials often rely on biphenyl or phenyl-cyclohexyl cores which, while stable, frequently suffer from insufficient optical anisotropy for next-generation fast-response applications. Conventional synthesis routes for high Δn materials often involve complex multi-step processes that result in lower overall yields and higher production costs due to the use of expensive intermediates or harsh reaction conditions. Furthermore, many existing aromatic heterocyclic liquid crystal compounds lack the necessary combination of wide nematic phase intervals and low viscosity, limiting their practical utility in thin-cell devices where rapid switching is paramount. The reliance on older chemical architectures restricts the ability to fine-tune physical properties without compromising thermal stability, creating a bottleneck for innovation in display technology. Consequently, there is a persistent industry demand for novel molecular designs that can break these performance ceilings without introducing unmanageable manufacturing complexities.
The Novel Approach
The patented methodology overcomes these historical constraints by employing a streamlined synthetic strategy centered around Sonogashira coupling and oxidative cyclization reactions. This approach efficiently constructs the conjugated system necessary for high birefringence by linking fluorinated benzene rings with alkyne units and benzoxazole moieties. The use of 2,3-difluoro-p-bromobenzaldehyde as a key starting material ensures the precise incorporation of fluorine atoms, which are critical for enhancing dielectric anisotropy and reducing rotational viscosity. The process is designed to be robust, utilizing standard catalysts such as cuprous iodide and tetrakis(triphenylphosphine)palladium under nitrogen protection to prevent oxidation of sensitive intermediates. 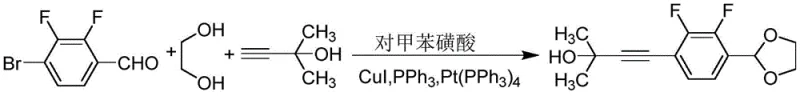
Mechanistic Insights into Sonogashira Coupling and Oxidative Cyclization
The core of this synthesis lies in the palladium-catalyzed cross-coupling reactions that build the molecular backbone with high precision. In the initial stages, the Sonogashira coupling facilitates the formation of the carbon-carbon triple bond between the aryl halide and the terminal alkyne, a reaction that proceeds efficiently at temperatures between 80-90 ℃. The catalytic cycle involves the oxidative addition of the aryl bromide to the palladium center, followed by transmetallation with the copper-acetylide species, and finally reductive elimination to release the coupled product. This mechanism is highly sensitive to the purity of reagents and the exclusion of oxygen, necessitating rigorous process controls to maintain high conversion rates. The subsequent steps involve nucleophilic additions and deprotections that prepare the molecule for the final ring-closing event, ensuring that the functional groups are correctly positioned for cyclization.
The final formation of the benzoxazole ring is achieved through an oxidative cyclization using dichlorodicyanoquinone (DDQ), which acts as a mild yet effective oxidant to aromatize the intermediate Schiff base. This step is crucial for establishing the planar, rigid structure that defines the liquid crystalline behavior of the final compound. Impurity control is managed through careful stoichiometry, such as maintaining a molar ratio of 1:1.1 to 1.5 between the aminophenol and the aldehyde precursor, minimizing the formation of side products. Purification is further enhanced by column chromatography using petroleum ether and ethyl acetate mixtures, ensuring that the final product meets the stringent purity specifications required for electronic grade materials. 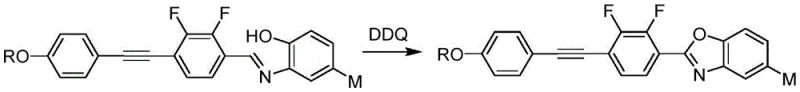
How to Synthesize Fluorine Substituted Benzoxazole Efficiently
The synthesis of these high-performance liquid crystal compounds requires a disciplined approach to reaction conditions and workup procedures to ensure consistent quality. The process begins with the protection of the aldehyde group followed by the critical coupling steps, each requiring specific temperature controls ranging from 50 ℃ to 90 ℃ depending on the stage. Operators must pay close attention to the molar ratios of catalysts, particularly the palladium and copper species, to maximize yield while minimizing metal residues that could affect device performance. The detailed standardized synthesis steps see the guide below, which outlines the precise sequence of reagent addition, reaction times, and purification techniques necessary for successful production. Adhering to these protocols ensures that the resulting material exhibits the desired wide nematic phase interval and high optical anisotropy.
- Perform Sonogashira coupling of 2,3-difluoro-p-bromobenzaldehyde with 2-methyl-3-butyn-2-ol using Pd/PPh3 catalyst.
- Execute deprotection and second coupling with 1-iodo-4-(alkoxy)benzene to extend the conjugated system.
- Complete the synthesis via condensation with aminophenol followed by DDQ-mediated oxidative cyclization to form the benzoxazole ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this patented synthesis route offers substantial advantages by utilizing readily available raw materials such as 2,3-difluoro-p-bromobenzaldehyde and common aminophenols. The simplicity of the reaction steps reduces the overall processing time and energy consumption, leading to a more cost-effective manufacturing profile compared to more convoluted synthetic pathways. The elimination of exotic or hard-to-source reagents minimizes supply chain risks, ensuring a stable and continuous flow of materials for large-scale production runs. Additionally, the high yields reported in the patent examples suggest that waste generation is minimized, aligning with modern environmental compliance standards and reducing disposal costs. These factors collectively contribute to a more resilient supply chain capable of meeting the demanding volumes of the consumer electronics market.
- Cost Reduction in Manufacturing: The streamlined nature of the synthetic route eliminates the need for multiple complex protection and deprotection cycles that typically drive up operational expenses. By using efficient catalysts that operate at moderate temperatures, the process reduces energy overheads associated with heating and cooling large reactors. The high selectivity of the reactions means less raw material is wasted on by-products, directly improving the atom economy and lowering the cost per kilogram of the final active ingredient. Furthermore, the use of standard solvents like trichloromethane and ethanol simplifies solvent recovery and recycling processes, adding another layer of cost efficiency to the overall operation.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals for the starting materials ensures that production is not vulnerable to the bottlenecks often associated with specialized fine chemical intermediates. This accessibility allows for the qualification of multiple suppliers for raw materials, thereby mitigating the risk of single-source dependency. The robustness of the reaction conditions also means that the process can be transferred between different manufacturing sites with minimal re-validation, providing flexibility in case of regional disruptions. Such stability is critical for long-term contracts with major display manufacturers who require guaranteed delivery schedules to maintain their own production lines.
- Scalability and Environmental Compliance: The reaction parameters described in the patent are well-suited for scale-up from laboratory glassware to industrial stainless steel reactors without significant modification. The absence of extremely hazardous reagents or extreme pressure conditions simplifies the safety engineering required for commercial plants. Moreover, the workup procedures involving standard extraction and crystallization techniques are easily adaptable to continuous processing technologies, which can further enhance throughput. The process inherently supports green chemistry principles by maximizing yield and minimizing waste, facilitating easier compliance with increasingly strict environmental regulations in key manufacturing regions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these fluorine-substituted benzoxazole compounds. The answers are derived directly from the experimental data and technical disclosures within the patent documentation to ensure accuracy. Understanding these details is essential for R&D teams evaluating the material for new formulations and procurement officers assessing the feasibility of sourcing. We encourage stakeholders to review these insights to fully appreciate the value proposition of this technology.
Q: What is the primary advantage of the fluorine-substituted benzoxazole structure?
A: The introduction of fluorine atoms and acetylene bonds significantly increases the optical anisotropy (delta n) and reduces viscosity, enabling faster response times in liquid crystal devices compared to conventional phenyl-cyclohexyl structures.
Q: How does the patented method improve yield and purity?
A: The process utilizes specific molar ratios of catalysts like tetrakis(triphenylphosphine)palladium and rigorous purification steps including column chromatography and recrystallization, ensuring high purity suitable for electronic applications.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the reaction conditions operate at moderate temperatures (50-90 ℃) and use commercially available raw materials, making the transition from laboratory scale to commercial tonnage feasible with standard chemical engineering controls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fluorine Substituted Benzoxazole Liquid Crystal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced liquid crystal materials play in the future of display technology and optoelectronics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative patents like CN110437846B can be successfully translated into market-ready products. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the exacting standards required for high-performance electronic applications. Our commitment to quality and consistency makes us an ideal partner for companies seeking to integrate next-generation liquid crystals into their product portfolios.
We invite you to collaborate with us to explore the full potential of these fluorine-substituted benzoxazole derivatives for your specific needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your development timeline and help you secure a competitive advantage in the global market.
