Efficient One-Step Synthesis of 3,4-Dihydro-2H-1,3-Thiazine Derivatives for Pharma
Introduction to Advanced Thiazine Synthesis Technology
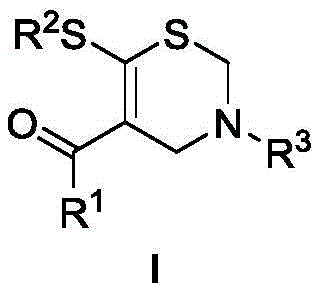
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and environmentally benign synthetic routes. A significant breakthrough in this domain is detailed in patent CN111848547B, which introduces a novel methodology for the preparation of 3,4-dihydro-2H-1,3-thiazine derivatives. These sulfur-containing heterocycles are critical scaffolds in medicinal chemistry, serving as key precursors for biologically active molecules including antihistamines and antipsychotic agents. The traditional reliance on complex multi-step sequences often hampers the rapid development of these vital compounds. This new approach leverages alpha-enol dithioesters and 1,3,5-triazines to construct the thiazine core in a single, high-yielding operation. By utilizing readily available starting materials and avoiding harsh reaction conditions, this technology offers a compelling solution for scaling up the production of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of thiazine and related benzothiazine derivatives has been fraught with synthetic challenges that impede commercial viability. Conventional pathways often rely on multi-component reactions involving isothiocyanates, hydrazines, and alkynes, which necessitate precise stoichiometric control and frequently result in regioselectivity issues. Furthermore, many established protocols depend heavily on the use of strong Lewis acid catalysts, such as Ytterbium triflate (Yb(OTf)3), which not only inflate raw material costs but also introduce significant downstream purification burdens. The requirement for strict inert atmosphere protection (e.g., nitrogen or argon blanketing) adds another layer of operational complexity and equipment cost, particularly when transitioning from laboratory benchtop to kilogram-scale production. These factors collectively contribute to longer lead times and reduced overall process efficiency, creating bottlenecks for supply chains dependent on these specialized heterocyclic building blocks.
The Novel Approach
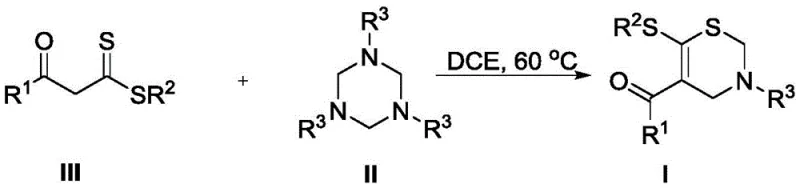
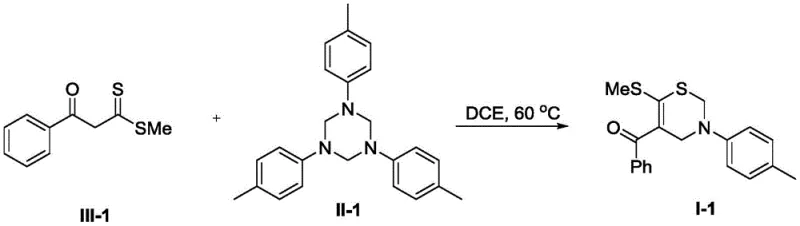
In stark contrast to these legacy methods, the technology disclosed in CN111848547B presents a streamlined, one-pot cyclization strategy that fundamentally simplifies the manufacturing process. By reacting alpha-enol dithioesters with 1,3,5-triazines in dichloroethane (DCE) at a moderate temperature of 60°C, the method achieves the construction of the sulfur-containing six-membered ring in a single step. Crucially, this transformation proceeds without the addition of any external acid, base, or metal catalyst, thereby eliminating the risk of heavy metal contamination in the final product. The operational simplicity is further enhanced by the fact that no inert gas protection is required, allowing the reaction to be run under ambient atmospheric conditions. This robust protocol not only delivers generally high yields across a broad range of substrates but also drastically reduces the environmental footprint and operational overhead associated with traditional thiazine synthesis.
Mechanistic Insights into Metal-Free Cyclization
The efficacy of this novel synthetic route lies in the unique reactivity profile of the alpha-enol dithioester functionality. Mechanistically, the reaction likely initiates through a nucleophilic attack by the nitrogen atoms of the 1,3,5-triazine ring onto the electrophilic centers of the dithioester, facilitated by the thermal energy provided at 60°C. This interaction triggers a cascade of bond rearrangements that ultimately lead to the expulsion of volatile byproducts and the formation of the stable 3,4-dihydro-2H-1,3-thiazine core. The absence of a metal catalyst suggests that the electronic properties of the enol dithioester are sufficiently activated to drive the cyclization forward without external promotion. This metal-free nature is a distinct advantage for pharmaceutical applications, as it inherently avoids the formation of metal-complexed impurities that are notoriously difficult to remove and strictly regulated in final drug substances. Consequently, the impurity profile of the resulting intermediates is significantly cleaner, simplifying the quality control processes required for regulatory compliance.
Furthermore, the versatility of this mechanism allows for extensive structural diversification, which is essential for structure-activity relationship (SAR) studies in drug discovery. The R1 group on the carbonyl moiety can tolerate a wide array of aromatic and heteroaromatic systems, including phenyl rings substituted with electron-donating methoxy groups or electron-withdrawing bromo and chloro atoms. Similarly, the nitrogen substituent (R3) can be varied from simple phenyl groups to more complex functionalities like ethoxycarbonyl or cyclopropyl groups without compromising reaction efficiency. This broad substrate scope indicates that the transition state of the cyclization is not overly sensitive to steric hindrance or electronic variations, making it a reliable platform for generating diverse libraries of thiazine derivatives. Such flexibility ensures that R&D teams can rapidly access novel chemical space for biological evaluation without being constrained by synthetic limitations.
How to Synthesize 3,4-Dihydro-2H-1,3-Thiazine Derivatives Efficiently
Implementing this synthesis in a production environment requires adherence to specific operational parameters to maximize yield and purity. The process begins with the dissolution of the alpha-enol dithioester and the 1,3,5-triazine precursor in dichloroethane, maintaining a solution concentration of approximately 0.1M to ensure optimal reaction kinetics. The mixture is then heated to 60°C, a temperature that balances reaction rate with energy efficiency and safety. Monitoring the reaction progress is straightforward, typically involving TLC or HPLC analysis to confirm the complete disappearance of the starting dithioester. Once the reaction is complete, the workup is remarkably simple, involving only the removal of the solvent under reduced pressure followed by standard silica gel chromatography. This ease of execution makes it an ideal candidate for both rapid prototyping in the lab and large-scale manufacturing.
- Dissolve the alpha-enol dithioester (Compound III) and 1,3,5-triazine (Compound II) in dichloroethane (DCE) solvent at a concentration of 0.1M.
- Heat the reaction mixture to 60°C and maintain stirring until the starting material (Compound III) is completely consumed, typically requiring approximately 24 hours.
- Remove the organic solvent under reduced pressure and purify the crude residue using silica gel column chromatography with a petroleum ether and ethyl acetate gradient.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis protocol offers tangible strategic benefits that extend beyond mere chemical curiosity. The elimination of precious metal catalysts represents a direct reduction in raw material costs, removing the volatility associated with the pricing of rare earth elements and transition metals. Moreover, the simplified post-treatment process, which does not require specialized metal scavenging resins or complex extraction protocols, translates into significant savings in processing time and consumable usage. The ability to run the reaction without inert gas protection further lowers the barrier to entry for manufacturing, as it reduces the demand for high-specification reactors and continuous nitrogen supply, thereby enhancing overall plant throughput and asset utilization.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the complete avoidance of expensive catalytic systems. Traditional methods often rely on lanthanide or noble metal catalysts which are not only costly to purchase but also require rigorous recovery or disposal procedures to meet environmental standards. By operating under catalyst-free conditions, the process eliminates these line items entirely. Additionally, the high atom economy and single-step nature of the reaction minimize solvent consumption and waste generation, leading to a leaner, more cost-effective manufacturing profile that directly improves the gross margin of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity-grade starting materials. Alpha-enol dithioesters and 1,3,5-triazines can be synthesized from widely available ketones, amines, and formaldehyde, ensuring a stable and diversified supply base. This reduces the risk of production stoppages caused by the shortage of specialized reagents. Furthermore, the robustness of the reaction conditions—specifically the tolerance to ambient atmosphere—means that production is less susceptible to disruptions in utility supplies such as high-purity nitrogen. This reliability ensures consistent delivery schedules for downstream customers, a critical factor in maintaining long-term partnerships in the competitive pharmaceutical market.
- Scalability and Environmental Compliance: From a scale-up perspective, the mild thermal requirements (60°C) and the use of standard organic solvents like dichloroethane make this process highly adaptable to existing infrastructure. There is no need for cryogenic cooling or high-pressure equipment, which simplifies engineering controls and reduces capital expenditure for new capacity. Environmentally, the absence of heavy metals simplifies wastewater treatment and solid waste disposal, aligning with increasingly stringent global regulations on chemical manufacturing. This 'green' aspect not only mitigates regulatory risk but also enhances the corporate sustainability profile of the manufacturer, a growing priority for multinational buyers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thiazine synthesis technology. These answers are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a factual basis for decision-making. Understanding these details is crucial for R&D directors evaluating process feasibility and procurement officers assessing vendor capabilities. The clarity of the reaction mechanism and the simplicity of the workup procedure are key differentiators that set this method apart from older, more cumbersome synthetic routes.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the patented method (CN111848547B) operates under metal-free conditions, eliminating the need for costly Lewis acids or precious metal catalysts often required in traditional heterocycle synthesis.
Q: What are the typical reaction conditions for this cyclization?
A: The reaction proceeds efficiently in dichloroethane (DCE) at a mild temperature of 60°C, without the necessity for inert gas protection, making it highly suitable for standard reactor setups.
Q: Can this method accommodate diverse substituent groups?
A: Yes, the process demonstrates excellent substrate scope, tolerating various phenyl substituents (methoxy, bromo, chloro), heterocyclic groups (pyridyl, furyl), and alkyl chains on the thiazine core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,4-Dihydro-2H-1,3-Thiazine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthetic methodologies play in accelerating drug development timelines. Our technical team has thoroughly analyzed the potential of this metal-free cyclization route and is fully prepared to leverage it for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial supply is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 3,4-dihydro-2H-1,3-thiazine derivatives meets the highest standards required for pharmaceutical applications.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this protocol for your specific target molecules. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your portfolio. Let us collaborate to bring high-quality, cost-effective sulfur-containing heterocycles to your pipeline with speed and precision.
