Advanced Synthesis of Paclitaxel Muramyl Dipeptide Conjugates for Scalable API Manufacturing
Introduction to Next-Generation Taxane Immunotherapy Intermediates
The pharmaceutical landscape for oncology treatments is constantly evolving, driven by the need to overcome the limitations of established drugs like paclitaxel. Patent CN1712399B introduces a groundbreaking class of conjugates linking paclitaxel with muramyl dipeptide (MDP), a potent immunopotentiator. This innovation addresses the critical challenges of poor water solubility and dose-limiting toxicity associated with conventional paclitaxel formulations. By chemically conjugating these two distinct pharmacophores, the technology creates a prodrug system that not only improves physicochemical properties but also merges chemotherapy with immunotherapy. For R&D directors and procurement specialists, understanding the synthesis of these complex molecules is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials for clinical development.
The core innovation lies in the strategic attachment of the MDP moiety to specific positions on the taxane skeleton, namely the 7-O, 2'-O, or 3'-N positions. This structural modification is not merely a derivatization exercise; it is a calculated effort to enhance bioavailability and reduce the reliance on harmful excipients like Cremophor EL. The patent details a sophisticated hybrid methodology combining liquid-phase synthesis for the taxane intermediates and solid-phase peptide synthesis (SPPS) for the MDP component. This dual approach ensures precise control over stereochemistry and linkage stability, which are paramount for maintaining biological activity. As we delve deeper into the technical specifics, it becomes clear that this process represents a significant leap forward in the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the clinical administration of paclitaxel has been hampered by its extremely low water solubility, measured at approximately 0.025 mg/mL. To make the drug injectable, manufacturers have relied on a vehicle containing 50% polyethoxylated castor oil (Cremophor EL) and 50% ethanol. This formulation strategy introduces severe drawbacks, including life-threatening hypersensitivity reactions that necessitate pre-medication with corticosteroids and antihistamines. Furthermore, Cremophor EL can alter the pharmacokinetics of paclitaxel, leading to unpredictable plasma levels and increased toxicity. From a manufacturing perspective, the requirement for such complex and hazardous formulation additives increases the cost reduction in API manufacturing challenges and complicates the supply chain due to stringent safety regulations surrounding these excipients. The inability to simply dissolve the drug in aqueous media limits the flexibility of dosage forms and administration routes.
The Novel Approach
The novel approach outlined in the patent circumvents these formulation hurdles by fundamentally altering the drug molecule itself rather than just its vehicle. By covalently bonding muramyl dipeptide to paclitaxel via a succinic acid linker, the resulting conjugate exhibits markedly improved water solubility. This chemical modification allows for the potential development of formulations that are free from Cremophor EL, thereby mitigating the risk of allergic reactions. The synthesis utilizes a robust solid-phase strategy where the MDP sequence is assembled on a 2-Chlorotrityl resin, providing a stable anchor for subsequent conjugation. 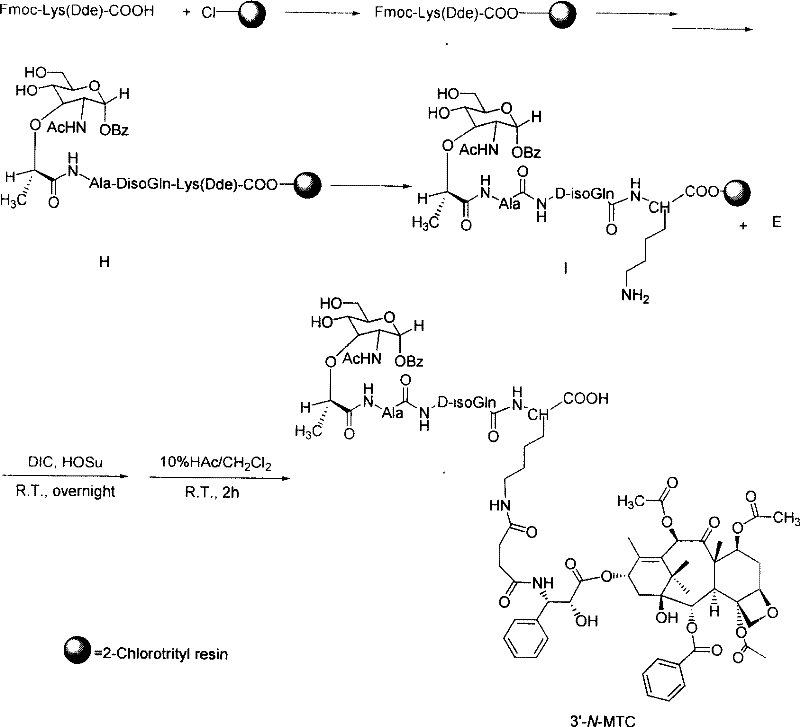 This method facilitates easy purification through simple washing steps, removing excess reagents and by-products before the final cleavage. The result is a high-purity intermediate that simplifies downstream processing and enhances the overall reliability of the production workflow for high-purity pharmaceutical intermediates.
This method facilitates easy purification through simple washing steps, removing excess reagents and by-products before the final cleavage. The result is a high-purity intermediate that simplifies downstream processing and enhances the overall reliability of the production workflow for high-purity pharmaceutical intermediates.
Mechanistic Insights into Hybrid Solid-Liquid Phase Conjugation
The chemical architecture of these conjugates relies on a precise sequence of protection, activation, and coupling reactions. The process begins with the liquid-phase modification of the taxane core. For instance, to prepare the 3'-N conjugate, the 7-acetoxy-Baccatin III is first reacted with a protected side chain to form the C-13 ester. Subsequent deprotection reveals the 3'-amino group, which is then acylated with succinic anhydride to introduce a carboxylic acid handle. This handle serves as the attachment point for the MDP peptide. The reaction conditions are meticulously controlled, often employing reagents like diisopropylcarbodiimide (DIC) and 4-dimethylaminopyridine (DMAP) in anhydrous toluene at elevated temperatures, such as 80°C, to drive the esterification to completion with yields reaching up to 61%.
Parallel to the taxane modification, the muramyl dipeptide fragment is constructed using standard Fmoc-based solid-phase peptide synthesis. The use of a lysine residue with an orthogonal Dde protecting group on the epsilon-amino side chain is a critical design feature. This allows for the selective deprotection of the epsilon-amino group while keeping the alpha-amino group intact or vice versa, enabling the directional coupling of the taxane moiety. 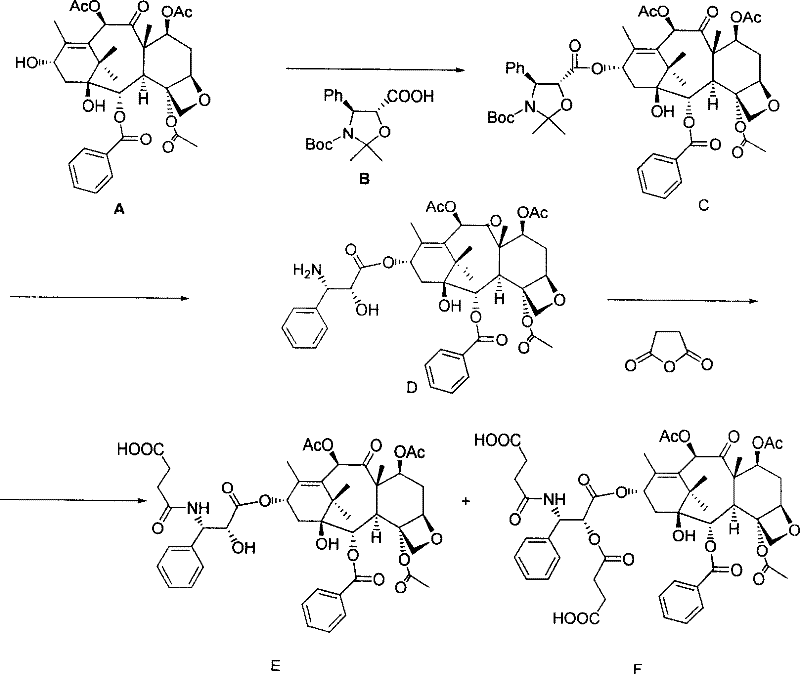 Once the MDP sequence is fully assembled on the resin, the Dde group is removed using hydrazine, exposing the amine for nucleophilic attack on the activated succinate of the taxane intermediate. The final cleavage from the resin using acidic conditions, such as 10% acetic acid in dichloromethane, releases the target conjugate. This mechanistic pathway ensures that the sensitive oxetane ring and ester functionalities of the taxane remain intact throughout the synthesis, preserving the cytotoxic potency of the final product.
Once the MDP sequence is fully assembled on the resin, the Dde group is removed using hydrazine, exposing the amine for nucleophilic attack on the activated succinate of the taxane intermediate. The final cleavage from the resin using acidic conditions, such as 10% acetic acid in dichloromethane, releases the target conjugate. This mechanistic pathway ensures that the sensitive oxetane ring and ester functionalities of the taxane remain intact throughout the synthesis, preserving the cytotoxic potency of the final product.
How to Synthesize Paclitaxel-MDP Conjugates Efficiently
Executing this synthesis requires a seamless integration of solution-phase organic chemistry and solid-phase techniques. The process demands strict adherence to anhydrous conditions and precise stoichiometry to minimize impurities. The following guide outlines the critical operational phases derived from the patent examples, focusing on the preparation of the key intermediates and their final assembly. For detailed laboratory protocols and safety data sheets, please refer to the standardized synthesis steps provided below.
- Prepare paclitaxel intermediates by introducing a succinic acid monoester or monoamide linker at the 7-O, 2'-O, or 3'-N position via liquid-phase chemistry.
- Synthesize the muramyl dipeptide (MDP) sequence on a 2-Chlorotrityl resin using Fmoc protection strategies, ensuring a free epsilon-amino group on the lysine linker.
- Couple the functionalized paclitaxel intermediate to the resin-bound MDP using DIC and HOSu condensing agents, followed by cleavage and purification to obtain the final conjugate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this conjugate technology offers substantial strategic benefits beyond mere chemical novelty. The primary advantage lies in the potential for significant cost reduction in pharmaceutical intermediate manufacturing by simplifying the final drug formulation. By eliminating the need for Cremophor EL, manufacturers can reduce the costs associated with sourcing, handling, and disposing of hazardous excipients. Furthermore, the improved solubility of the conjugate may allow for higher drug loading in formulations, reducing the volume of packaging and shipping materials required. This efficiency translates directly into a more streamlined and cost-effective supply chain operation.
- Cost Reduction in Manufacturing: The solid-phase synthesis protocol inherently simplifies purification. Unlike traditional solution-phase peptide synthesis which often requires tedious chromatography after each step, the resin-bound intermediates can be purified by simple filtration and washing. This reduction in solvent consumption and chromatography media drastically lowers the variable costs of production. Additionally, the high purity achieved (>97% as per patent examples) minimizes the loss of expensive taxane starting materials during final purification, ensuring better overall mass balance and yield efficiency.
- Enhanced Supply Chain Reliability: The modular nature of this synthesis allows for the independent production of the taxane linker intermediate and the MDP-resin. This decoupling means that supply chain disruptions in one area do not necessarily halt the entire production line. Manufacturers can stockpile stable intermediates and perform the final conjugation on demand, offering greater flexibility in meeting market needs. The use of commercially available reagents like DIC, HOSu, and standard Fmoc-amino acids further ensures that the raw material supply is robust and less prone to geopolitical or logistical bottlenecks.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the well-established infrastructure for solid-phase peptide synthesis in the fine chemical industry. The process avoids the use of heavy metal catalysts, aligning with increasingly stringent environmental regulations regarding residual metals in APIs. The reduction in organic solvent usage during purification steps also contributes to a lower environmental footprint, supporting corporate sustainability goals and reducing the costs associated with waste treatment and regulatory compliance audits.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of paclitaxel-MDP conjugates. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into your existing product pipeline.
Q: How does the paclitaxel-MDP conjugate improve upon traditional Taxol formulations?
A: Traditional Taxol requires Cremophor EL as a solubilizer, which causes severe hypersensitivity reactions. The conjugate described in CN1712399B significantly enhances water solubility through covalent bonding with the hydrophilic muramyl dipeptide, potentially eliminating the need for toxic emulsifiers while adding immunotherapeutic benefits.
Q: What are the critical reaction conditions for the solid-phase coupling step?
A: The coupling reaction typically utilizes diisopropylcarbodiimide (DIC) and N-hydroxysuccinimide (HOSu) in anhydrous dimethylformamide (DMF). The reaction is conducted at room temperature with shaking overnight to ensure complete conversion, monitored by ninhydrin testing for free amino groups.
Q: Does the conjugation process affect the anticancer activity of paclitaxel?
A: Biological assays indicate that the conjugates retain potent antitumor activity comparable to paclitaxel itself. Furthermore, the MDP moiety provides a synergistic immunostimulatory effect, enhancing TNF-alpha and IL-12 secretion in macrophages, which may improve overall therapeutic efficacy against tumor metastasis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Paclitaxel-MDP Conjugate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the technology described in CN1712399B for the future of oncology therapeutics. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical trials to market launch is seamless. We are committed to delivering high-purity pharmaceutical intermediates that meet the most stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex conjugation chemistries with the utmost precision and safety.
We invite you to collaborate with us to explore the full commercial viability of these innovative conjugates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us today to request specific COA data and route feasibility assessments. Let us partner with you to bring this next-generation immunotherapy to patients faster and more efficiently, leveraging our expertise in reducing lead time for high-purity pharmaceutical intermediates.
