Advanced Hydroformylation Technology for Commercial Scale Production of 4-Hydroxybutyraldehyde Intermediates
The chemical industry continuously seeks advancements in catalytic processes that enhance selectivity and economic viability, particularly for high-volume intermediates like 4-hydroxybutyraldehyde. Patent CN101652179B introduces a transformative hydroformylation method that addresses long-standing challenges in the production of this critical compound. By utilizing a specialized rhodium catalyst system paired with a unique diphosphine ligand, this technology achieves an unexpectedly high ratio of linear 4-hydroxybutyraldehyde to branched by-products. For R&D Directors and Procurement Managers, this represents a significant opportunity to optimize supply chains for downstream derivatives such as 1,4-Butanediol (BDO). The ability to control regioselectivity at the molecular level not only improves yield but also simplifies purification, making it a cornerstone technology for any reliable fine chemical intermediates supplier aiming to deliver high-purity OLED material or pharmaceutical precursors. This report analyzes the technical merits and commercial implications of adopting this novel synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional hydroformylation processes for converting allyl alcohol into 4-hydroxybutyraldehyde have historically relied on catalyst systems employing simple tri-substituted phosphines, such as triphenylphosphine. While these conventional methods are well-established, they suffer from inherent limitations regarding regioselectivity and by-product formation. A significant disadvantage is the concurrent production of branched isomers like 3-hydroxy-2-methylpropanal (HMPA) and various C3 by-products such as n-propanol and propionaldehyde. These impurities not only reduce the overall yield of the desired linear product but also complicate the downstream separation processes, leading to increased operational costs and waste generation. Furthermore, the formation of these co-products effectively represents a loss of raw material efficiency, which can have serious adverse effects on process economics when scaled to industrial levels. The inability to sufficiently suppress these side reactions often necessitates complex purification trains, thereby extending production timelines and reducing the overall throughput of manufacturing facilities.
The Novel Approach
The innovative process described in the patent data overcomes these deficiencies through the introduction of a sterically demanding diphosphine ligand known as trans-1,2-bis(bis(3,5-di-n-alkylphenyl)phosphinomethyl)cyclobutane. This specific ligand architecture modifies the electronic and steric environment of the rhodium active site, favoring the formation of the linear aldehyde over the branched isomer. Experimental data indicates that this novel catalyst system yields a significantly higher ratio of 4-hydroxybutyraldehyde to 3-hydroxy-2-methylpropanal compared to standard ligands like DIOP or simple bis(diphenylphosphinomethyl)cyclobutane derivatives. By minimizing the generation of unwanted C3 by-products and branched isomers, the process enhances the overall atom economy and reduces the burden on separation units. This technological leap allows for cost reduction in pharma intermediates manufacturing by streamlining the workflow and ensuring a more consistent quality of the crude product before it even enters the purification stage, thereby supporting the commercial scale-up of complex fine chemical intermediates.
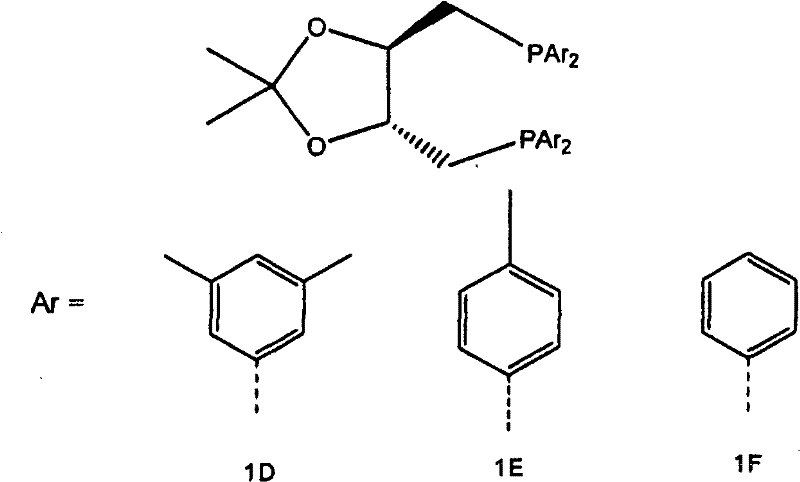
Mechanistic Insights into Rhodium-Catalyzed Hydroformylation
The core of this technological advancement lies in the precise interaction between the rhodium metal center and the bulky diphosphine ligand during the catalytic cycle. The ligand, trans-1,2-bis(bis(3,5-di-n-alkylphenyl)phosphinomethyl)cyclobutane, features 3,5-di-n-alkylphenyl groups that create a specific steric pocket around the metal. This configuration discourages the coordination of the olefin in a manner that would lead to branched product formation, effectively steering the reaction pathway towards the linear 4-hydroxybutyraldehyde. The electronic properties of the alkyl-substituted phenyl rings also modulate the electron density on the phosphorus atoms, which in turn influences the migratory insertion steps of the hydroformylation cycle. Understanding this mechanistic nuance is crucial for R&D teams aiming to replicate or further optimize the process, as slight variations in the alkyl chain length (methyl, ethyl, or propyl) can impact the catalyst's performance. The stability of the rhodium complex under reaction conditions is also enhanced by this chelating ligand, reducing metal leaching and ensuring longer catalyst life.
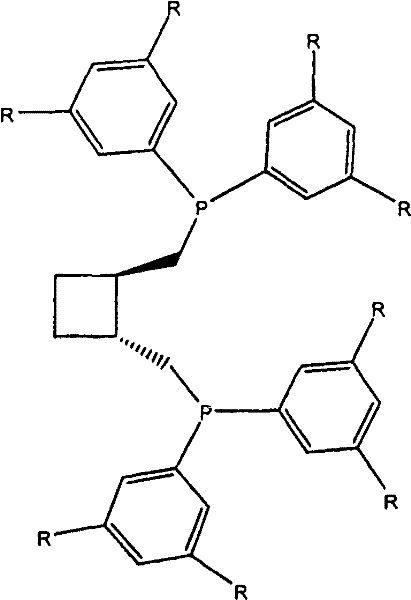
Impurity control is another critical aspect governed by the catalyst design. In conventional systems, the formation of C3 by-products like propionaldehyde often results from competing hydrogenation or decarbonylation pathways that are not sufficiently suppressed. The novel ligand system described in the patent data demonstrates a remarkable ability to minimize these side reactions, maintaining a high conversion rate of allyl alcohol while keeping by-product levels negligible. This high selectivity is vital for maintaining the purity specifications required for downstream applications, such as the synthesis of high-purity 4-hydroxybutyraldehyde for polymer or pharmaceutical use. By reducing the load of impurities early in the synthesis, the need for aggressive purification methods is diminished, which aligns with green chemistry principles and reduces the environmental footprint of the manufacturing process. This level of control over the impurity profile is a key differentiator for suppliers targeting high-value markets where consistency and quality are paramount.
How to Synthesize 4-Hydroxybutyraldehyde Efficiently
Implementing this synthesis route requires careful attention to catalyst preparation and reaction conditions to fully realize the benefits of the novel ligand system. The process begins with the formation of the active catalyst species, typically by combining a rhodium precursor such as rhodium acetylacetonate dicarbonyl with the diphosphine ligand in a solvent like toluene. It is essential to maintain an inert atmosphere during this preparation to prevent oxidation of the phosphine ligand, which could deactivate the catalyst. Once the catalyst is formed, the hydroformylation reaction proceeds under mild temperatures ranging from 50-80°C and moderate pressures, which are favorable for industrial safety and energy efficiency. The detailed standardized synthesis steps see the guide below for specific molar ratios and operational parameters that ensure optimal performance.
- Prepare the catalyst system by combining a rhodium complex precursor with trans-1,2-bis(bis(3,5-di-n-alkylphenyl)phosphinomethyl)cyclobutane ligand in a suitable organic solvent such as toluene.
- Charge the reactor with allyl alcohol and pressurize with a carbon monoxide and hydrogen gas mixture, maintaining specific partial pressures to optimize the linear-to-branched product ratio.
- Conduct the reaction at controlled temperatures between 50-80°C, followed by water extraction to separate the 4-hydroxybutyraldehyde product from the organic catalyst phase.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this advanced hydroformylation technology offers tangible benefits that extend beyond mere chemical yield. The primary advantage lies in the substantial cost savings achieved through improved selectivity and reduced waste. By significantly increasing the ratio of linear to branched products, the process minimizes the loss of valuable raw materials into unusable by-products, thereby lowering the effective cost per unit of the final product. This efficiency gain is compounded by the simplified downstream processing, as the reduced impurity load requires less energy and fewer solvents for purification. Consequently, this leads to a more robust and predictable manufacturing cost structure, allowing for better budget forecasting and pricing stability for long-term contracts. The elimination of complex purification steps also reduces the turnaround time for production batches, enhancing the overall responsiveness of the supply chain to market demands.
- Cost Reduction in Manufacturing: The implementation of this catalyst system directly translates to financial efficiency by maximizing the utilization of allyl alcohol and synthesis gas. Since the formation of unwanted C3 by-products and branched isomers is drastically suppressed, the volume of waste requiring treatment or disposal is significantly reduced. This reduction in waste handling costs, combined with the higher yield of the target molecule, results in a lower overall production cost without compromising on quality. Furthermore, the mild reaction conditions reduce energy consumption associated with heating and pressurization, contributing to a leaner operational expenditure profile. These factors collectively enable a competitive pricing strategy while maintaining healthy margins, which is essential for sustaining a reliable fine chemical intermediates supplier status in a volatile market.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by process inefficiencies and yield fluctuations, but this technology mitigates those risks through its robust catalytic performance. The high conversion rates and consistent selectivity ensure that production targets are met reliably, reducing the likelihood of delays caused by off-spec batches. Additionally, the catalyst system's stability allows for longer operational runs without frequent regeneration or replacement, minimizing downtime for maintenance. This reliability is crucial for reducing lead time for high-purity chemical intermediates, as it ensures a steady flow of material to downstream customers. By securing a more predictable production schedule, companies can better manage inventory levels and fulfill large-scale orders with greater confidence, strengthening partnerships with key stakeholders in the pharmaceutical and polymer industries.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often introduces new challenges, but this hydroformylation method is designed with scalability in mind. The use of common solvents like toluene and the ability to separate the product via simple water extraction simplify the engineering requirements for large-scale reactors. This ease of scale-up facilitates the transition from pilot plants to full commercial production, ensuring that the benefits observed in the lab are retained at the 100 MT level. Moreover, the reduction in by-products and the use of efficient separation techniques align with stringent environmental regulations, reducing the burden of waste management and emissions control. This environmental compliance not only avoids potential regulatory fines but also enhances the corporate sustainability profile, which is increasingly important for global supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydroformylation technology. These answers are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical principles involved. Understanding these details is essential for technical teams evaluating the feasibility of integrating this process into their existing manufacturing frameworks. The insights provided here highlight the specific improvements over prior art and the practical benefits for industrial application.
Q: How does the novel ligand improve the linear-to-branched ratio compared to conventional triphenylphosphine?
A: The specialized trans-1,2-bis(bis(3,5-di-n-alkylphenyl)phosphinomethyl)cyclobutane ligand provides superior steric control around the rhodium center, significantly suppressing the formation of branched 3-hydroxy-2-methylpropanal by-products and enhancing the yield of the desired linear 4-hydroxybutyraldehyde.
Q: What are the primary operational advantages of this hydroformylation process for large-scale manufacturing?
A: This process operates under relatively mild conditions (50-80°C) and utilizes a catalyst system that allows for efficient separation via water extraction, reducing energy consumption and simplifying the downstream purification workflow for commercial scale-up of complex fine chemical intermediates.
Q: Can this technology be adapted for the production of 1,4-Butanediol (BDO)?
A: Yes, the 4-hydroxybutyraldehyde produced via this method is a direct precursor to 1,4-Butanediol. The high purity and selectivity of the aldehyde intermediate ensure a more efficient subsequent hydrogenation step, minimizing impurities in the final BDO product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxybutyraldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes for high-value intermediates like 4-hydroxybutyraldehyde. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in patent literature can be successfully translated into industrial reality. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch meets the exacting standards required by the global pharmaceutical and fine chemical industries. Our infrastructure is designed to handle complex catalytic processes safely and efficiently, providing a secure foundation for your supply chain needs.
We invite you to collaborate with us to optimize your sourcing strategy for this key intermediate. By leveraging our technical expertise, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project goals. Together, we can drive innovation and efficiency in the production of essential chemical building blocks.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
