Scalable Synthesis of Phenylalanine Esters via Optimized Schiff Base Alkylation for Commercial Production
Scalable Synthesis of Phenylalanine Esters via Optimized Schiff Base Alkylation for Commercial Production
The pharmaceutical industry continuously demands more efficient and cost-effective routes for synthesizing critical amino acid derivatives, particularly phenylalanine esters which serve as pivotal intermediates for endothelin antagonists and other bioactive molecules. Patent CN107778194B introduces a robust preparation and resolution method that addresses the longstanding challenges of low yield and complex purification associated with traditional syntheses. This innovative approach leverages a streamlined alkylation strategy followed by controlled hydrolysis, offering a pathway that is not only chemically elegant but also commercially viable for large-scale operations. By optimizing reaction parameters such as temperature and molar ratios, the process ensures high conversion rates while minimizing the formation of difficult-to-remove impurities. For R&D directors and procurement managers alike, this technology represents a significant opportunity to enhance the reliability of the supply chain for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phenylalanine compounds has relied on methodologies disclosed in earlier patents, such as U.S. Pat. No. 5,943 and U.S. Pat. No. 5,567,943 assigned to Warner-Lambert Company. These conventional routes often involve cumbersome multi-step sequences that require harsh reaction conditions and expensive catalysts, leading to suboptimal overall yields. The complexity of these legacy processes frequently results in the generation of significant byproduct profiles, necessitating rigorous and costly purification steps to meet the stringent purity specifications required for API manufacturing. Furthermore, the reliance on specialized reagents in these older methods can introduce supply chain vulnerabilities, causing delays and price volatility for downstream manufacturers. The inability to easily scale these intricate pathways without compromising quality has long been a bottleneck for the commercial production of phenylalanine derivatives.
The Novel Approach
In stark contrast, the novel methodology presented in the patent data utilizes a direct alkylation of a Schiff base compound with a specifically prepared alkylating agent, followed by a straightforward hydrolysis step. This streamlined sequence drastically reduces the number of unit operations required, thereby lowering the overall processing time and resource consumption. The core of this innovation lies in the efficient coupling of the alkylating agent with the Schiff base under mild basic conditions, which preserves the integrity of sensitive functional groups while driving the reaction to completion. 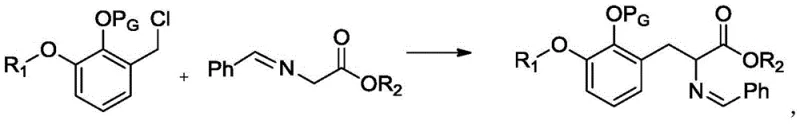 As illustrated in the reaction scheme, this approach bypasses the need for complex protecting group manipulations often seen in prior art, resulting in a cleaner reaction profile. The subsequent hydrolysis is equally efficient, allowing for the precise control of the final product form, whether as a free base or an acid salt, simply by adjusting the amount of acid used.
As illustrated in the reaction scheme, this approach bypasses the need for complex protecting group manipulations often seen in prior art, resulting in a cleaner reaction profile. The subsequent hydrolysis is equally efficient, allowing for the precise control of the final product form, whether as a free base or an acid salt, simply by adjusting the amount of acid used.
Mechanistic Insights into Alkylation and Hydrolysis Mechanisms
The success of this synthetic route hinges on the precise execution of the alkylating agent preparation and the subsequent nucleophilic substitution. The process begins with the conversion of a protected benzyl alcohol derivative into the corresponding chloride using a chlorinating agent such as thionyl chloride. 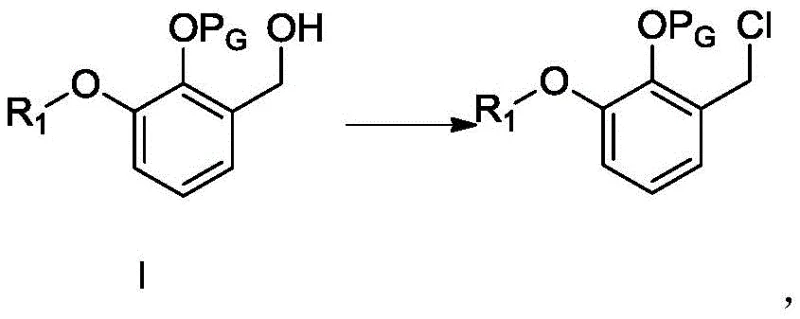 This transformation is critical, as the resulting alkyl chloride serves as a highly reactive electrophile for the subsequent alkylation step. The reaction is typically conducted at temperatures ranging from 0 to 50°C, with a preference for the lower end of this spectrum to minimize side reactions. Following this, the alkylating agent reacts with the Schiff base in the presence of a base like sodium ethoxide. The base deprotonates the active methylene group of the Schiff base, generating a nucleophile that attacks the alkyl chloride. This step is exothermic and requires careful temperature control, ideally between 0 and 20°C, to prevent racemization or decomposition of the Schiff base.
This transformation is critical, as the resulting alkyl chloride serves as a highly reactive electrophile for the subsequent alkylation step. The reaction is typically conducted at temperatures ranging from 0 to 50°C, with a preference for the lower end of this spectrum to minimize side reactions. Following this, the alkylating agent reacts with the Schiff base in the presence of a base like sodium ethoxide. The base deprotonates the active methylene group of the Schiff base, generating a nucleophile that attacks the alkyl chloride. This step is exothermic and requires careful temperature control, ideally between 0 and 20°C, to prevent racemization or decomposition of the Schiff base.
Following the alkylation, the intermediate undergoes hydrolysis to reveal the free amine and ester functionalities. The patent specifies the use of inorganic acids, particularly hydrochloric acid, to facilitate this cleavage. 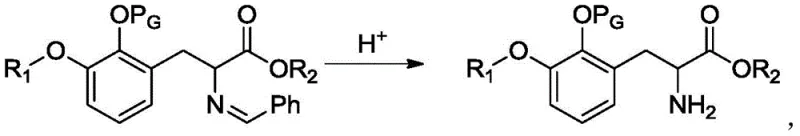 The mechanism involves the protonation of the imine nitrogen, making it susceptible to nucleophilic attack by water, which eventually leads to the release of the amine and the carbonyl byproduct. A key feature of this process is the ability to control the stoichiometry of the acid to dictate the final product form. By maintaining a molar ratio of acid to alkylated Schiff base between 1.5 and 3.0, the free phenylalanine ester is obtained, whereas increasing this ratio to 3.0-4.0 yields the acid salt directly. This flexibility eliminates the need for a separate salt formation step, further enhancing the process efficiency and reducing waste generation.
The mechanism involves the protonation of the imine nitrogen, making it susceptible to nucleophilic attack by water, which eventually leads to the release of the amine and the carbonyl byproduct. A key feature of this process is the ability to control the stoichiometry of the acid to dictate the final product form. By maintaining a molar ratio of acid to alkylated Schiff base between 1.5 and 3.0, the free phenylalanine ester is obtained, whereas increasing this ratio to 3.0-4.0 yields the acid salt directly. This flexibility eliminates the need for a separate salt formation step, further enhancing the process efficiency and reducing waste generation.
How to Synthesize Phenylalanine Ester Efficiently
Implementing this synthesis requires strict adherence to the optimized conditions outlined in the patent to ensure maximum yield and purity. The process is divided into three distinct phases: the preparation of the alkylating agent, the alkylation of the Schiff base, and the final hydrolysis. Each phase demands precise control over temperature and reagent addition rates to manage exotherms and prevent the formation of impurities. The detailed standardized synthesis steps below provide a roadmap for replicating this high-efficiency route in a pilot or production setting, ensuring that the critical quality attributes of the final phenylalanine ester are consistently met.
- Prepare the alkylating agent by reacting the protected benzyl alcohol derivative with thionyl chloride at 0-50°C.
- Perform the alkylation reaction by mixing the alkylating agent with the Schiff base compound in the presence of a base like sodium ethoxide at 0-20°C.
- Hydrolyze the alkylated Schiff base using an inorganic acid such as hydrochloric acid to obtain the final phenylalanine ester compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers transformative benefits that extend far beyond simple chemical yield improvements. The simplification of the synthetic pathway directly translates to a reduction in the number of raw materials required, which streamlines inventory management and reduces the risk of supply disruptions caused by niche reagent shortages. By utilizing common industrial chemicals like thionyl chloride and sodium ethoxide, manufacturers can leverage existing supply networks to secure materials at competitive prices, thereby achieving substantial cost savings in pharmaceutical intermediates manufacturing. Furthermore, the robustness of the reaction conditions means that the process is less sensitive to minor variations in input quality, enhancing the overall reliability of the production schedule.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences and the avoidance of expensive transition metal catalysts significantly lower the operational expenditure associated with production. The high yield reported in the examples minimizes the loss of valuable starting materials, ensuring that the cost per kilogram of the final product is drastically reduced compared to legacy methods. Additionally, the ability to isolate the product as a salt directly from the hydrolysis step removes the need for a separate neutralization and crystallization stage, further cutting down on utility consumption and labor costs.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals rather than proprietary or hard-to-source reagents ensures a stable and continuous supply of inputs. This stability is crucial for maintaining uninterrupted production runs, especially when scaling up to meet the demands of global pharmaceutical markets. The simplified workflow also reduces the lead time for high-purity phenylalanine esters, allowing suppliers to respond more agilely to fluctuating market demands and urgent customer orders without compromising on quality standards.
- Scalability and Environmental Compliance: The mild reaction temperatures and the use of standard solvents like dichloromethane and ethyl acetate make this process highly scalable from laboratory to commercial tonnage. The reduced number of steps inherently generates less waste, aligning with modern green chemistry principles and easing the burden on wastewater treatment facilities. This environmental compatibility not only lowers disposal costs but also ensures compliance with increasingly stringent regulatory frameworks regarding industrial emissions and solvent usage.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phenylalanine ester synthesis technology. These answers are derived directly from the experimental data and technical disclosures within the patent, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the key advantages of this phenylalanine ester synthesis method over prior art?
A: The method described in CN107778194B offers significantly higher yields and avoids the complex multi-step sequences found in older Warner-Lambert patents. It utilizes readily available reagents like thionyl chloride and sodium ethoxide, simplifying the supply chain and reducing production costs.
Q: How is optical purity achieved in this process?
A: Optical purity is achieved through a resolution step using chiral acids such as L-tartaric acid or L-benzoyl tartaric acid. This allows for the isolation of specific enantiomers, such as the S-configuration phenylalanine ethyl ester, which is critical for biological activity in pharmaceutical applications.
Q: Can this process be scaled for industrial manufacturing?
A: Yes, the patent explicitly states the route is suitable for industrialization. The reaction conditions are mild (0-50°C), and the workup involves standard extraction and chromatography techniques that are easily adaptable to large-scale reactors, ensuring consistent quality and supply continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylalanine Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch. Our expertise in handling complex organic syntheses allows us to troubleshoot potential issues proactively, guaranteeing a consistent supply of phenylalanine esters that your R&D and production teams can rely on.
We invite you to collaborate with us to explore how this advanced synthesis route can optimize your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how this technology can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us partner with you to drive innovation and efficiency in your pharmaceutical supply chain.
