Advanced Synthesis of 2-(7-Methoxy-1-Naphthyl) Ethylamine Hydrochloride for Commercial API Production
The pharmaceutical industry continuously seeks robust, scalable, and environmentally benign pathways for the production of critical active pharmaceutical ingredient (API) intermediates. Patent CN103113243A, published in May 2013, introduces a significant technological advancement in the synthesis of 2-(7-methoxy-1-naphthyl) ethylamine hydrochloride, a pivotal precursor for the antidepressant Agomelatine. This novel methodology addresses longstanding challenges associated with traditional synthetic routes, specifically targeting the elimination of hazardous reagents and extreme reaction conditions. By leveraging a strategic combination of Knoevenagel-type condensation, 2,3-Dichloro-5,6-dicyano-1,4-benzoquinone (DDQ) mediated dehydrogenation, and a modified sodium borohydride reduction system, this process establishes a new benchmark for efficiency and safety. For global procurement leaders and R&D directors, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials while adhering to stringent regulatory and cost constraints.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of naphthalene-based ethylamine derivatives has been plagued by operational complexities and safety hazards that hinder large-scale manufacturing. Prior art, such as the methods described in patent EP0447285, relied on multi-step sequences involving ethyl bromoacetate, which is known for its severe pungency and adverse effects on labor protection. Furthermore, these legacy processes frequently utilized benzene as a solvent, a carcinogen that poses unacceptable environmental and health risks in modern green chemistry frameworks. Another prevalent approach, documented in patents like EP156420 and US20050182276, attempted to streamline the process but introduced different critical bottlenecks. These methods employed toxic allyl methacrylate (AMA) as a hydrogen acceptor during the dehydrogenation phase, creating significant waste disposal challenges. Moreover, the subsequent reduction steps often necessitated high-pressure hydrogenation environments, reaching up to 300 atmospheres, which requires specialized, capital-intensive equipment and introduces substantial safety liabilities for any commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
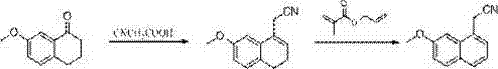
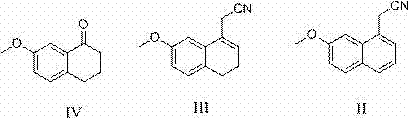
In stark contrast to these cumbersome legacy techniques, the methodology outlined in CN103113243A presents a streamlined, three-step trajectory that prioritizes operational simplicity and chemical safety. The process initiates with the condensation of 7-methoxy-1-tetralone and cyanoacetic acid, followed immediately by an elegant dehydrogenation step utilizing DDQ. This oxidant effectively aromatizes the tetralone ring system under mild thermal conditions, completely bypassing the need for toxic hydrogen acceptors or precious metal catalysts like palladium on carbon. The final transformation involves a sophisticated reduction protocol where sodium borohydride, activated by calcium chloride in anhydrous tetrahydrofuran, converts the nitrile functionality to the corresponding amine. This specific modification allows the reduction to proceed at atmospheric pressure and near-ambient temperatures, drastically simplifying the engineering requirements. For stakeholders focused on cost reduction in API manufacturing, this shift from high-pressure catalytic hydrogenation to chemical reduction represents a fundamental improvement in process economics and facility flexibility.
Mechanistic Insights into DDQ-Mediated Aromatization and Activated Borohydride Reduction
The core chemical innovation of this patent lies in the precise control of oxidation and reduction potentials to achieve high selectivity without compromising yield. The dehydrogenation step utilizes DDQ, a potent quinone oxidant, which operates through a hydride transfer mechanism. In this process, the dihydronaphthalene intermediate donates hydride ions to the DDQ molecule, resulting in the formation of the fully aromatic naphthalene system and the reduced hydroquinone byproduct. This mechanism is highly advantageous because it avoids the radical pathways often associated with catalytic dehydrogenation, thereby minimizing the formation of polymeric tars or over-oxidized side products that can complicate downstream purification. The reaction is typically conducted in dichloromethane, a solvent that provides excellent solubility for both the organic substrate and the polar quinone, ensuring homogeneous reaction kinetics and consistent heat transfer throughout the batch.
Furthermore, the reduction of the nitrile group to the primary amine demonstrates a masterful application of Lewis acid activation. While sodium borohydride alone is generally insufficient for reducing nitriles under mild conditions, the addition of calcium chloride generates a more reactive species in situ. The calcium cation coordinates with the nitrogen lone pair of the nitrile, increasing the electrophilicity of the cyano carbon and facilitating nucleophilic attack by the borohydride anion. This synergistic effect allows the reaction to proceed efficiently in tetrahydrofuran at temperatures ranging from 0°C to room temperature. From an impurity control perspective, this mildness is crucial; it prevents the over-reduction of the aromatic ring or the cleavage of the methoxy ether group, which are common degradation pathways in harsher hydrogenation protocols. Consequently, the resulting crude amine exhibits a superior purity profile, reducing the burden on final crystallization steps.
How to Synthesize 2-(7-Methoxy-1-Naphthyl) Ethylamine Efficiently
The execution of this synthetic route requires careful attention to stoichiometry and thermal management to maximize the benefits of the patented process. The initial condensation is performed under reflux with water removal to drive the equilibrium towards the nitrile product, after which the crude oil is carried forward without isolation. The subsequent DDQ oxidation must be monitored to ensure complete conversion while managing the exotherm associated with quinone reduction. Finally, the reduction step demands strict anhydrous conditions to maintain the activity of the borohydride-calcium chloride complex.
- Condense 7-methoxy-1-tetralone with cyanoacetic acid to form (7-methoxy-3,4-dihydro-1-naphthyl) acetonitrile.
- Perform dehydrogenation using DDQ in dichloromethane to aromatize the ring system.
- Reduce the nitrile group using sodium borohydride and calcium chloride in anhydrous THF, followed by acidification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical elegance. The elimination of high-pressure hydrogenation equipment significantly lowers the barrier to entry for contract manufacturing organizations, allowing for a broader base of qualified suppliers. This diversification enhances supply chain resilience, reducing the risk of bottlenecks that often occur when production is limited to facilities with specialized high-pressure capabilities. Additionally, the replacement of toxic reagents like allyl methacrylate and carcinogenic solvents like benzene aligns the manufacturing process with increasingly rigorous global environmental, health, and safety (EHS) standards. This compliance not only mitigates regulatory risk but also reduces the long-term costs associated with hazardous waste disposal and worker safety monitoring.
- Cost Reduction in Manufacturing: The transition from catalytic hydrogenation to chemical reduction using sodium borohydride and calcium chloride eliminates the need for expensive noble metal catalysts and the high-energy infrastructure required for high-pressure reactors. This shift results in substantial capital expenditure savings and lower operational overheads. Furthermore, the ability to telescope the first two steps without isolating the intermediate dihydronaphthyl acetonitrile reduces solvent consumption, filtration time, and drying energy. These cumulative efficiencies drive down the cost of goods sold (COGS), enabling more competitive pricing structures for the final API while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, including DDQ, sodium borohydride, and calcium chloride, are commodity chemicals with robust global supply chains. Unlike specialized catalysts or hazardous gases that may face logistical restrictions or supply volatility, these materials are readily available from multiple vendors. This accessibility ensures consistent production scheduling and reduces the lead time for high-purity pharmaceutical intermediates. Moreover, the mild reaction conditions minimize the risk of unplanned shutdowns due to equipment failure or safety incidents, guaranteeing a steady flow of material to downstream formulation partners.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing standard glass-lined or stainless steel reactors commonly found in multipurpose fine chemical plants. The absence of high-pressure operations simplifies the scale-up trajectory from pilot plant to commercial tonnage, reducing the time and cost required for process validation. From an environmental standpoint, the avoidance of benzene and allyl methacrylate significantly lowers the toxicity profile of the effluent stream. This facilitates easier wastewater treatment and helps manufacturers meet strict discharge limits, ensuring long-term operational sustainability and community acceptance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic pathway. Understanding these details is vital for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing supplier capabilities.
Q: Why is the DDQ dehydrogenation method preferred over catalytic dehydrogenation?
A: Traditional methods often utilize toxic allyl methacrylate (AMA) as a hydrogen acceptor with Pd/C catalysts, posing environmental and safety risks. The DDQ method offers a cleaner, more controllable oxidation process without requiring heavy metal catalysts or generating hazardous byproducts.
Q: What is the role of Calcium Chloride in the reduction step?
A: Calcium chloride acts as a Lewis acid additive that activates sodium borohydride, significantly enhancing its reducing power towards nitriles under mild conditions. This allows the reaction to proceed at ambient pressure and temperature, avoiding the need for dangerous high-pressure hydrogenation equipment.
Q: Does the intermediate require purification between steps?
A: No, the patent specifies that the crude (7-methoxy-3,4-dihydro-1-naphthyl) acetonitrile obtained from the first step can be directly used in the subsequent dehydrogenation reaction. This telescoping capability reduces solvent usage, labor intensity, and overall processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(7-Methoxy-1-Naphthyl) Ethylamine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex antidepressants like Agomelatine depends on a secure and high-quality supply of key intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative route described in CN103113243A can be seamlessly translated from the laboratory to the manufacturing floor. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 2-(7-methoxy-1-naphthyl) ethylamine hydrochloride meets the exacting standards required for GMP API synthesis. Our commitment to quality assurance ensures that potential impurities are identified and controlled at every stage of the process.
We invite global pharmaceutical partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements. By leveraging our optimized manufacturing protocols, we can help you achieve significant efficiencies in your supply chain without compromising on quality or compliance. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our expertise in advanced organic synthesis can become a cornerstone of your strategic sourcing initiative, driving value and reliability for your entire product portfolio.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
