Scalable Mannich Reaction Protocol for High-Purity Nucleoside Phosphorylase Inhibitors
The pharmaceutical industry constantly seeks more efficient pathways to synthesize complex bioactive molecules, particularly those targeting critical metabolic enzymes. Patent CN1771254A introduces a transformative approach to preparing a specific class of nucleoside analogs that serve as potent inhibitors for Purine Nucleoside Phosphorylase (PNP), purine phosphoribosyl transferase (PPRT), and related enzymes like MTAP and MTAN. These compounds, often referred to as transition state analogs or Immucillins, hold immense therapeutic potential for treating T-cell malignancies, autoimmune disorders, and parasitic infections. The core innovation lies in utilizing a modified Mannich reaction to directly couple cyclic amines with heteroaromatic bases, bypassing the laborious multi-step sequences traditionally required for such intricate molecular architectures. This technological leap not only simplifies the synthetic route but also enhances the overall economic viability of producing these high-value pharmaceutical intermediates.
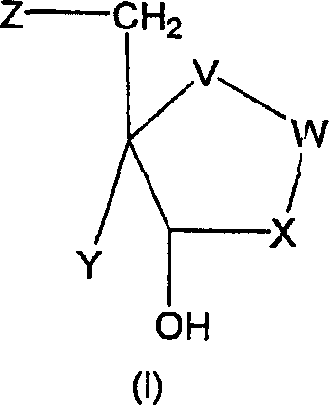
For research and development teams focused on process chemistry, understanding the structural versatility of Formula (I) is crucial. The patent delineates a broad scope where variables V, W, X, Y, and Z can be modified to tune the pharmacological profile, allowing for the creation of diverse libraries of inhibitors. The ability to access these structures through a convergent synthesis strategy means that medicinal chemists can rapidly iterate on lead compounds without being bottlenecked by complex synthesis timelines. This flexibility is essential in the early stages of drug discovery where speed to market and the ability to explore structure-activity relationships (SAR) are paramount. By establishing a robust platform technology for these deazapurine derivatives, manufacturers can support a wide range of downstream drug development programs with a single, optimized process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nucleoside analogs involving carbon-linked sugar mimics and heterocyclic bases has been plagued by inefficiencies inherent in traditional glycosylation or alkylation methods. Conventional routes often necessitate the use of sensitive organometallic reagents, strict anhydrous conditions, and multiple protection and deprotection steps to prevent side reactions on the numerous hydroxyl and amine functionalities present in the molecule. These multi-step sequences inevitably lead to cumulative yield losses, where the overall efficiency drops precipitously with each additional transformation. Furthermore, the reliance on expensive catalysts or difficult-to-handle reagents increases the cost of goods sold (COGS) and introduces significant safety hazards during scale-up. The purification of intermediates in these long sequences often requires extensive chromatography, which is not only solvent-intensive but also difficult to translate from laboratory glassware to industrial reactors, creating a significant barrier to commercialization.
The Novel Approach
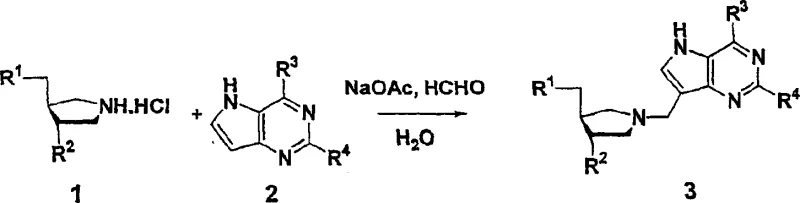
In stark contrast, the methodology disclosed in CN1771254A leverages the power of the Mannich condensation to achieve direct C-C bond formation between the amine and the heterocycle in a single operational step. This novel approach utilizes formaldehyde or its equivalents as a bridging unit, reacting spontaneously with the cyclic amine and the 9-deazapurine base under relatively mild aqueous or semi-aqueous conditions. The elegance of this method is its atom economy and step convergence; it effectively merges three components into the final scaffold without the need for pre-activating the heterocycle or the amine with complex leaving groups. As illustrated in the reaction scheme below, the process tolerates various functional groups, reducing the need for orthogonal protection strategies that typically bloat synthetic routes. This simplification translates directly into reduced processing time, lower solvent consumption, and a drastically simplified workup procedure, making it an ideal candidate for green chemistry initiatives in pharmaceutical manufacturing.
Mechanistic Insights into Mannich-Type Condensation
The mechanistic pathway of this transformation relies on the generation of an reactive iminium ion intermediate from the cyclic amine and formaldehyde. In the presence of a buffer system, typically sodium acetate, the equilibrium favors the formation of this electrophilic species, which is then susceptible to nucleophilic attack by the electron-rich position on the heteroaromatic ring, specifically the C-9 position of the 9-deazapurine system. This electrophilic aromatic substitution is facilitated by the specific electronic properties of the deazapurine ring, which possesses sufficient nucleophilicity to displace the proton from the iminium intermediate. The reaction kinetics are influenced by the pH of the medium and the temperature, with the patent specifying conditions around 95°C to ensure complete conversion while minimizing degradation of the sensitive heterocyclic base. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as the stoichiometry of formaldehyde, to drive the equilibrium towards the desired product and suppress the formation of bis-alkylated byproducts.
Impurity control is a critical aspect of this mechanism, particularly regarding the regioselectivity of the alkylation. The patent highlights that the reaction predominantly occurs at the nitrogen or carbon positions that offer the most stable transition state, thereby ensuring high regio-purity of the final inhibitor. The use of water as a co-solvent plays a dual role: it solubilizes the inorganic salts and formaldehyde while also helping to moderate the exothermic nature of the condensation. From a quality control perspective, the absence of heavy metal catalysts means that the final product is free from toxic metal residues, a significant advantage for regulatory filing. The robustness of this mechanism against varying substituents on the pyrrolidine ring suggests that the electronic demand of the nucleophile is well-matched by the electrophilicity of the iminium ion, providing a reliable platform for synthesizing a wide array of analogs with consistent purity profiles.
How to Synthesize Deazapurine Pyrrolidine Derivatives Efficiently
Executing this synthesis requires careful attention to the preparation of the starting materials, particularly the cyclic amine hydrochloride salts and the heterocyclic bases. The general procedure involves dissolving the amine salt and a stoichiometric amount of sodium acetate in a mixture of water and 1,4-dioxane, creating a homogeneous phase for the reaction to proceed. Formaldehyde is then introduced, either as an aqueous solution or as paraformaldehyde, followed by the addition of the 9-deazapurine derivative. The mixture is heated to reflux temperatures, typically around 95°C, and stirred for a period ranging from 1 to 16 hours depending on the specific reactivity of the substrates involved. Upon completion, the reaction mixture is concentrated, and the crude product is purified using silica gel chromatography with a gradient of dichloromethane, methanol, and ammonium hydroxide. For detailed standardized operating procedures and specific parameter optimizations, please refer to the guide below.
- Dissolve the cyclic amine hydrochloride and sodium acetate in a water and 1,4-dioxane mixture to create the reaction medium.
- Add aqueous formaldehyde solution and the substituted 9-deazapurine heterocycle to the mixture under stirring conditions.
- Stir the reaction at elevated temperatures (e.g., 95°C) for several hours, then purify the crude product via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this Mannich-based synthesis offers profound strategic benefits that extend beyond simple yield improvements. The elimination of multiple synthetic steps inherently reduces the exposure to supply chain disruptions associated with sourcing specialized reagents for each individual transformation. By consolidating the synthesis into a one-pot reaction, manufacturers can significantly decrease the lead time required to produce kilogram quantities of these critical intermediates. This agility is vital for pharmaceutical companies needing to ramp up production for clinical trials or commercial launch without the long validation cycles associated with complex multi-step processes. Furthermore, the use of commodity chemicals like formaldehyde and common solvents ensures a stable and predictable cost structure, shielding the project from the volatility often seen with exotic catalysts or custom-synthesized building blocks.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the drastic reduction in unit operations. Fewer reaction steps mean less labor, lower energy consumption for heating and cooling cycles, and a significant decrease in solvent usage for extractions and washes. Since the process avoids the use of expensive transition metal catalysts, there is no need for costly metal scavenging steps or the associated analytical testing to prove residual metal compliance. This streamlining of the process flow directly lowers the cost of goods sold, allowing for more competitive pricing in the generic or specialty chemical markets. Additionally, the higher overall yield resulting from fewer purification losses means that less raw material is required to produce the same amount of final API, further enhancing the economic efficiency of the manufacturing campaign.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as cyclic amines and simple heterocycles ensures a robust supply chain that is less prone to bottlenecks. Unlike processes dependent on single-source suppliers for chiral catalysts or unstable intermediates, the reagents for this Mannich reaction are produced by multiple global chemical manufacturers, fostering a competitive sourcing environment. This redundancy mitigates the risk of production halts due to raw material shortages. Moreover, the stability of the intermediates allows for potential stockpiling strategies, enabling manufacturers to build inventory buffers that can absorb demand spikes. The simplicity of the reaction conditions also means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without extensive re-validation, ensuring continuity of supply across the global network.
- Scalability and Environmental Compliance: Scaling this reaction from the bench to the plant is straightforward due to the use of conventional equipment and non-hazardous reagents. The aqueous nature of the reaction medium reduces the fire hazard profile compared to purely organic solvent systems, simplifying safety protocols and insurance costs. From an environmental standpoint, the reduction in solvent waste and the avoidance of heavy metals align perfectly with modern green chemistry principles and increasingly stringent environmental regulations. This compliance reduces the burden of waste disposal and treatment, lowering the operational overhead associated with environmental health and safety (EHS) management. The ability to run the reaction at atmospheric pressure without specialized high-pressure reactors further simplifies the engineering requirements for scale-up, making it accessible to a wider range of manufacturing partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing production portfolios. For more specific technical data or custom synthesis requests, direct consultation with our technical team is recommended.
Q: What is the primary advantage of the Mannich reaction method described in CN1771254A?
A: The primary advantage is the ability to perform the coupling in a single step without requiring extensive protecting group strategies on non-reactive functional groups, significantly reducing process time and material costs.
Q: Can this synthesis method be scaled for commercial API production?
A: Yes, the use of common solvents like water and 1,4-dioxane, along with stable reagents like paraformaldehyde or aqueous formaldehyde, makes the process highly amenable to large-scale commercial manufacturing.
Q: What types of biological activities do these deazapurine compounds target?
A: These compounds act as potent inhibitors for enzymes such as Purine Nucleoside Phosphorylase (PNP) and 5'-methylthioadenosine phosphorylase (MTAP), targeting conditions like T-cell malignancies and parasitic infections.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Deazapurine Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in the development of next-generation therapeutics. Our team of expert chemists has extensively evaluated the Mannich reaction protocol described in CN1771254A and confirmed its viability for large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from clinical supply to commercial market is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for pharmaceutical intermediates. We are committed to delivering high-quality deazapurine derivatives that meet the exacting standards of the global pharmaceutical industry, supporting your mission to bring life-saving medicines to patients faster.
We invite you to collaborate with us to optimize your supply chain for these valuable enzyme inhibitors. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By leveraging our expertise in process optimization, we can help you identify further opportunities for efficiency gains and cost reduction. Please contact us today to request specific COA data and route feasibility assessments for your projects. Let us be your partner in turning innovative chemistry into commercial success.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
