Advanced Synthesis of 2-Benzyl-1,2,3,4-Tetrahydroisoquinoline-8-Carboxylic Acid for Commercial Scale-Up
Introduction to Patent CN103772278A and Tetrahydroisoquinoline Derivatives
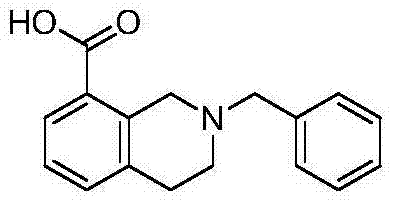
The pharmaceutical landscape is continuously evolving, driven by the demand for novel heterocyclic scaffolds that exhibit potent biological activities. Patent CN103772278A discloses a significant advancement in the synthesis of tetrahydroisoquinoline derivatives, specifically focusing on the production of 2-benzyl-1,2,3,4-tetrahydroisoquinoline-8-carboxylic acid. This compound serves as a critical building block for developing new drug candidates with antihypertensive, antiviral, and antioxidant properties. The disclosed method offers a robust pathway starting from readily available 8-bromoisoquinoline and benzyl bromide, proceeding through a sequence of quaternization, reduction, cyanation, and hydrolysis. For R&D directors and procurement specialists, understanding this specific synthetic route is vital as it represents a shift towards more accessible and scalable manufacturing processes for complex nitrogen-containing heterocycles.
The strategic importance of this intermediate lies in its versatility; the carboxylic acid moiety at the 8-position allows for further derivatization into amides, esters, or other functional groups essential for Structure-Activity Relationship (SAR) studies. By establishing a reliable supply chain for such precursors, pharmaceutical companies can accelerate their drug discovery timelines. This report analyzes the technical merits of the patented process, highlighting its potential for cost reduction in pharma intermediate manufacturing and its suitability for commercial scale-up of complex heterocyclic intermediates. The methodology described eliminates several bottlenecks associated with traditional isoquinoline functionalization, offering a streamlined approach that aligns with modern green chemistry principles and industrial efficiency standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of the isoquinoline ring system, particularly at the 8-position, has presented significant challenges due to the electronic deactivation of the benzene ring by the adjacent nitrogen atom. Conventional routes often rely on harsh electrophilic aromatic substitution conditions or multi-step sequences involving protecting groups that increase both material costs and waste generation. Furthermore, direct metal-catalyzed coupling reactions at the 8-position frequently suffer from low regioselectivity and require expensive palladium or rhodium catalysts, which pose contamination risks and necessitate rigorous purification steps to meet residual metal specifications. These factors collectively contribute to extended lead times and inflated production costs, creating a bottleneck for the rapid development of tetrahydroisoquinoline-based therapeutics.
The Novel Approach
The novel approach detailed in the patent circumvents these issues by employing a stepwise strategy that leverages the inherent reactivity of the isoquinoline nitrogen. By first forming a quaternary ammonium salt, the electron density of the ring system is modulated, facilitating subsequent reduction and functionalization steps under milder conditions. This method utilizes cost-effective reagents such as cuprous cyanide for the introduction of the nitrile group, avoiding the need for precious metals. The sequential nature of the synthesis allows for precise control over reaction parameters, minimizing the formation of by-products and simplifying downstream processing. This strategic redesign of the synthetic pathway not only enhances the overall yield but also significantly improves the operational safety and environmental profile of the manufacturing process.
Mechanistic Insights into the Four-Step Synthetic Sequence
The synthesis begins with a Menshutkin-type reaction where 8-bromoisoquinoline acts as a nucleophile attacking benzyl bromide. This quaternization step is crucial as it activates the heterocyclic ring for the subsequent reduction. The reaction is typically conducted in tetrahydrofuran (THF) under reflux, ensuring complete conversion to the onium salt. Following this, the onium salt undergoes selective reduction using sodium cyanoborohydride in dimethylformamide (DMF). Unlike stronger reducing agents that might reduce the aromatic ring indiscriminately, sodium cyanoborohydride selectively targets the iminium double bond generated in situ, preserving the aromatic integrity of the benzene ring while saturating the nitrogen-containing ring to form the tetrahydroisoquinoline skeleton.
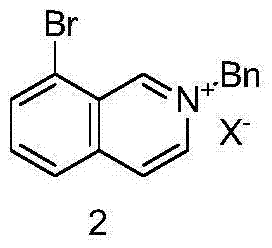
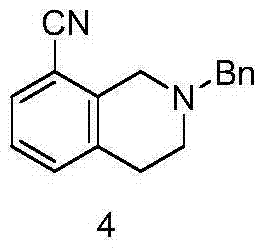
The third and perhaps most chemically significant step involves the nucleophilic substitution of the bromine atom at the 8-position with a cyano group. This transformation utilizes cuprous cyanide (CuCN) in DMF at elevated temperatures. This Rosenmund-von Braun type reaction is highly effective for introducing a versatile nitrile handle onto the aromatic core. The use of copper cyanide is advantageous from a cost perspective compared to palladium-catalyzed cyanation, and the reaction conditions are robust enough for industrial application. The resulting 2-benzyl-8-cyano-1,2,3,4-tetrahydroisoquinoline is a stable intermediate that can be isolated or carried forward directly, providing flexibility in the manufacturing workflow.
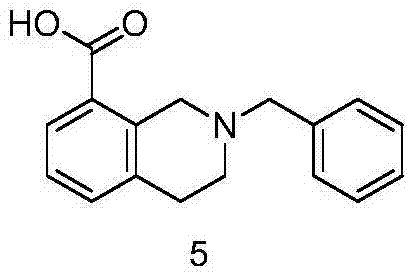
The final step entails the hydrolysis of the nitrile group to the corresponding carboxylic acid. This is achieved by heating the cyano-intermediate with sodium hydroxide in an ethanol-water mixture under reflux. The alkaline conditions facilitate the conversion of the nitrile to the carboxylate salt, which is subsequently acidified to precipitate the free acid. This hydrolysis step is clean and high-yielding, producing the target 2-benzyl-1,2,3,4-tetrahydroisoquinoline-8-carboxylic acid as a white solid. The simplicity of the workup, involving filtration and washing, ensures that the final product meets stringent purity specifications required for pharmaceutical applications without the need for complex chromatographic purification.
How to Synthesize 2-Benzyl-1,2,3,4-Tetrahydroisoquinoline-8-Carboxylic Acid Efficiently
Implementing this synthesis requires careful attention to solvent quality and temperature control, particularly during the reduction and cyanation steps. The process is designed to be telescoped where possible, although isolation of intermediates is recommended for quality control in a GMP environment. The use of common industrial solvents like THF, DMF, and ethanol simplifies solvent recovery and recycling, further enhancing the economic viability of the route. Operators should be trained on the handling of cuprous cyanide and sodium cyanoborohydride to ensure safety compliance. Detailed standard operating procedures (SOPs) regarding addition rates and exotherm management are essential for safe scale-up.
- Quaternization of 8-bromoisoquinoline with benzyl bromide in THF under reflux to form the onium salt.
- Selective reduction of the onium salt using sodium cyanoborohydride in DMF at room temperature.
- Nucleophilic substitution of the bromo group with cuprous cyanide in DMF to introduce the cyano group.
- Alkaline hydrolysis of the nitrile group using sodium hydroxide to yield the final carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers substantial advantages by relying on commodity chemicals that are widely available in the global market. The starting materials, 8-bromoisoquinoline and benzyl bromide, are produced at scale by numerous suppliers, mitigating the risk of supply chain disruptions. Furthermore, the elimination of precious metal catalysts such as palladium removes a significant cost driver and reduces the complexity of metal scavenging processes, which are often required to meet regulatory limits for heavy metals in APIs. This translates directly into cost reduction in pharma intermediate manufacturing, allowing for more competitive pricing of the final active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with copper salts significantly lowers the raw material costs. Additionally, the avoidance of cryogenic conditions or high-pressure hydrogenation equipment reduces capital expenditure (CAPEX) and operational expenditure (OPEX). The process utilizes standard reflux conditions which are energy-efficient and easy to maintain in existing reactor infrastructure. By simplifying the purification steps to crystallization and filtration, the consumption of silica gel and chromatography solvents is minimized, leading to lower waste disposal costs and a reduced environmental footprint.
- Enhanced Supply Chain Reliability: The reliance on robust, non-proprietary reagents ensures that the supply chain is resilient against geopolitical or logistical shocks. Since the reagents are not single-source dependent, procurement managers can negotiate better terms and secure multiple supply lines. The stability of the intermediates allows for batch production and inventory stocking, enabling manufacturers to respond quickly to fluctuating demand from downstream API producers. This reliability is crucial for maintaining continuous production schedules and reducing lead time for high-purity API building blocks.
- Scalability and Environmental Compliance: The reaction conditions are inherently scalable, moving seamlessly from kilogram to tonne scale without significant re-optimization. The use of aqueous workups and the ability to recycle solvents like ethanol and THF align with green chemistry initiatives. The waste stream primarily consists of inorganic salts and organic solvents that can be treated using standard wastewater treatment protocols, ensuring compliance with increasingly strict environmental regulations. This scalability supports the commercial scale-up of complex heterocyclic intermediates, meeting the growing demand for tetrahydroisoquinoline derivatives in the pharmaceutical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of this tetrahydroisoquinoline derivative. These answers are derived from the technical data provided in the patent literature and general industry knowledge regarding heterocyclic synthesis. Understanding these aspects helps stakeholders make informed decisions about integrating this intermediate into their supply chains.
Q: What are the key advantages of this synthesis route over traditional methods?
A: This route avoids the use of expensive precious metal catalysts often required for direct functionalization, utilizing cost-effective copper cyanide instead. It also employs mild reduction conditions that minimize side reactions, leading to a cleaner impurity profile.
Q: Is this intermediate suitable for large-scale API production?
A: Yes, the process utilizes common solvents like THF and DMF and operates at standard reflux temperatures, making it highly amenable to scale-up in standard stainless steel reactors without requiring specialized high-pressure equipment.
Q: How is the purity of the final carboxylic acid ensured?
A: The final step involves precipitation by pH adjustment followed by rigorous washing with water, methanol, and acetone, which effectively removes inorganic salts and organic impurities, ensuring high purity suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Benzyl-1,2,3,4-Tetrahydroisoquinoline-8-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the drug development pipeline. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-benzyl-1,2,3,4-tetrahydroisoquinoline-8-carboxylic acid meets the highest industry standards. Our commitment to quality and consistency makes us a trusted partner for pharmaceutical companies seeking to optimize their synthesis routes.
We invite you to collaborate with us to explore how this advanced synthetic route can benefit your specific projects. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your supply chain efficiency and reduce your overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
