Scalable Synthesis of 9-Anthrone Lactone Frameworks for Antitumor Drug Development
Scalable Synthesis of 9-Anthrone Lactone Frameworks for Antitumor Drug Development
The pharmaceutical industry is constantly seeking robust synthetic routes for complex marine natural product scaffolds that exhibit potent biological activity. Patent CN109096239B introduces a groundbreaking preparation route for the 9-anthrone lactone framework, a rigid tetracyclic structure found in the marine microorganism secondary metabolite Aspergillus A (AspA). This patent details the first successful chemical construction of this specific skeleton, overcoming the historical reliance on difficult natural extraction methods. By establishing a concise two-step synthesis involving high-temperature condensation and acid-mediated cyclization, this technology provides a reliable foundation for developing novel antitumor agents. For R&D directors and procurement specialists, this represents a significant shift from unpredictable biological sourcing to controlled, high-yield chemical manufacturing, ensuring a steady supply of high-purity intermediates for drug discovery programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, obtaining 9-anthrone lactone compounds like Aspergillus A relied heavily on the isolation from marine filamentous fungi, specifically strains collected from mangrove forest root mud. This biological extraction approach suffers from inherent limitations that severely hinder commercial viability and research scalability. The natural abundance of these compounds is extremely low, leading to poor yields that cannot meet the demands of preclinical or clinical development. Furthermore, the native structure of AspA contains polyphenol hydroxyl substitutions that render the molecule chemically unstable, prone to easy oxidation, and characterized by large polarity and poor solubility. These physicochemical properties complicate formulation and lead to rapid metabolic elimination in vivo, resulting in a short half-life that limits therapeutic efficacy. Consequently, relying on natural sources creates a bottleneck in the supply chain, making cost reduction in API manufacturing nearly impossible due to the intensive resources required for fermentation and purification.
The Novel Approach
The innovative synthetic strategy outlined in the patent circumvents these biological constraints by constructing the 9-anthrone lactone framework entirely through organic synthesis. This method utilizes readily available benzophenone compounds and substituted ethyl benzoylacetates as starting materials, which are condensed under the catalysis of DBU at high temperatures to form key coumarin intermediates. Subsequently, the rigid tetracyclic framework is assembled via intramolecular dehydration cyclization using Eaton's Reagent. This chemical approach not only achieves high yields but also allows for structural simplification, producing derivatives (Compounds I and II) that retain antitumor activity while exhibiting superior chemical stability compared to the natural product. By shifting to this synthetic paradigm, manufacturers can achieve consistent quality and significantly reduced lead times for high-purity pharmaceutical intermediates, effectively decoupling production from the vagaries of marine biology.
Mechanistic Insights into DBU-Catalyzed Condensation and Cyclization
The core of this synthetic breakthrough lies in the efficient formation of the carbon-carbon and carbon-oxygen bonds that define the tetracyclic skeleton. The process initiates with a base-catalyzed condensation where DBU acts as a non-nucleophilic base to deprotonate the active methylene group of the ethyl benzoylacetate. At an elevated temperature of 160°C, this nucleophile attacks the carbonyl carbon of the benzophenone derivative, facilitating a Claisen-type condensation that constructs the initial coumarin ring system. This step is critical as it sets the stereochemical and regiochemical stage for the subsequent ring closure. The use of DBU is particularly advantageous as it minimizes side reactions often associated with stronger, nucleophilic bases, thereby preserving the integrity of the sensitive functional groups on the aromatic rings and ensuring a clean reaction profile that simplifies downstream purification.
Following the formation of the acyclic or monocyclic intermediate, the construction of the rigid anthrone core is achieved through an acid-mediated cyclization. Eaton's Reagent, a solution of phosphorus pentoxide in methanesulfonic acid, serves as a powerful dehydrating agent that promotes the intramolecular Friedel-Crafts acylation or similar electrophilic aromatic substitution. This step effectively closes the final ring, locking the molecule into its planar, rigid tetracyclic conformation. The reaction proceeds with high selectivity at moderate temperatures (50°C), preventing thermal degradation of the product. In the case of Compound I, a subsequent demethylation step using boron trifluoride diethyl etherate at 80°C selectively removes a methyl protecting group to reveal the phenolic hydroxyl, fine-tuning the electronic properties of the final molecule. This precise control over functional group transformation is essential for optimizing the biological activity and solubility profile of the final drug candidate.
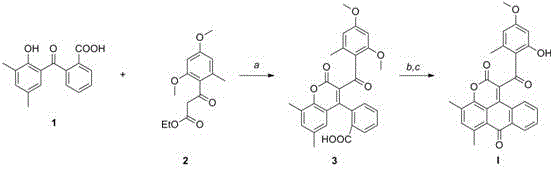
The visual representation of the synthesis pathway for Compound I clearly illustrates the efficiency of this two-step sequence. Starting from the benzophenone precursor and the ethyl ester, the reaction flows smoothly through the intermediate stage to the final cyclized product. The structural evolution from a flexible linear precursor to a rigid fused ring system is evident, highlighting how the synthetic design mimics the complexity of nature while utilizing robust industrial reagents. This mechanistic clarity provides confidence to process chemists that the route is reproducible and amenable to optimization, a key factor when considering the commercial scale-up of complex pharmaceutical intermediates.
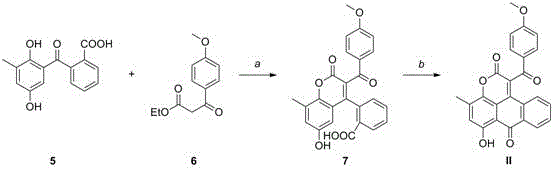
Similarly, the synthesis of Compound II demonstrates the versatility of this platform technology. By varying the substitution pattern on the starting benzophenone and ester components, the same fundamental reaction conditions can be applied to generate diverse analogues. This modularity is invaluable for medicinal chemistry campaigns where structure-activity relationship (SAR) studies require a library of related compounds. The ability to access these structurally complex molecules through a unified synthetic strategy significantly accelerates the drug discovery timeline, allowing R&D teams to focus on biological evaluation rather than struggling with bespoke synthesis for each new analogue.
How to Synthesize 9-Anthrone Lactone Compounds Efficiently
The practical execution of this synthesis is designed to be straightforward, utilizing standard laboratory equipment and reagents that are commercially available in bulk quantities. The process begins with the precise weighing of the benzophenone and ethyl benzoylacetate precursors, which are mixed with a catalytic amount of DBU. The mixture is heated to 160°C with stirring for approximately 4 hours to ensure complete conversion to the coumarin intermediate. Following the reaction, the mixture is quenched with dilute hydrochloric acid and extracted with ethyl acetate. The crude product is then purified via silica gel column chromatography using a dichloromethane and methanol solvent system to isolate the intermediate in high purity. Detailed standardized synthesis steps see the guide below.
- Condense benzophenone compounds with substituted ethyl benzoylacetate at 160°C using DBU as a catalyst to form the coumarin intermediate.
- Perform intramolecular dehydration cyclization on the intermediate using Eaton's Reagent at 50°C to construct the tetracyclic skeleton.
- Optionally remove methyl protection groups using BF3·Et2O at 80°C to obtain the final hydroxyl-substituted target compounds.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from natural extraction to this synthetic route offers profound strategic benefits that directly impact the bottom line and operational reliability. The primary advantage lies in the drastic simplification of the supply chain; instead of depending on the seasonal and geographical variability of marine fungal collection, manufacturers can source stable, commodity-grade chemical raw materials year-round. This shift eliminates the risk of supply disruption caused by environmental factors or regulatory changes regarding marine resource harvesting. Furthermore, the synthetic route's high yield and selectivity mean that less raw material is wasted, leading to substantial cost savings in material procurement and waste disposal. The ability to produce these intermediates on demand ensures that project timelines are met without the long lead times typically associated with isolating trace natural products.
- Cost Reduction in Manufacturing: The synthetic pathway eliminates the need for expensive and labor-intensive fermentation processes and the subsequent complex extraction protocols required to isolate minute quantities of natural AspA. By utilizing cost-effective reagents like DBU and Eaton's Reagent, and achieving high conversion rates, the overall cost of goods sold (COGS) is significantly lowered. Additionally, the simplified purification steps reduce the consumption of chromatography media and solvents, further driving down operational expenses. This economic efficiency makes the development of 9-anthrone lactone-based drugs financially viable, opening up market opportunities that were previously closed due to prohibitive production costs.
- Enhanced Supply Chain Reliability: Relying on a chemical synthesis route guarantees a consistent and predictable supply of the active intermediate. Unlike natural products, where batch-to-batch variability in potency and impurity profiles is a common challenge, this synthetic method produces a well-defined chemical entity with a consistent impurity spectrum. This consistency is crucial for regulatory compliance and quality control, reducing the risk of batch failures during drug manufacturing. The use of stable starting materials ensures that inventory can be managed effectively, allowing for just-in-time production strategies that minimize capital tied up in stock while maximizing responsiveness to market demand.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are inherently scalable, moving seamlessly from gram-scale laboratory experiments to kilogram and ton-scale production. The absence of exotic catalysts or extreme pressure conditions simplifies the engineering requirements for reactor design. Moreover, the process generates fewer hazardous byproducts compared to traditional multi-step total syntheses of natural products. The streamlined workflow reduces the volume of chemical waste, aligning with modern green chemistry principles and easing the burden on environmental treatment facilities. This environmental compatibility is increasingly important for maintaining a social license to operate and meeting the stringent sustainability goals of global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these 9-anthrone lactone derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this technology into your existing drug development pipeline.
Q: What are the stability advantages of the synthesized 9-anthrone lactone derivatives compared to natural AspA?
A: The synthesized derivatives feature a simplified structure that eliminates unstable polyphenol hydroxyl substitutions found in natural Aspergillus A. This modification significantly reduces susceptibility to oxidation and metabolic elimination, thereby extending the half-life and improving chemical stability while maintaining antitumor activity.
Q: How does the DBU-catalyzed condensation step impact the overall yield?
A: The use of DBU as a catalyst at elevated temperatures (160°C) facilitates an efficient condensation between the benzophenone and ethyl benzoylacetate precursors. This method avoids harsh conditions that typically degrade sensitive intermediates, resulting in high yields (e.g., 62% for intermediate 3) and simplifying the purification process.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the route is designed for scalability. It utilizes readily available raw materials and standard reagents like Eaton's Reagent and DBU. The process involves straightforward workup procedures such as extraction and silica gel chromatography, making it highly adaptable for industrial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Anthrone Lactone Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the 9-anthrone lactone framework in the field of oncology. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory methodology into a robust industrial process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest international standards, providing a solid foundation for your clinical trials and eventual market launch.
We invite you to collaborate with us to unlock the full commercial potential of these antitumor agents. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can accelerate your path to a novel cancer therapy.
