Advanced Metal-Free Synthesis of Benzoxazole Derivatives for Commercial Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and economically viable synthetic routes for heterocyclic compounds, particularly benzoxazole derivatives which serve as critical scaffolds in numerous bioactive molecules. Patent CN108484517B introduces a groundbreaking methodology that addresses long-standing challenges in the construction of the benzoxazole core. This innovation leverages a metal-free oxidative cyclization strategy, utilizing N-hydroxyphthalimide (NHPI) as a potent organocatalyst to facilitate the coupling of beta-hydroxylamines and o-aminophenols. By shifting away from traditional transition metal catalysis, this technology offers a cleaner, more sustainable pathway that aligns perfectly with modern green chemistry principles and stringent regulatory requirements for residual metals in active pharmaceutical ingredients. The ability to utilize molecular oxygen from ambient air as the terminal oxidant further simplifies the operational complexity, removing the need for specialized high-pressure oxygen equipment or hazardous stoichiometric oxidants. For a reliable pharmaceutical intermediates supplier, adopting such a streamlined process translates directly into enhanced supply chain stability and reduced production costs, making it an attractive option for the commercial manufacture of high-value drug candidates.
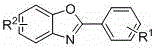
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzoxazole frameworks has relied heavily on transition metal catalysts such as copper, palladium, or iron, often requiring harsh reaction conditions and expensive ligands to achieve acceptable yields. These conventional pathways frequently suffer from significant drawbacks, including the generation of toxic heavy metal waste which necessitates complex and costly purification steps to meet pharmacopeial standards for residual metals. Furthermore, many traditional methods employ stoichiometric oxidants that are not only hazardous to handle on a large scale but also produce substantial amounts of chemical waste, thereby increasing the environmental footprint and disposal costs associated with cost reduction in heterocyclic compound manufacturing. The sensitivity of these metal-catalyzed reactions to moisture and oxygen often demands inert atmosphere conditions, adding another layer of operational expense and technical difficulty that can hinder the commercial scale-up of complex fine chemicals. Additionally, the limited substrate scope of some older methodologies restricts the diversity of derivatives that can be efficiently accessed, forcing process chemists to develop custom solutions for each new analog.
The Novel Approach
In stark contrast, the methodology disclosed in CN108484517B represents a paradigm shift by employing an organocatalytic system that operates efficiently under aerobic conditions without any transition metals. This novel approach utilizes beta-hydroxylamines and o-aminophenols as readily available starting materials, which are converted into the target benzoxazole structure through a seamless oxidative cyclization mediated by NHPI. The reaction conditions are notably mild, typically proceeding in common organic solvents like toluene or N-methyl pyrrolidone at temperatures ranging from 90°C to 140°C, which are easily achievable in standard industrial reactors. By using air as the oxidant, the process inherently improves atom economy and safety, eliminating the risks associated with storing and handling pure oxygen or aggressive chemical oxidants. This simplicity not only accelerates the development timeline for new drug candidates but also ensures that the resulting high-purity benzoxazole derivatives are free from metal contamination, significantly reducing the burden on downstream purification teams and quality control laboratories.
Mechanistic Insights into NHPI-Catalyzed Oxidative Cyclization
The mechanistic elegance of this transformation lies in the unique ability of N-hydroxyphthalimide to generate phthalimide-N-oxyl (PINO) radicals in the presence of molecular oxygen, initiating a cascade of single-electron transfer events. The process begins with the activation of the beta-hydroxylamine substrate, where the PINO radical abstracts a hydrogen atom to form an intermediate radical species that undergoes oxidation to an aldehyde functionality via decarboxylation and deamination. This generated aldehyde then condenses spontaneously with the o-aminophenol to form an imine intermediate, which is subsequently oxidized by the catalytic system to close the oxazole ring and restore aromaticity. Understanding this radical-mediated pathway is crucial for process optimization, as it highlights the importance of maintaining adequate oxygen flow and controlling the concentration of the NHPI catalyst to maximize turnover frequency. The absence of metal coordination complexes simplifies the kinetic profile of the reaction, allowing for more predictable scale-up behavior and easier troubleshooting during technology transfer to manufacturing sites.
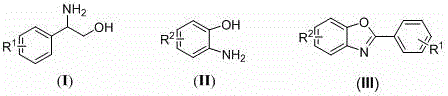
From an impurity control perspective, this metal-free mechanism offers distinct advantages by avoiding the formation of metal-complexed byproducts that are notoriously difficult to separate from the final product. The primary side reactions are typically limited to over-oxidation or polymerization of the reactive intermediates, which can be effectively managed by optimizing the reaction temperature and the molar ratio of the starting materials. The patent data suggests that maintaining a specific stoichiometry between the beta-hydroxylamine and the o-aminophenol, along with a controlled loading of the NHPI catalyst, ensures high selectivity for the desired benzoxazole core. This level of control is essential for producing high-purity API intermediates that meet the rigorous specifications required by global regulatory agencies, thereby minimizing the risk of batch rejection and ensuring consistent product quality across multiple production campaigns.
How to Synthesize Benzoxazole Derivatives Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the dispersion of reagents and the management of the gas-liquid interface to ensure efficient oxygen uptake. The general procedure involves dispersing the beta-hydroxylamine and o-aminophenol substrates along with the NHPI catalyst in a selected solvent, followed by heating the mixture under an air atmosphere with vigorous stirring. Detailed standard operating procedures regarding specific molar ratios, solvent choices, and workup protocols are critical for reproducibility and are outlined in the technical documentation below. Process engineers should note that while the reaction is robust, the choice of solvent can influence the reaction rate and ease of product isolation, with non-polar solvents like toluene often facilitating simpler crystallization processes compared to polar aprotic solvents.
- Disperse beta-hydroxylamine, o-aminophenol, and N-hydroxyphthalimide (NHPI) catalyst in a suitable organic solvent such as toluene or NMP.
- Heat the reaction mixture to a temperature range of 90°C to 140°C under an air atmosphere with continuous stirring.
- Maintain reflux for 5 to 20 hours to complete the oxidative cyclization, followed by purification via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this NHPI-catalyzed technology presents a compelling value proposition driven by significant operational efficiencies and cost avoidance strategies. The elimination of transition metal catalysts removes a major cost center associated with both the purchase of expensive precious metals and the implementation of specialized scavenging resins or extraction processes required to reduce metal levels to parts-per-million specifications. This simplification of the downstream processing workflow directly contributes to cost reduction in heterocyclic compound manufacturing by shortening the overall production cycle time and reducing the consumption of auxiliary materials. Furthermore, the reliance on air as an oxidant means that the process does not depend on the supply chain volatility of specialized chemical oxidants, enhancing the resilience of the manufacturing schedule against external market disruptions.
- Cost Reduction in Manufacturing: The economic benefits of this process are substantial, primarily derived from the removal of costly metal catalysts and the associated purification infrastructure. By utilizing an organocatalyst that is inexpensive and readily available in bulk quantities, manufacturers can achieve a lower cost of goods sold without compromising on the quality or purity of the final intermediate. The simplified workup procedure, which often avoids complex aqueous extractions required to remove metal salts, further reduces utility consumption and waste treatment costs, leading to a more sustainable and profitable production model.
- Enhanced Supply Chain Reliability: The use of commodity chemicals such as beta-hydroxylamines and o-aminophenols ensures a stable supply of raw materials, as these are produced by multiple vendors globally and are not subject to the geopolitical constraints often seen with rare earth metals. This diversification of the supply base significantly mitigates the risk of production stoppages due to raw material shortages, ensuring reducing lead time for high-purity API intermediates and allowing for more accurate delivery commitments to downstream pharmaceutical clients. The robustness of the reaction conditions also means that the process can be transferred between different manufacturing sites with minimal re-validation effort.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this methodology aligns perfectly with green chemistry initiatives by minimizing hazardous waste generation and energy consumption. The absence of heavy metals simplifies the disposal of process waste streams, reducing the regulatory burden and liability associated with hazardous waste management. The mild reaction temperatures and atmospheric pressure operation make the process inherently safer to scale up to multi-ton quantities, facilitating the commercial scale-up of complex fine chemicals while maintaining a low environmental footprint that appeals to sustainability-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzoxazole synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for project managers evaluating the feasibility of integrating this route into their existing production portfolios.
Q: Does this synthesis method require expensive transition metal catalysts?
A: No, the patented method utilizes N-hydroxyphthalimide (NHPI) as an organocatalyst and molecular oxygen from air as the oxidant, completely eliminating the need for costly transition metals and subsequent heavy metal removal steps.
Q: What are the typical reaction conditions for this benzoxazole synthesis?
A: The reaction typically proceeds in solvents like toluene or NMP at temperatures between 90°C and 140°C for a duration of 5 to 20 hours, depending on the specific substrate reactivity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of air as an oxidant and readily available starting materials makes this process highly scalable and economically viable for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoxazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic technologies to maintain a competitive edge in the global pharmaceutical market. Our team of expert process chemists has extensively evaluated the NHPI-catalyzed pathway described in CN108484517B and confirmed its potential for robust, large-scale production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of detecting impurities at trace levels, guaranteeing that every batch of benzoxazole intermediate we deliver meets stringent purity specifications required for GMP manufacturing.
We invite you to collaborate with us to leverage this advanced chemistry for your next drug development program. By partnering with our technical team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments that demonstrate how we can optimize your supply chain for benzoxazole derivatives.
