Advanced Biocatalytic Synthesis of Dextro-Cis-Chrysanthemic Acid for Commercial Pyrethroid Production
The pharmaceutical and agrochemical industries are constantly seeking more efficient pathways to produce chiral intermediates with high optical purity. Patent CN112301065A discloses a groundbreaking preparation method for dextro-cis-first chrysanthemic acid, a critical building block for synthetic pyrethroid sanitary pesticides. This innovation leverages a biocatalytic resolution strategy, utilizing specific esterase enzymes to selectively hydrolyze racemic first chrysanthemic acid esters in an aqueous environment. Unlike traditional methods that struggle with low enantiomeric excess, this novel approach consistently delivers a final product with an ee value exceeding 99%. By bypassing the cumbersome crystallization and purification steps typically associated with low-ee intermediates, this technology offers a streamlined, cost-effective, and environmentally superior route for manufacturing high-value agrochemical precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of optically pure chrysanthemic acid derivatives has relied heavily on chemical resolution techniques, which are fraught with significant inefficiencies and environmental drawbacks. Traditional chemical resolving agents are becoming increasingly scarce and expensive, driving up the raw material costs for manufacturers. Furthermore, these chemical methods often yield intermediates with suboptimal ee values, necessitating multiple rounds of recrystallization and physical purification to achieve the requisite stereochemical purity for pyrethroid synthesis. These additional processing steps not only extend the production cycle and increase energy consumption but also generate substantial amounts of chemical waste, posing challenges for waste treatment and regulatory compliance in modern chemical plants.
The Novel Approach
The patented biocatalytic method represents a paradigm shift by employing engineered esterase enzymes to achieve highly selective hydrolysis under mild conditions. By selecting the appropriate esterase, such as the CEYN series commercialized by Sigma, the process effectively discriminates between enantiomers in the racemic ester mixture. This biological specificity allows for the direct production of dextro-cis-first chrysanthemic acid with an ee value greater than 99% in a single hydrolysis step. The elimination of downstream purification stages significantly simplifies the overall production workflow, reducing both the operational complexity and the total time required to bring the intermediate to market readiness for pyrethroid formulation.
Mechanistic Insights into Esterase-Catalyzed Selective Hydrolysis
The core of this technological advancement lies in the precise stereoselectivity of the biocatalyst within a carefully controlled aqueous buffer system. The reaction mechanism involves the esterase enzyme recognizing and binding specifically to the cis-configuration of the chrysanthemic acid ester substrate. Upon binding, the enzyme catalyzes the hydrolysis of the ester bond, releasing the free carboxylic acid (dextro-cis-first chrysanthemic acid) into the aqueous phase while leaving the trans-isomer or the opposite enantiomer largely intact in the organic phase or as unreacted ester. This kinetic resolution is highly dependent on the microenvironment provided by the buffer system, which can include ammonia water, organic amines, or inorganic bases like sodium carbonate.
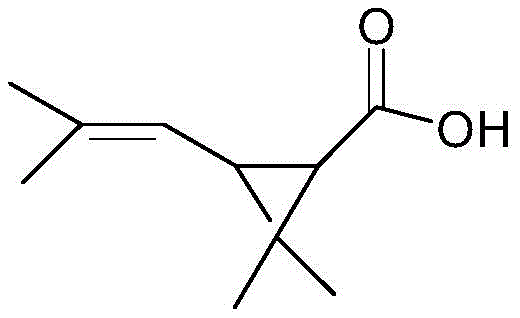
To maintain optimal enzyme activity and stability, the reaction parameters are tightly regulated, with the pH maintained between 4 and 8 through the addition of sodium hydroxide during the process. The use of an aqueous medium not only supports the biological function of the esterase but also facilitates the separation of the product, as the resulting acid salt is water-soluble. Following the reaction, simple acidification allows for the extraction of the pure acid product. This mechanism ensures that impurities are minimized at the source, as the enzyme's inherent selectivity prevents the formation of unwanted byproducts that are common in non-specific chemical hydrolysis reactions.
How to Synthesize Dextro-Cis-Chrysanthemic Acid Efficiently
The synthesis protocol outlined in the patent provides a robust framework for scaling this biocatalytic process from laboratory to industrial levels. The procedure begins with the preparation of a reaction mixture containing the racemic ester substrate, water, and the specific esterase catalyst. The system is then agitated at a controlled temperature ranging from 20 to 60°C for a duration of 6 to 60 hours, depending on the specific enzyme loading and substrate concentration. Throughout the reaction, the pH is monitored and adjusted to ensure the enzyme remains in its active state, facilitating the continuous conversion of the ester to the desired acid. For a detailed breakdown of the standardized operating procedures and specific reagent quantities, please refer to the technical guide below.
- Prepare an aqueous reaction system containing racemic first chrysanthemic acid ester (such as methyl or ethyl ester) and a specific biocatalyst esterase (e.g., CEYN205).
- Maintain the reaction temperature between 20-60°C and adjust pH to 4-8 using sodium hydroxide while stirring for 6-60 hours to allow selective hydrolysis.
- Separate the oil-water layers, recover the enzyme, acidify the aqueous layer to pH 2, and extract with toluene to isolate the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this biocatalytic technology translates into tangible strategic benefits regarding cost structure and supply reliability. The shift from chemical resolving agents to reusable enzymes fundamentally alters the cost equation by removing the dependency on volatile and expensive chiral auxiliaries. Additionally, the simplified workflow reduces the demand for extensive solvent usage and energy-intensive purification equipment, leading to a leaner manufacturing footprint. These operational efficiencies contribute to a more resilient supply chain capable of meeting the rigorous demands of the global agrochemical market without the bottlenecks associated with traditional multi-step resolution processes.
- Cost Reduction in Manufacturing: The implementation of this enzymatic route drives down production costs through several key mechanisms, primarily by eliminating the need for costly chemical resolving agents that are difficult to source. The ability to recover and reuse the biocatalyst through centrifugation or membrane filtration further amortizes the cost of the enzyme over multiple batches, significantly lowering the variable cost per kilogram of product. Moreover, the reduction in processing steps means less consumption of utilities such as steam and electricity, as well as a decrease in solvent waste disposal fees, resulting in substantial overall cost savings for the manufacturing facility.
- Enhanced Supply Chain Reliability: Supply continuity is greatly improved because the process is not reliant on the fluctuating availability of specialized chemical resolving agents, which have historically been a supply chain vulnerability. The raw materials required, such as various chrysanthemic acid esters (methyl, ethyl, etc.), are commodity chemicals with stable supply lines, ensuring that production can proceed without interruption. The flexibility of the process to accept feedstocks with varying cis-trans ratios also means that manufacturers are not forced to wait for highly fractionated raw materials, allowing for faster inventory turnover and more responsive production scheduling.
- Scalability and Environmental Compliance: Scaling this process to commercial volumes is straightforward due to the use of standard aqueous reaction conditions that do not require exotic high-pressure or cryogenic equipment. The environmental profile of the process is exceptionally clean, as it avoids the generation of heavy metal waste or toxic organic byproducts often associated with chemical resolution. This alignment with green chemistry principles simplifies regulatory compliance and environmental permitting, reducing the risk of production shutdowns due to environmental violations and enhancing the corporate sustainability profile of the manufacturer.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this biocatalytic resolution technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on the feasibility and advantages of adopting this method for large-scale production. Understanding these details is crucial for technical teams evaluating the integration of this process into existing manufacturing lines.
Q: What is the optical purity (ee value) achieved by this biocatalytic method?
A: The patented biocatalytic resolution method achieves an exceptional enantiomeric excess (ee) value of greater than 99% for dextro-cis-first chrysanthemic acid. This high level of stereochemical purity eliminates the need for subsequent crystallization or purification steps often required in traditional chemical resolution processes, allowing the intermediate to be directly used in the synthesis of sanitary pyrethroids.
Q: Can this process handle raw materials with varying cis-trans ratios?
A: Yes, the process demonstrates significant flexibility regarding the raw material composition. The enzymatic system selectively hydrolyzes the cis-configuration ester while leaving the trans-configuration ester largely unreacted. This means the initial cis-trans ratio of the chrysanthemic acid ester feedstock is not strictly limited, although a higher cis-isomer content is preferred for industrial efficiency. The unreacted trans-ester can be separated and recovered, enhancing overall atom economy.
Q: What are the environmental advantages of using esterase over chemical resolving agents?
A: This method replaces scarce and polluting chemical resolving agents with a biocatalyst (esterase) in an aqueous medium. The enzyme can be recovered and reused via centrifugation or ultrafiltration, significantly reducing waste generation. Furthermore, the reaction operates under mild conditions (20-60°C) without harsh organic solvents during the hydrolysis phase, aligning with green chemistry principles and reducing the environmental footprint of agrochemical intermediate manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dextro-Cis-Chrysanthemic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this biocatalytic technology for the agrochemical sector. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that verify every batch meets the highest standards for optical purity and chemical integrity required by top-tier pyrethroid manufacturers globally.
We invite you to collaborate with us to leverage these advanced synthesis capabilities for your supply chain. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us demonstrate how we can optimize your sourcing strategy for high-purity chrysanthemic acid derivatives.
