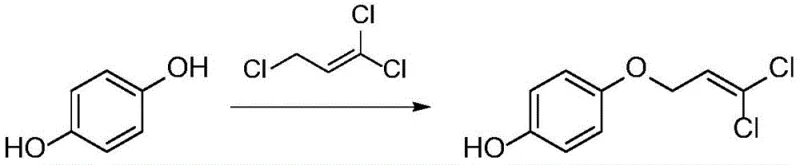
Advanced Synthesis of 4-(3,3-dichloroallyloxy)phenol for Commercial Agrochemical Production
The chemical landscape for agrochemical intermediate production is undergoing a significant transformation, driven by the urgent need for more sustainable and selective synthetic routes. Patent CN111574337B, published in late 2022, introduces a groundbreaking methodology for the synthesis of 4-(3,3-dichloroallyloxy)phenol, a critical precursor in the manufacturing of the novel pesticide pyridalyl. This compound is essential for controlling lepidoptera and thysanoptera pests while maintaining low toxicity profiles for mammals and birds. The disclosed technology represents a paradigm shift from traditional etherification methods, offering a robust solution that addresses long-standing challenges in selectivity and environmental compliance. For R&D directors and procurement specialists, understanding the nuances of this patent is vital for securing a reliable agrochemical intermediate supplier capable of delivering high-purity materials consistently. The innovation lies not just in the chemical transformation but in the holistic process design that minimizes waste and maximizes yield through a sophisticated water-oil two-phase system.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-(3,3-dichloroallyloxy)phenol has been plagued by significant inefficiencies that hinder cost reduction in agrochemical manufacturing. Traditional literature methods often rely on the use of high-boiling polar solvents such as N,N-dimethylformamide (DMF) under inorganic alkali conditions. A major drawback of these conventional routes is the poor selectivity, which frequently results in the formation of substantial amounts of double-etherification by-products. To mitigate this, prior art suggests using a large excess of hydroquinone, with molar ratios exceeding 3.0:1.0 relative to the alkylating agent. While this improves selectivity marginally, it drastically inflates raw material costs and creates a massive burden on downstream recovery processes. Furthermore, the use of DMF complicates the isolation of the product and generates hazardous organic wastewater, posing severe environmental compliance risks. Alternative strategies involving protection and deprotection of phenolic hydroxyl groups add unnecessary synthetic steps, reducing overall atom economy and extending production cycles, which is detrimental to supply chain reliability.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a streamlined etherification reaction that eliminates the need for protecting groups and hazardous polar solvents. By employing hydroquinone and 1,3-trichloropropene as primary raw materials in the presence of alkali liquor and sodium dodecyl benzene sulfonate, the process achieves exceptional mono-substitution selectivity. The core innovation is the implementation of a water-oil two-phase system, which fundamentally alters the reaction kinetics to favor the desired mono-ether product. This method not only simplifies the reaction conditions but also facilitates easier product separation and solvent recovery. The avoidance of high-boiling solvents means that energy consumption is significantly reduced, and the generation of organic wastewater is effectively avoided. For supply chain heads, this translates to a more resilient production model with reduced lead time for high-purity agrochemical intermediates, as the simplified workup allows for faster turnaround from reactor to finished goods without compromising on quality or safety standards.
Mechanistic Insights into Phase Transfer Etherification
The success of this synthesis hinges on the precise mechanistic role of sodium dodecyl benzene sulfonate (SDBS) within the reaction matrix. SDBS acts as a potent surfactant and phase transfer catalyst, facilitating the interaction between the aqueous phenolate salt and the organic soluble 1,3-trichloropropene. In the absence of this auxiliary agent, the reaction would suffer from mass transfer limitations inherent to biphasic systems. The surfactant molecules form micelles that solubilize the organic reactants within the aqueous phase or at the interface, thereby increasing the effective concentration of reactive species. This microenvironment promotes the nucleophilic attack of the phenolate anion on the allylic chloride, ensuring that the reaction proceeds efficiently at mild temperatures ranging from 35°C to 50°C. The controlled addition of alkali liquor ensures the gradual formation of the sodium phenolate, preventing local overheating and minimizing side reactions such as hydrolysis of the trichloropropene. This delicate balance is crucial for maintaining the structural integrity of the dichloroallyl group, which is essential for the biological activity of the final pesticide.
Impurity control is another critical aspect where this mechanism excels, directly impacting the purity profile required by R&D directors. The two-phase system inherently suppresses the formation of di-ether by-products, which are common in homogeneous polar solvent systems. The selectivity is reported to reach between 71% and 85%, a substantial improvement over conventional methods. The reaction conditions are optimized to ensure that the mono-substituted product precipitates or partitions favorably, allowing for straightforward isolation. Acidification with hydrochloric acid in the final step converts the remaining phenolate salts back to phenol, which can be separated, while the product remains in the organic phase or precipitates as a solid depending on the specific solvent choice like toluene or n-heptane. This rigorous control over the reaction pathway ensures that the impurity spectrum is manageable, reducing the need for complex chromatographic purification and making the process viable for commercial scale-up of complex agrochemical intermediates.
How to Synthesize 4-(3,3-dichloroallyloxy)phenol Efficiently
Implementing this synthesis route requires careful attention to the preparation of the phenol sodium salt and the subsequent etherification steps to ensure optimal yield and safety. The process begins with the suspension of hydroquinone in an aqueous solution containing sodium dodecyl benzene sulfonate, followed by the controlled addition of sodium hydroxide to generate the reactive phenolate species. Temperature control during this exothermic neutralization is paramount to prevent degradation of the reactants. Once the phenolate is formed, the 1,3-trichloropropene, dissolved in a suitable organic solvent such as toluene, is added dropwise to maintain a steady reaction rate. The detailed standardized synthetic steps, including specific molar ratios, stirring speeds, and precise temperature profiles for each stage of the reaction, are outlined in the guide below for technical teams to follow.
- Prepare sodium phenolate by reacting hydroquinone with sodium hydroxide and sodium dodecyl benzene sulfonate in water at 35-50°C.
- Add 1,3-trichloropropene dissolved in toluene dropwise to the aqueous phase for etherification.
- Separate layers, acidify the aqueous phase with hydrochloric acid, and filter to isolate the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers compelling economic and operational benefits that go beyond simple yield improvements. The elimination of expensive, high-boiling polar solvents like DMF removes a significant cost center associated with solvent purchase, recovery, and waste disposal. This shift directly contributes to substantial cost savings in the overall manufacturing budget. Furthermore, the use of readily available raw materials such as hydroquinone and 1,3-trichloropropene ensures that the supply chain is not vulnerable to the volatility of specialty chemical markets. The simplified workup procedure, which avoids complex protection and deprotection sequences, reduces the number of unit operations required. This streamlining of the process flow enhances throughput and reduces the overall production cycle time, allowing for more responsive inventory management and faster fulfillment of customer orders without compromising on quality.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of the need for high-boiling polar solvents and the reduction of raw material excess. Conventional methods often require a three-fold excess of hydroquinone to drive selectivity, which ties up capital in unreacted starting material that is difficult to recover. By achieving high selectivity with near-stoichiometric ratios, the new method drastically reduces raw material consumption. Additionally, the avoidance of DMF eliminates the energy-intensive distillation steps required for solvent recovery, leading to lower utility costs. The simplified purification process also reduces the consumption of auxiliary materials like silica gel or extraction solvents, further driving down the variable cost per kilogram of the produced intermediate.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the use of commodity chemicals that are widely available from multiple global sources. Unlike specialized catalysts or protected intermediates that may have single-source suppliers, hydroquinone and trichloropropene are produced at scale for various industries. This diversity in sourcing options mitigates the risk of supply disruptions. Moreover, the robustness of the two-phase reaction system makes it less sensitive to minor fluctuations in raw material quality, ensuring consistent output even when feedstock specifications vary slightly. This reliability is crucial for maintaining production schedules and meeting the just-in-time delivery requirements of downstream pesticide formulators, thereby strengthening the partnership between the supplier and the end manufacturer.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this method is inherently designed for industrial amplification. The water-oil two-phase system is well-understood in chemical engineering and can be easily managed in large-scale reactors with standard agitation and temperature control systems. The reduction in organic wastewater generation is a significant advantage for environmental compliance, as it lowers the load on effluent treatment plants and reduces the risk of regulatory penalties. The mild reaction temperatures (35-50°C) also enhance operational safety by minimizing the risk of thermal runaways, making the process safer for operators and equipment. These factors combined make the technology highly attractive for long-term investment and capacity expansion.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of 4-(3,3-dichloroallyloxy)phenol, based on the detailed analysis of the patent data and industry standards. These insights are intended to clarify the operational feasibility and strategic value of adopting this new synthetic route for your supply chain. Understanding these details will help stakeholders make informed decisions about process integration and vendor selection.
Q: How does the new method improve selectivity compared to traditional DMF-based processes?
A: The novel method utilizes a water-oil two-phase system with sodium dodecyl benzene sulfonate, achieving 71-85% mono-substitution selectivity, significantly reducing double-etherification by-products common in polar solvent systems.
Q: What are the environmental benefits of this synthesis route?
A: By avoiding high-boiling polar solvents like DMF and utilizing a recyclable two-phase system, the process drastically reduces organic wastewater treatment pressure and energy consumption associated with solvent recovery.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the reaction operates at mild temperatures (35-50°C) with easily available raw materials and simple workup procedures, making it highly adaptable for commercial scale-up of complex agrochemical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(3,3-dichloroallyloxy)phenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation agrochemicals. Our technical team has extensively analyzed the pathway described in CN111574337B and is fully prepared to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 4-(3,3-dichloroallyloxy)phenol meets the exacting standards required for pesticide synthesis. We are committed to being a partner that not only supplies chemicals but also provides technical solutions that enhance your overall operational efficiency.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific production requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic impact of switching to this more efficient method. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your project timeline. Our goal is to support your R&D and manufacturing goals with reliable supply and expert technical guidance, ensuring that your journey from intermediate to final product is smooth, cost-effective, and compliant with global standards.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
