Scalable Synthesis of (13cis,15trans)-Octadecadienal for Advanced Pest Control Solutions
Scalable Synthesis of (13cis,15trans)-Octadecadienal for Advanced Pest Control Solutions
The agricultural sector continuously demands more effective and environmentally friendly solutions for pest management, driving the need for high-purity insect pheromones. Patent CN111943821A introduces a robust and efficient synthetic methodology for producing (13cis,15trans)-octadecadienal, the critical active sex pheromone component for controlling the poplar moth (Micromelalopha troglodyta). This technical breakthrough addresses significant bottlenecks in previous manufacturing processes by utilizing inexpensive starting materials such as 12-bromo-1-dodecanol and acetylene gas. The disclosed route not only simplifies the operational complexity but also ensures high production efficiency and product purity, making it an ideal candidate for reliable agrochemical intermediate supplier networks seeking to optimize their portfolios. By streamlining the synthesis into five distinct unit operations, this technology offers a pathway to substantial cost reduction in pheromone manufacturing while maintaining rigorous quality standards required for biological efficacy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing long-chain unsaturated aldehydes often suffer from prohibitive production costs and convoluted reaction sequences that hinder industrial adoption. As noted in the background of the patent, earlier approaches involved complex reaction processes and operational procedures that resulted in low production efficiency and suboptimal yields. These traditional routes frequently rely on expensive reagents or require harsh conditions that complicate purification and increase waste generation. Furthermore, the difficulty in controlling stereochemistry at multiple double bond positions often leads to isomeric impurities that reduce the biological activity of the final pheromone product. Such inefficiencies create significant supply chain vulnerabilities, leading to inconsistent availability and inflated pricing for downstream formulators who depend on these specialized chemicals for integrated pest management programs.
The Novel Approach
The innovative strategy outlined in CN111943821A overcomes these historical challenges through a streamlined five-step sequence that prioritizes atom economy and operational simplicity. By initiating the synthesis with the nucleophilic substitution of 12-bromo-1-dodecanol with acetylene, the process efficiently constructs the carbon backbone using commodity chemicals. The subsequent steps involving lithiation, dehydration, selective hydrogenation, and mild oxidation are carefully optimized to maximize yield at each stage. This approach eliminates the need for exotic catalysts or extreme conditions, thereby reducing the technical barrier for commercial scale-up of complex agrochemical intermediates. The result is a manufacturing protocol that delivers high-purity products with minimal byproduct formation, directly translating to enhanced supply chain reliability and reduced lead time for high-purity pheromone intermediates needed in seasonal agricultural applications.
Mechanistic Insights into the Multi-Step Synthetic Route
The core of this synthesis lies in the precise construction of the 18-carbon chain with specific cis/trans geometry, achieved through a logical progression of functional group transformations. The process begins with the formation of 13-tetradecyn-1-ol via a base-catalyzed substitution where acetylene gas acts as the nucleophile against the bromo-alcohol. Following this, the carbon chain is extended through an organolithium-mediated addition to n-butyraldehyde, generating the diol intermediate 13-octadecyne-1,15-diol. The critical stereochemical control is established in the subsequent dehydration step, where acidic conditions promote the elimination of water to form the trans-alkyne alcohol. Finally, the use of a P-2 nickel catalyst allows for the stereoselective cis-hydrogenation of the triple bond, setting the 13-cis configuration, before a final mild oxidation converts the primary alcohol to the target aldehyde functionality.
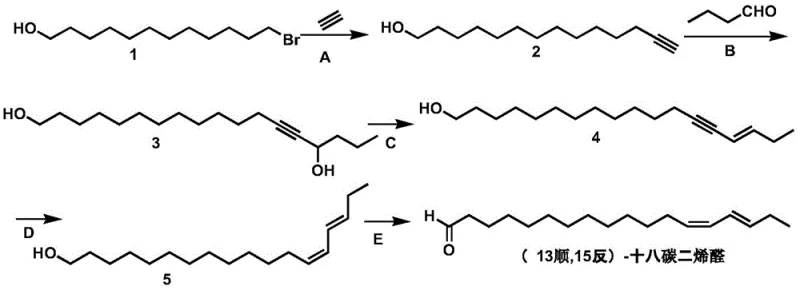
Impurity control is meticulously managed throughout the reaction sequence to ensure the final product meets stringent purity specifications. In the initial substitution step, the use of potassium iodide as a catalyst enhances the reaction rate and selectivity, minimizing the formation of elimination byproducts. During the lithiation step, maintaining temperatures between -30°C and 0°C is crucial to prevent side reactions such as polymerization or over-addition. The dehydration step utilizes trifluoroacetic acid, which provides a controlled acidic environment to favor the thermodynamic trans-product without degrading the sensitive alkyne moiety. Furthermore, the final oxidation using Dess-Martin periodinane is chosen specifically for its ability to oxidize primary alcohols to aldehydes under neutral conditions, preventing over-oxidation to carboxylic acids or isomerization of the sensitive diene system, thus ensuring the biological integrity of the pheromone.
How to Synthesize (13cis,15trans)-Octadecadienal Efficiently
Implementing this synthesis requires careful attention to reaction parameters and workup procedures to replicate the high yields reported in the patent data. The process is designed to be modular, allowing for the isolation and purification of intermediates if necessary, although the high purity of the crude products often allows for telescoping steps to improve throughput. Operators must ensure strict temperature control during the exothermic lithiation and hydrogenation phases to maintain safety and selectivity. The detailed standardized synthesis steps below outline the specific molar ratios, solvent choices, and processing conditions required to achieve the reported 90% to 99% yields across the individual transformation stages.
- Perform nucleophilic substitution of 12-bromo-1-dodecanol with acetylene gas using KOH and KI catalyst to form 13-tetradecyn-1-ol.
- Conduct lithiation of the alkyne followed by addition to n-butyraldehyde to extend the carbon chain to 13-octadecyne-1,15-diol.
- Execute acid-catalyzed dehydration to form the trans-alkyne, followed by stereoselective cis-hydrogenation and final oxidation to the aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers compelling economic benefits driven by the accessibility of raw materials and the efficiency of the chemical transformations. The reliance on 12-bromo-1-dodecanol and acetylene, which are commodity chemicals with stable global supply chains, mitigates the risk of raw material shortages that often plague specialty chemical manufacturing. Additionally, the simplified operational workflow reduces the demand for specialized equipment and extensive labor hours, contributing to significant cost savings in production overhead. The high yields achieved in each step minimize material waste and maximize the output per batch, enhancing the overall cost-effectiveness of the manufacturing process. These factors combine to create a resilient supply model that can adapt to fluctuating market demands without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences found in prior art directly lowers the cost of goods sold by reducing solvent consumption and energy usage. By avoiding expensive transition metal catalysts in favor of more economical options like P-2 nickel and utilizing efficient oxidation protocols, the process minimizes the financial burden associated with catalyst recovery and heavy metal removal. This streamlined approach ensures that the final pheromone active ingredient can be produced at a price point that supports widespread adoption in large-scale agricultural pest control programs.
- Enhanced Supply Chain Reliability: The use of robust and well-understood chemical reactions ensures consistent batch-to-batch reproducibility, which is critical for maintaining long-term contracts with agrochemical formulators. The moderate reaction conditions, often ranging from ambient to mildly elevated temperatures, reduce the risk of process upsets and equipment failures that can cause production delays. Furthermore, the scalability of the route from laboratory to industrial reactors means that suppliers can rapidly ramp up production volumes to meet seasonal peaks in demand for pest management solutions.
- Scalability and Environmental Compliance: The synthesis is designed with environmental sustainability in mind, utilizing solvents and reagents that are easier to manage and dispose of in compliance with modern regulatory standards. The high atom economy of the coupling reactions reduces the generation of hazardous waste streams, lowering the costs associated with waste treatment and environmental remediation. This alignment with green chemistry principles not only improves the corporate sustainability profile but also future-proofs the manufacturing process against tightening environmental regulations in the chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specific pheromone intermediate. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this molecule into new formulations and for procurement specialists assessing supplier capabilities. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance.
Q: What are the key advantages of this synthesis route compared to prior art?
A: This method utilizes cheap and readily available starting materials like 12-bromo-1-dodecanol and acetylene. It simplifies the operation process, reduces reaction steps, and achieves high yields suitable for industrial scale-up, addressing the low efficiency of previous methods.
Q: How is stereochemistry controlled in the production of the diene system?
A: The process ensures the correct geometry through a specific sequence: acid-catalyzed dehydration establishes the trans-alkene configuration at the 15-position, while subsequent catalytic hydrogenation using a P-2 nickel catalyst selectively reduces the triple bond to a cis-double bond at the 13-position.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the patent explicitly states the method is suitable for volume production. The reaction conditions are mild (often between -30°C to 45°C), and the workup procedures involve standard extraction and purification techniques, facilitating reliable supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (13cis,15trans)-Octadecadienal Supplier
As the global demand for sustainable pest control solutions grows, the ability to deliver high-quality pheromone intermediates at scale becomes a critical competitive advantage. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring this advanced synthesis technology to the market. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of (13cis,15trans)-octadecadienal meets the exacting standards required for effective biological activity. We are committed to supporting our partners with a stable supply of this key agrochemical intermediate, enabling the development of next-generation eco-friendly pest management products.
We invite industry leaders to collaborate with us to explore the full potential of this synthetic route for their specific applications. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your production volumes and quality requirements. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a reliable supply chain for your pheromone-based pest control initiatives.
