Advanced Phosphonate Coupling Strategy for Commercial Scale-up of High-Purity Decaisoprenol
Advanced Phosphonate Coupling Strategy for Commercial Scale-up of High-Purity Decaisoprenol
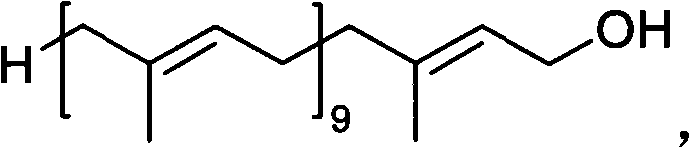
The pharmaceutical and nutritional industries rely heavily on the consistent supply of high-purity intermediates for the synthesis of vital compounds like Coenzyme Q10. Patent CN101863740B, granted in October 2013, introduces a transformative preparation method for Decaisoprenol, a critical C50 polyprenol precursor. This technology addresses long-standing challenges in terpene synthesis by replacing hazardous heavy metal reductions with a cleaner, phosphonate-based coupling strategy. The core innovation lies in the efficient condensation of Solanone with accessible phosphonate esters, followed by a highly selective hydrolysis or reduction step that preserves the delicate all-trans stereochemistry of the polyene chain. This approach not only mitigates environmental risks associated with mercury usage but also streamlines the purification process, making it an ideal candidate for reliable Decaisoprenol supplier operations aiming for industrial scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
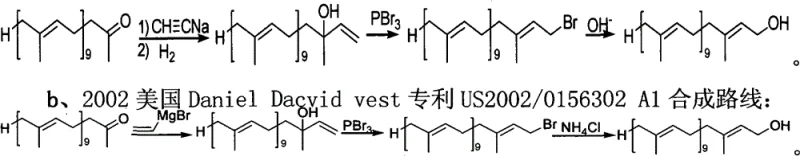
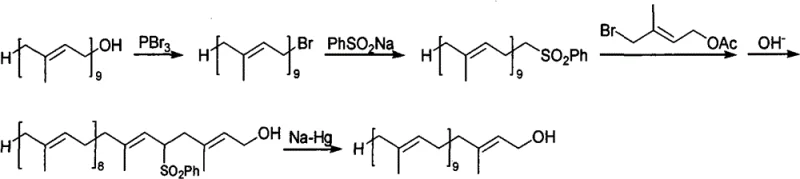
Historically, the synthesis of Decaisoprenol has been plagued by complex multi-step sequences that introduce significant impurities and safety hazards. Early methodologies, such as those described by R. Ruegg, relied on the addition of sodium acetylide or vinyl magnesium bromide to Solanone to form a tertiary alcohol, which was subsequently converted via phosphorus tribromide and hydrolysis. As illustrated in the reaction schemes below, these routes often resulted in a mixture of cis-trans isomers, necessitating difficult and costly separation processes to isolate the biologically active all-trans form required for ubiquinone production. Furthermore, alternative routes developed by researchers like Wang Chaojie utilized a sulfone elimination strategy involving sodium amalgam (Na/Hg). While effective in forming the double bond, the use of mercury poses severe environmental toxicity risks and creates substantial regulatory burdens for waste disposal, rendering these methods increasingly obsolete for modern green chemistry standards.
The Novel Approach
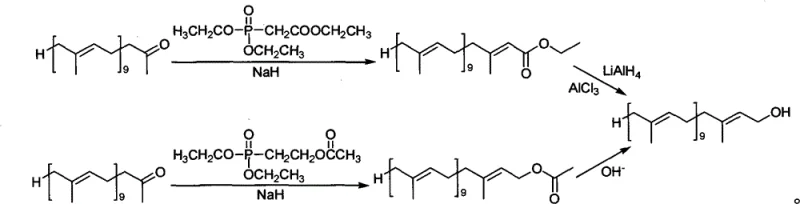
In stark contrast to these legacy methods, the novel approach detailed in CN101863740B utilizes a Horner-Wadsworth-Emmons type condensation that fundamentally simplifies the molecular construction. By reacting Solanone with acetoxyl group ethyl phosphonic acid diethyl ester or triethyl phosphate in the presence of mild bases such as sodium hydride or sodium ethylate, the process achieves high stereoselectivity directly. The subsequent conversion to the final alcohol is achieved through either straightforward alkaline hydrolysis or a controlled reduction using a modified lithium aluminum hydride system. This eliminates the need for toxic mercury reagents and avoids the use of expensive, pyrophoric organometallics like diisobutylaluminum hydride. The result is a robust, two-stage process that delivers high yields of the target Decaisoprenol with minimal byproduct formation, significantly enhancing the feasibility of cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Phosphonate-Mediated Chain Extension
The chemical elegance of this synthesis rests on the precise control of the olefination reaction and the subsequent functional group transformation. In the first stage, the deprotonation of the phosphonate ester by a strong base generates a nucleophilic carbanion which attacks the carbonyl carbon of Solanone. The reaction conditions, typically maintained between 25°C and 61°C in solvents like tetrahydrofuran or toluene, favor the formation of the thermodynamically stable E-alkene geometry. This intrinsic stereoselectivity is crucial because it minimizes the formation of Z-isomers that would otherwise complicate downstream purification. The use of specific molar ratios, such as 1.79 to 2.36 equivalents of phosphonate to ketone, ensures complete conversion of the valuable Solanone starting material, maximizing atom economy and reducing the burden of recovering unreacted precursors from the reaction mixture.
Furthermore, the patent highlights a sophisticated modification in the reduction step to ensure product integrity. When reducing the intermediate ester to the alcohol, pure lithium aluminum hydride is often too aggressive, risking the hydrogenation of the conjugated double bonds which would destroy the biological activity of the molecule. The invention solves this by introducing aluminum chloride to the reaction mixture at temperatures between -20°C and 0°C. This modification effectively tunes the reducing power of the hydride species, creating a milder reagent capable of selectively reducing the ester functionality to a primary alcohol while leaving the sensitive polyene backbone intact. This level of chemoselectivity is paramount for maintaining the high purity specifications required for clinical-grade Coenzyme Q10 production.
How to Synthesize Decaisoprenol Efficiently
The operational protocol derived from this patent offers a clear pathway for laboratory and pilot-scale production, emphasizing safety and reproducibility. The process begins with the preparation of the phosphonate anion in an inert atmosphere, followed by the controlled addition of Solanone to manage the exotherm. Following the coupling reaction, the workup involves standard aqueous washing and drying procedures to isolate the ester intermediate. Depending on the desired throughput and equipment availability, manufacturers can choose between the hydrolysis route using sodium hydroxide for simplicity or the reduction route for specific substrate variations. For a comprehensive breakdown of the exact reagent quantities, temperature profiles, and workup procedures necessary to replicate these high yields, please refer to the standardized synthesis guide below.
- Condense Solanone with acetoxyl group ethyl phosphonic acid diethyl ester or triethyl phosphate using a base catalyst like NaH or sodium ethylate in THF or toluene at 25-61°C.
- For acetate intermediates, perform hydrolysis using 10% NaOH alcoholic solution at normal temperature to yield the target alcohol.
- Alternatively, reduce the ester intermediate using a modified LiAlH4/AlCl3 system at -20 to 0°C to ensure selectivity without damaging carbon-carbon double bonds.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates into tangible operational improvements and risk mitigation. The shift away from mercury-based chemistry removes a significant environmental liability, simplifying compliance with increasingly stringent global regulations regarding heavy metal discharge. Additionally, the reliance on commodity chemicals such as triethyl phosphate and sodium hydride, rather than specialized custom synthons, stabilizes the raw material supply chain against market volatility. The simplified purification requirements, driven by the high stereoselectivity of the phosphonate coupling, reduce the consumption of chromatography media and solvents, leading to substantial cost savings in terms of both materials and waste disposal fees.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like sodium amalgam and diisobutylaluminum hydride directly lowers the bill of materials. By utilizing widely available phosphonate esters and common inorganic bases, the process avoids the premium pricing associated with specialty organometallic reagents. Furthermore, the high yield reported in the patent embodiments suggests a more efficient utilization of the expensive Solanone starting material, reducing the cost per kilogram of the final active pharmaceutical ingredient precursor significantly.
- Enhanced Supply Chain Reliability: The raw materials identified in this synthesis route, including tetrahydrofuran, toluene, and sodium hydroxide, are bulk commodities with robust global supply networks. This reduces the risk of production stoppages due to single-source supplier failures. The mild reaction conditions also imply that the process can be executed in standard stainless steel reactors without the need for exotic lining or specialized containment systems required for handling large quantities of mercury or pyrophoric liquids, thereby increasing plant availability and uptime.
- Scalability and Environmental Compliance: The absence of mercury waste streams drastically simplifies the environmental permitting process for scaling up production. Traditional sulfone routes generate toxic sludge that requires specialized treatment facilities, whereas the effluent from this phosphonate route consists primarily of phosphate salts and organic solvents that are easier to treat or recycle. This environmental friendliness facilitates faster regulatory approval for new manufacturing sites and aligns with the sustainability goals of major multinational pharmaceutical clients seeking green supply chain partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Decaisoprenol synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a factual basis for evaluating its suitability for your specific production needs. Understanding these nuances is essential for R&D teams planning technology transfer and for procurement officers negotiating supply contracts based on process efficiency.
Q: How does this patent avoid the toxicity issues of traditional Decaisoprenol synthesis?
A: The patented method eliminates the use of sodium amalgam (Na/Hg) required in traditional sulfone elimination routes, thereby removing the risk of heavy metal mercury contamination and simplifying waste treatment protocols.
Q: What represents the primary cost advantage of this phosphonate-based route?
A: By utilizing readily available phosphonate esters and avoiding expensive, moisture-sensitive reducing agents like diisobutylaluminum hydride (DIBAL-H) or complex organolithium reagents, the overall raw material cost is significantly lowered.
Q: Why is the LiAlH4/AlCl3 modification critical in the reduction step?
A: Pure lithium aluminum hydride is too reactive and risks reducing the sensitive polyene chain; adding aluminum chloride moderates the reducing power, ensuring selective reduction of the ester group while preserving the all-trans stereochemistry essential for Coenzyme Q10 activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Decaisoprenol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires deep technical expertise and rigorous quality control. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the CN101863740B process are fully realized in a GMP-compliant environment. Our state-of-the-art facilities are equipped to handle the specific solvent systems and low-temperature reduction steps required by this chemistry, guaranteeing stringent purity specifications and consistent batch-to-batch reproducibility for your Coenzyme Q10 projects.
We invite you to leverage our technical capabilities to optimize your supply chain for high-purity Decaisoprenol. Our team is prepared to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this mercury-free route can improve your margins. Please contact our technical procurement team today to request specific COA data from our pilot runs and to discuss route feasibility assessments for your upcoming development campaigns.
