Scalable Synthesis of Chiral Inchworm Sex Pheromone Intermediates for Advanced Pest Control
The global demand for environmentally friendly pest control solutions has intensified the search for efficient synthetic routes to insect sex pheromones, particularly for the management of destructive inchworm species. Patent CN109053637B, published in March 2021, introduces a robust and versatile methodology for synthesizing chiral inchworm sex pheromones and their critical epoxy intermediates. This technology addresses the longstanding challenges associated with the low natural abundance of these compounds and the complexity of establishing the requisite stereocenters. By leveraging a convergent strategy starting from inexpensive propargyl alcohol and halogenated hydrocarbons, the disclosed method offers a streamlined pathway that bypasses the need for complex resolution steps or exotic reagents. For R&D directors and procurement specialists in the agrochemical sector, this represents a significant opportunity to secure a reliable agrochemical intermediate supplier capable of delivering high-purity materials essential for Integrated Pest Management (IPM) programs.
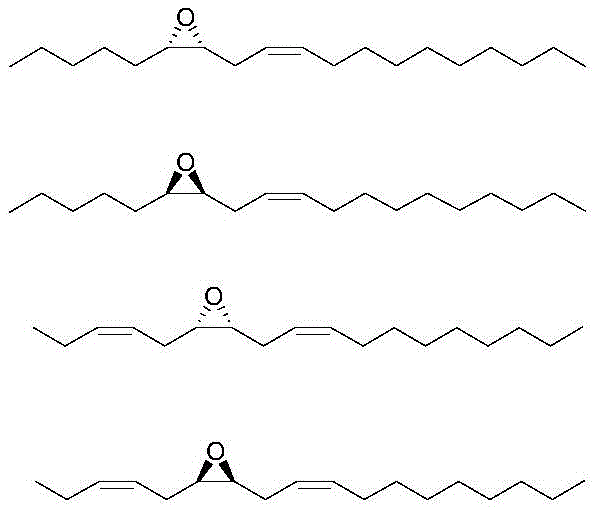
The structural complexity of inchworm sex pheromones typically involves long carbon chains with specific unsaturation patterns and, crucially, a chiral epoxy moiety. As illustrated in the provided structural data, these molecules often possess multiple stereocenters that dictate their biological specificity. Traditional extraction from natural sources is economically unviable due to minute yields, while early synthetic attempts often suffered from poor stereocontrol or excessive step counts. The innovation detailed in CN109053637B lies in its ability to construct the chiral epoxy core with high fidelity using a modified Sharpless asymmetric epoxidation protocol, followed by functionalization that preserves the stereochemical integrity throughout the subsequent derivatization steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of lepidopteran sex pheromones has been plagued by inefficiencies that hinder commercial viability. Conventional routes often rely on the chiral pool approach, starting from expensive natural amino acids or sugars, which limits scalability and drives up the cost of goods significantly. Alternatively, racemic synthesis followed by chiral resolution is frequently employed, but this inherently caps the maximum theoretical yield at 50% and generates substantial chemical waste in the form of the unwanted enantiomer. Furthermore, many traditional methods utilize harsh reaction conditions or sensitive organometallic reagents that require rigorous anhydrous environments and cryogenic temperatures, posing significant safety and engineering challenges for commercial scale-up of complex agrochemical intermediates. The accumulation of impurities from multi-step sequences also complicates downstream purification, often requiring repeated column chromatography which is impractical for ton-scale manufacturing.
The Novel Approach
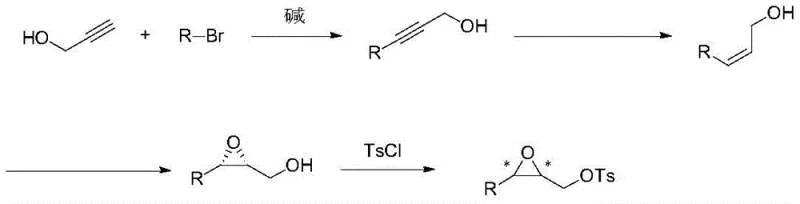
In stark contrast, the novel approach outlined in the patent utilizes a linear, high-efficiency sequence that maximizes atom economy and operational simplicity. The process initiates with a straightforward nucleophilic substitution to build the carbon skeleton, followed by a selective hydrogenation to install the cis-double bond geometry required for biological activity. The centerpiece of this strategy is the asymmetric epoxidation step, which directly installs the chiral oxygen functionality with excellent enantioselectivity. This route eliminates the need for resolution and minimizes the number of isolation steps. As shown in the general reaction scheme below, the transformation from simple alkynes to the functionalized epoxy tosylate is achieved with remarkable efficiency, providing a versatile intermediate that can be further elaborated into various specific pheromone analogues.
Mechanistic Insights into Sharpless Asymmetric Epoxidation
The cornerstone of this synthetic success is the application of the Sharpless Asymmetric Epoxidation (SAE), a reaction renowned for its ability to predictably generate chiral epoxides from allylic alcohols. In this specific protocol, the 1-substituted allyl alcohol serves as the substrate, coordinating with a titanium center formed in situ from titanium tetraisopropoxide (Ti(O-iPr)4) and a chiral tartrate ligand, specifically D-(-)-DIPT or D-(+)-DIPT depending on the desired absolute configuration. The mechanism involves the formation of a dimeric titanium-tartrate complex that creates a rigid chiral pocket. When tert-butyl hydroperoxide (TBHP) is introduced as the oxidant, it coordinates to the titanium, facilitating the transfer of an oxygen atom to the olefinic bond of the allylic alcohol. The chirality of the tartrate ligand dictates the facial selectivity of this oxygen delivery, ensuring that the epoxide ring forms on only one face of the double bond. This precise control is critical, as the biological activity of inchworm pheromones is often strictly dependent on the absolute stereochemistry of the epoxide ring.
Following the epoxidation, the resulting chiral epoxy alcohol undergoes a sulfonylation reaction with p-toluenesulfonyl chloride (TsCl) in the presence of a base like triethylamine. This step converts the hydroxyl group into a tosylate, a superior leaving group that activates the molecule for subsequent nucleophilic displacement reactions. This activation is strategically designed to allow for the attachment of various alkyl or alkenyl chains in later steps, enabling the modular synthesis of different pheromone variants from a single common intermediate. The preservation of the epoxide ring during this tosylation is paramount, and the mild conditions employed (0°C to room temperature) ensure that no ring-opening or racemization occurs. This mechanistic robustness translates directly to high product purity and consistent quality, key metrics for any high-purity agrochemical intermediate intended for field application.
How to Synthesize Chiral Epoxy Intermediate Efficiently
The practical execution of this synthesis involves four distinct operational stages that have been optimized for reproducibility and yield. The process begins with the generation of an acetylide anion from propargyl alcohol using n-butyllithium at low temperatures, followed by alkylation with a bromoalkane to extend the carbon chain. Subsequent hydrogenation over a Lindlar catalyst ensures the formation of the Z-alkene without over-reduction to the alkane. The critical asymmetric epoxidation is then performed under controlled conditions to establish chirality, followed by the final tosylation. Each step has been validated through multiple examples in the patent, demonstrating consistent yields and high optical purity. For detailed operational parameters, stoichiometry, and workup procedures, please refer to the standardized synthesis guide below.
- Perform nucleophilic substitution between propargyl alcohol and a halogenated hydrocarbon using n-butyllithium as a base to form 1-substituted propargyl alcohol.
- Conduct a selective hydrogenation reaction using a Lindlar catalyst to convert the alkyne group into a cis-alkene, yielding 1-substituted allyl alcohol.
- Execute asymmetric epoxidation using Ti(O-iPr)4 and a chiral ligand (DIPT), followed by sulfonylation with TsCl to finalize the chiral epoxy intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this synthetic route offers transformative benefits regarding cost stability and manufacturing agility. The reliance on commodity feedstocks such as propargyl alcohol and simple alkyl halides decouples production from the volatility associated with specialized chiral starting materials. This fundamental shift in raw material sourcing significantly mitigates supply risk and ensures a continuous flow of materials even during market fluctuations. Moreover, the high yield observed at each individual step compounds to provide a superior overall process efficiency, reducing the quantity of raw materials required per kilogram of final product. This efficiency is a primary driver for cost reduction in agrochemical intermediate manufacturing, allowing for more competitive pricing structures without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive chiral auxiliaries or resolution agents drastically lowers the direct material costs. Furthermore, the use of heterogeneous catalysts like Lindlar catalyst allows for easy filtration and potential recycling, minimizing catalyst loss. The simplified purification protocols, which often rely on extraction and distillation rather than preparative chromatography, reduce solvent consumption and waste disposal costs. These factors collectively contribute to a leaner manufacturing cost structure, enabling significant savings that can be passed down the supply chain or reinvested in R&D.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions means that the process is less susceptible to minor variations in temperature or reagent quality, leading to higher batch-to-batch consistency. This reliability is crucial for maintaining steady inventory levels and meeting just-in-time delivery schedules for downstream formulators. The scalability of the chemistry ensures that production can be ramped up rapidly to meet seasonal demand spikes typical in the agricultural sector, thereby reducing lead time for high-purity agrochemical intermediates during critical application windows.
- Scalability and Environmental Compliance: The synthetic route is inherently green, generating fewer byproducts and utilizing safer solvents where possible. The high atom economy of the nucleophilic substitution and epoxidation steps minimizes waste generation, simplifying compliance with increasingly stringent environmental regulations. The process is designed for large-scale operation, with examples demonstrating successful translation from gram to multi-gram scales without loss of efficiency. This scalability ensures that the technology can support global demand volumes, making it a sustainable long-term solution for the industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on performance metrics and operational feasibility. Understanding these aspects is vital for technical teams evaluating the integration of this intermediate into their existing product portfolios or formulation pipelines.
Q: What is the enantiomeric excess (ee) achieved in this synthesis?
A: The patented method utilizing Sharpless asymmetric epoxidation consistently achieves high optical purity, with ee values exceeding 94% across various examples, ensuring effective biological activity for pest control applications.
Q: Are the raw materials for this process readily available?
A: Yes, the synthesis relies on commodity chemicals such as propargyl alcohol, common alkyl halides, and standard catalysts like Lindlar catalyst and titanium tetraisopropoxide, ensuring a stable and cost-effective supply chain.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The process features short reaction steps, mild conditions, and high yields at each stage, making it highly amenable to commercial scale-up from pilot plants to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Inchworm Sex Pheromone Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality chiral intermediates in the development of next-generation biopesticides. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of CN109053637B can be seamlessly translated into industrial reality. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced chiral HPLC and GC-MS instrumentation to guarantee that every batch meets the exacting standards required for effective pest control. Our commitment to quality assurance ensures that the enantiomeric excess and chemical purity of our intermediates remain consistent, safeguarding the efficacy of your final formulations.
We invite procurement leaders and R&D directors to collaborate with us to leverage this advanced synthetic technology for your specific applications. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your volume requirements and logistical needs. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and sample availability. Let us help you optimize your supply chain and accelerate the deployment of sustainable pest management solutions globally.
