Advanced Synthesis of Hyperfluorescent 9-Aminoacridine Derivatives for Commercial OLED Applications
The rapid evolution of the organic light-emitting diode (OLED) industry demands materials that combine exceptional fluorescence efficiency with structural stability and scalable manufacturability. Patent CN103755634A addresses this critical market need by disclosing a novel class of 9-aminoacridine derivatives characterized by extended conjugated systems and rigid planar structures. These molecular architectures are specifically engineered to maximize pi-electron delocalization, resulting in superior fluorescence properties compared to traditional acridine-based emitters. The patent outlines a sophisticated multi-step synthesis that leverages modern catalytic strategies, including magnetic nanoparticle catalysis and microwave-assisted coupling, to achieve high purity and yield. For R&D directors and procurement specialists in the electronic chemicals sector, this technology represents a significant advancement in the development of next-generation display materials. The general structure of these high-performance derivatives, featuring variable substituents R1 and R2 to tune electronic properties, is illustrated below.
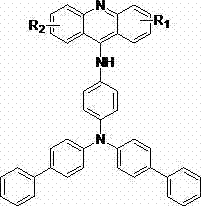
Furthermore, the specific embodiments detailed in the patent, such as compounds (1) through (12), demonstrate the versatility of this synthetic platform in accommodating various functional groups like methyl, ethyl, methoxy, and trifluoromethyl moieties. This structural flexibility allows manufacturers to fine-tune the emission wavelength and charge transport properties of the final OLED material without compromising the core synthetic efficiency. The ability to produce such complex, highly conjugated molecules with consistent quality is paramount for maintaining the color gamut and longevity of commercial display panels. As the demand for high-resolution and energy-efficient screens grows, the availability of reliable suppliers capable of delivering these specialized intermediates becomes a strategic asset for the supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for acridine derivatives often suffer from significant drawbacks that hinder their commercial viability for high-end electronic applications. Conventional methods frequently rely on harsh reaction conditions, such as extremely high temperatures or the use of corrosive reagents in stoichiometric excess, which can lead to the formation of complex impurity profiles that are difficult to remove. Moreover, standard protocols for introducing bulky amine substituents at the 9-position of the acridine core often result in poor regioselectivity and low conversion rates, necessitating extensive purification steps that drastically reduce overall yield. The lack of efficient catalytic systems in older methodologies means that heavy metal residues may persist in the final product, posing a severe risk to the performance and stability of OLED devices. Additionally, the absence of modular building blocks in traditional synthesis limits the ability to rapidly iterate on molecular designs to optimize fluorescence quantum yields.
The Novel Approach
In stark contrast, the methodology presented in CN103755634A introduces a streamlined and highly efficient pathway that overcomes these historical bottlenecks through the strategic application of transition metal catalysis. The novel approach utilizes a palladium-catalyzed cross-coupling reaction in the final step to attach the large triphenylamine-derived moiety, ensuring high selectivity and robust bond formation under relatively mild conditions. By employing a magnetic copper-iron oxide catalyst in the initial condensation step, the process facilitates easy catalyst recovery and reuse, which significantly simplifies the workup procedure and reduces chemical waste. The integration of microwave irradiation in the final coupling stage further accelerates reaction kinetics, allowing for shorter reaction times and improved energy efficiency compared to conventional thermal heating. This modern synthetic strategy not only enhances the purity of the final 9-aminoacridine derivative but also ensures a more consistent and reproducible manufacturing process suitable for industrial scale-up.
Mechanistic Insights into Pd-Catalyzed C-N Coupling and Magnetic Catalysis
The core of this innovative synthesis lies in the precise orchestration of two distinct catalytic cycles that build the complex molecular architecture step-by-step. The initial formation of the acridone skeleton involves a copper-mediated oxidative coupling where the CuFe2O4 magnetic catalyst plays a dual role as both an oxidant and a Lewis acid promoter. This heterogeneous catalytic system operates effectively in dimethyl sulfoxide (DMSO) at temperatures between 100°C and 105°C, facilitating the C-N bond formation between the aniline and benzoic acid precursors with high atom economy. Following cyclization and activation of the carbonyl group via chlorination and subsequent azide reduction, the pathway culminates in a sophisticated Buchwald-Hartwig amination. In this critical final step, a palladium catalyst, specifically Pd(dppf)Cl2, mediates the coupling between the 9-aminoacridine intermediate and the sterically demanding tris(biphenyl-4-yl)amine derivative. The complete reaction sequence, highlighting the transformation from simple starting materials to the highly conjugated final product, is depicted in the following schematic.
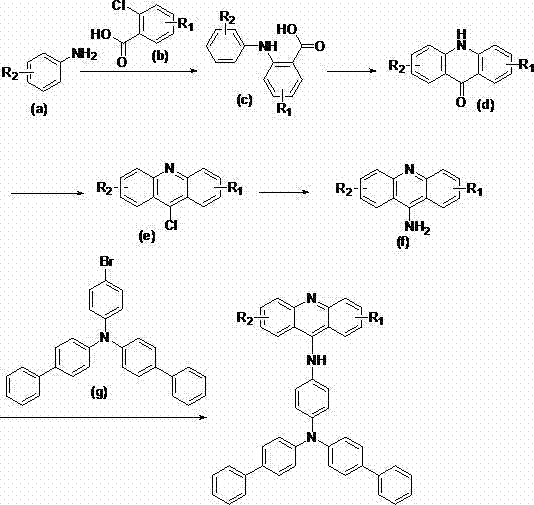
From a mechanistic perspective, the success of this route in generating hyperfluorescent materials is attributed to the preservation of the rigid planar structure throughout the synthesis. The extended pi-conjugation system, formed by the fusion of the acridine core with the biphenyl-amine side chains, creates a large delocalized electron cloud that is easily excited upon energy absorption. This structural feature is directly responsible for the observed red-shift in fluorescence emission and the enhanced quantum efficiency reported in the patent data. Furthermore, the use of cesium carbonate as a base in the final coupling step ensures effective deprotonation of the amine nucleophile without promoting side reactions that could degrade the sensitive acridine framework. The careful selection of solvents like 1,4-dioxane or toluene optimizes the solubility of the bulky intermediates, ensuring homogeneous reaction conditions that are critical for achieving the reported purities of over 99%. Understanding these mechanistic nuances is essential for R&D teams aiming to replicate or further optimize this process for specific commercial requirements.
How to Synthesize 9-Aminoacridine Derivative Efficiently
The synthesis of these high-value OLED intermediates requires strict adherence to the optimized parameters defined in the patent to ensure maximum yield and purity. The process begins with the preparation of the key acridone intermediate, followed by activation and functionalization to enable the final coupling. Each step, from the magnetic catalyzed condensation to the microwave-assisted amination, has been fine-tuned to balance reaction rate with product integrity. Operators must pay close attention to temperature control and reagent stoichiometry, particularly during the chlorination and azide reduction phases, to prevent the formation of hazardous byproducts. The detailed standardized synthetic steps, including specific molar ratios and reaction times for each transformation, are outlined in the guide below.
- Condense aniline and benzoic acid derivatives using CuFe2O4 magnetic catalyst in DMSO at 100-105°C to form intermediate (c).
- Perform acid-catalyzed cyclization in toluene at 100-110°C to generate the acridone core intermediate (d).
- Execute chlorination with thionyl chloride followed by azide substitution and reduction to obtain the amino-intermediate (f).
- Conduct the final Buchwald-Hartwig amination with a triphenylamine derivative using Pd(dppf)Cl2 under microwave irradiation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this patented synthetic route offers tangible benefits that extend beyond mere technical performance, directly impacting the bottom line through process optimization. The implementation of a magnetic catalyst in the early stages of synthesis eliminates the need for complex filtration processes typically associated with heterogeneous catalysis, thereby reducing solvent usage and waste disposal costs significantly. This simplification of the workup procedure translates into a more streamlined manufacturing workflow that requires less labor and equipment downtime. Moreover, the high yields reported across multiple embodiments, consistently exceeding 70%, indicate a robust process that minimizes raw material loss and maximizes output per batch. Such efficiency is crucial for maintaining competitive pricing in the volatile market of electronic chemicals while ensuring a steady supply of high-quality materials.
- Cost Reduction in Manufacturing: The elimination of expensive and difficult-to-remove transition metal catalysts in favor of a recoverable magnetic system drastically lowers the cost of goods sold. By avoiding the need for specialized scavenging resins or extensive chromatographic purification to remove metal residues, manufacturers can achieve substantial cost savings in downstream processing. The use of microwave heating in the final step also contributes to energy efficiency, reducing the overall utility consumption per kilogram of product produced. These cumulative efficiencies allow for a more aggressive pricing strategy without compromising profit margins, making the final OLED material more attractive to cost-sensitive display manufacturers.
- Enhanced Supply Chain Reliability: The synthetic route relies on commercially available and stable starting materials, such as substituted anilines and benzoic acids, which mitigates the risk of supply disruptions associated with exotic or proprietary reagents. The robustness of the reaction conditions, which tolerate a range of substituents without significant loss in yield, provides flexibility in sourcing raw materials from different vendors. This resilience ensures a continuous and reliable supply of the 9-aminoacridine derivative, preventing production bottlenecks that could delay the launch of new display technologies. Furthermore, the scalability of the process from gram to kilogram scale has been demonstrated, assuring partners of long-term availability.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing waste generation and utilizing safer solvents where possible. The magnetic separation technique reduces the volume of solid waste sent for treatment, aligning with increasingly stringent environmental regulations in the chemical industry. The high atom economy of the coupling reactions ensures that the majority of the input mass is incorporated into the final product, reducing the environmental footprint of the manufacturing operation. This commitment to sustainability not only aids in regulatory compliance but also enhances the brand reputation of suppliers who prioritize eco-friendly production methods in their value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel 9-aminoacridine derivatives. The answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and reliability. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this material into their existing product lines or supply networks. Comprehensive responses regarding purity standards, yield expectations, and structural customization options are provided below to facilitate informed decision-making.
Q: What is the fluorescence quantum yield of the synthesized 9-aminoacridine derivatives?
A: According to patent CN103755634A, the synthesized compounds exhibit high fluorescence quantum efficiencies ranging from approximately 0.40 to 0.77, significantly outperforming standard 9-aminoacridine references.
Q: How does the use of CuFe2O4 impact the manufacturing process?
A: The utilization of CuFe2O4 as a magnetic catalyst in the initial condensation step allows for simplified product isolation via magnetic separation, thereby reducing solvent consumption and improving overall process efficiency.
Q: Are these derivatives suitable for large-scale OLED production?
A: Yes, the patent reports yields exceeding 70% with purities above 99%, and the synthetic route utilizes robust reagents and scalable techniques like microwave-assisted heating, indicating strong potential for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 9-Aminoacridine Derivative Supplier
As the global demand for high-performance OLED materials continues to surge, partnering with a technically proficient manufacturer is essential for securing a competitive edge in the display market. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in complex organic synthesis to deliver custom solutions that meet the rigorous standards of international electronics giants. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from pilot trials to full-scale manufacturing. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 9-aminoacridine derivative meets the exacting requirements for fluorescence efficiency and stability.
We invite you to collaborate with us to explore the full potential of this patented technology for your specific applications. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can reduce your overall material costs. Please contact us today to request specific COA data and route feasibility assessments, and let us help you accelerate the development of your next-generation optoelectronic devices with our premium chemical intermediates.
