Advanced One-Step Synthesis of Flomoxef Intermediate 1 via Selenium Dioxide Oxidation for Commercial Scale-Up
Introduction to Next-Generation Flomoxef Intermediate Manufacturing
The pharmaceutical landscape for broad-spectrum antibiotics continues to evolve, driven by the urgent need for efficient and safe manufacturing processes for critical active ingredients. Patent CN114315858B, published in May 2023, introduces a transformative synthesis method for Flomoxef Intermediate 1, a pivotal precursor in the production of Sodium Flomoxef. This oxacephem antibiotic is renowned for its potent activity against Gram-positive cocci and Enterobacteriaceae, making its reliable supply chain essential for global healthcare infrastructure. The patented technology addresses long-standing bottlenecks in the industry by replacing hazardous chlorination steps with a mild, high-yield selenium dioxide oxidation protocol. 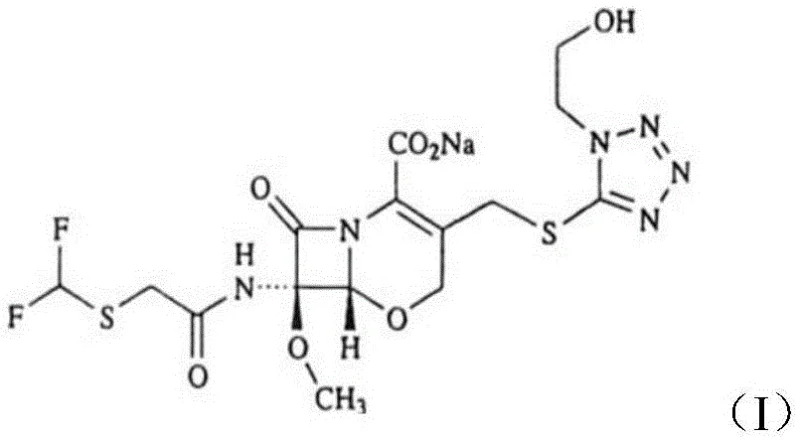 This innovation not only streamlines the synthetic pathway but also aligns with modern green chemistry principles by reducing waste and eliminating toxic reagents. For R&D directors and procurement specialists, understanding this shift is crucial for optimizing supply chains and ensuring the continuous availability of high-purity intermediates required for final drug formulation.
This innovation not only streamlines the synthetic pathway but also aligns with modern green chemistry principles by reducing waste and eliminating toxic reagents. For R&D directors and procurement specialists, understanding this shift is crucial for optimizing supply chains and ensuring the continuous availability of high-purity intermediates required for final drug formulation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Flomoxef Intermediate 1 has been plagued by complex, multi-step routes that pose significant safety and economic challenges. Traditional literature describes pathways starting from 6-APA (6-aminopenicillanic acid), which require a tedious sequence of acidification, esterification, oxidation, and chlorination. As illustrated in the prior art, these methods often involve the use of elemental chlorine gas, a highly toxic and corrosive substance that necessitates specialized equipment and rigorous safety protocols. 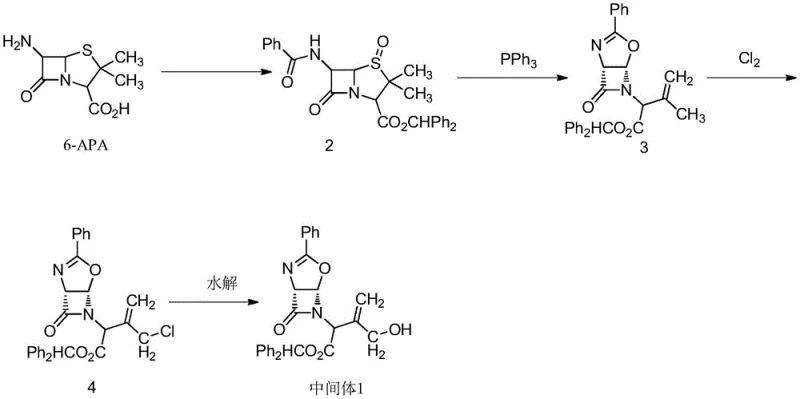 Furthermore, alternative routes utilizing silver nitrate for nitration steps introduce prohibitive costs due to the expense of silver salts, rendering them unsuitable for large-scale industrial application. These conventional processes typically suffer from low overall yields, often hovering around 30%, primarily due to material loss across numerous purification stages and the formation of difficult-to-remove impurities. The reliance on harsh reagents also generates substantial hazardous waste, complicating environmental compliance and increasing disposal costs for manufacturers.
Furthermore, alternative routes utilizing silver nitrate for nitration steps introduce prohibitive costs due to the expense of silver salts, rendering them unsuitable for large-scale industrial application. These conventional processes typically suffer from low overall yields, often hovering around 30%, primarily due to material loss across numerous purification stages and the formation of difficult-to-remove impurities. The reliance on harsh reagents also generates substantial hazardous waste, complicating environmental compliance and increasing disposal costs for manufacturers.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a direct oxidation strategy that dramatically simplifies the manufacturing landscape. The core innovation lies in the conversion of Compound 3 directly into Intermediate 1 using selenium dioxide (SeO2) in the presence of specific auxiliary agents. 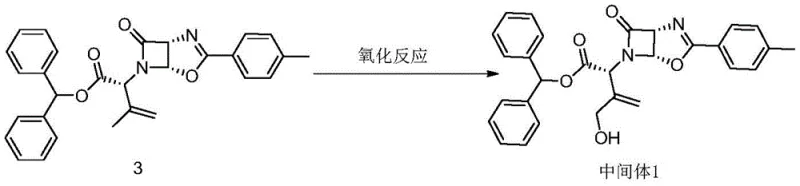 This single-step transformation bypasses the need for dangerous chlorination and expensive metal catalysts, offering a streamlined path from starting material to the key intermediate. The reaction conditions are remarkably mild, operating effectively at temperatures between 0°C and 35°C, which reduces energy consumption and thermal stress on the molecular structure. By employing auxiliaries such as t-butyl hydroperoxide or acetic acid, the catalytic efficiency of selenium dioxide is enhanced, allowing for lower reagent loading while maintaining high conversion rates. This methodological shift represents a paradigm change in how this critical pharmaceutical building block is produced, prioritizing safety, efficiency, and scalability.
This single-step transformation bypasses the need for dangerous chlorination and expensive metal catalysts, offering a streamlined path from starting material to the key intermediate. The reaction conditions are remarkably mild, operating effectively at temperatures between 0°C and 35°C, which reduces energy consumption and thermal stress on the molecular structure. By employing auxiliaries such as t-butyl hydroperoxide or acetic acid, the catalytic efficiency of selenium dioxide is enhanced, allowing for lower reagent loading while maintaining high conversion rates. This methodological shift represents a paradigm change in how this critical pharmaceutical building block is produced, prioritizing safety, efficiency, and scalability.
Mechanistic Insights into Selenium Dioxide Catalyzed Allylic Oxidation
The chemical elegance of this new process is rooted in the specific mechanism of selenium dioxide-mediated allylic oxidation. In this reaction, SeO2 acts as a selective oxidant that targets the allylic position of the alkene moiety present in Compound 3. The mechanism typically involves the formation of a selenite ester intermediate, followed by a [2,3]-sigmatropic rearrangement or hydrolysis to yield the allylic alcohol functionality found in Intermediate 1. The addition of auxiliary agents plays a critical role in this catalytic cycle; for instance, t-butyl hydroperoxide can serve as a co-oxidant, regenerating the active selenium species and driving the reaction to completion with higher atom economy. Acetic acid, another preferred auxiliary, likely functions by protonating intermediate species or stabilizing the transition state, thereby lowering the activation energy required for the oxidation. This precise control over the reaction pathway ensures that the oxidation occurs selectively at the desired carbon center without affecting other sensitive functional groups within the complex beta-lactam fused ring system.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based chlorination methods. Traditional chlorination often leads to a broad spectrum of side products, including polychlorinated derivatives and ring-opened by-products, which are notoriously difficult to separate from the target molecule. The SeO2 oxidation, being more ionic and selective in nature, minimizes the formation of such structural analogs. The patent data indicates that this selectivity translates directly into superior product quality, with HPLC purity consistently exceeding 98%. The avoidance of aggressive halogenating agents also prevents the formation of genotoxic impurities often associated with alkyl halides, a critical consideration for regulatory compliance in API manufacturing. Furthermore, the work-up procedure involving simple alkaline quenching and solvent extraction effectively removes selenium residues, ensuring the final intermediate meets stringent heavy metal specifications required for downstream pharmaceutical synthesis.
How to Synthesize Flomoxef Intermediate 1 Efficiently
Implementing this novel synthesis route requires careful attention to reagent stoichiometry and temperature control to maximize the benefits of the SeO2 catalytic system. The process is designed to be robust, tolerating a range of solvents including dichloromethane, ethyl acetate, and methanol, which provides flexibility for process engineers optimizing for cost or environmental impact. The following guide outlines the standardized operational parameters derived from the patent examples, ensuring reproducible high-yield results suitable for pilot and commercial scale operations. Detailed standard operating procedures regarding specific mixing rates and crystallization kinetics should be consulted for full GMP compliance.
- Prepare the oxidant solution by dissolving selenium dioxide in a suitable solvent such as dichloromethane or water, and cool the mixture to between 0°C and 10°C.
- Add an auxiliary agent such as t-butyl hydroperoxide or acetic acid to the cooled oxidant solution and stir to activate the catalytic system.
- Slowly add a solution of Compound 3 dropwise to the reaction mixture, maintain temperature between 0°C and 35°C, then quench with base and crystallize the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this SeO2-based synthesis route offers compelling strategic advantages that extend beyond mere technical feasibility. The elimination of hazardous chlorine gas from the process map significantly de-risks the supply chain, removing the need for specialized storage facilities and reducing insurance premiums associated with toxic chemical handling. This simplification allows for manufacturing in a broader range of facilities, enhancing supply continuity and reducing the risk of production stoppages due to safety audits or regulatory crackdowns on dangerous processes. Moreover, the reduction in synthetic steps from a multi-stage sequence to a single oxidation event drastically cuts down on processing time and labor costs, leading to a more responsive supply chain capable of meeting fluctuating market demands for antibiotics.
- Cost Reduction in Manufacturing: The economic implications of this new method are profound, primarily driven by the removal of expensive reagents and the consolidation of process steps. By avoiding the use of silver nitrate and elemental chlorine, manufacturers can achieve substantial raw material cost savings, as these commodities are subject to volatile market pricing and high logistical costs. Additionally, the higher molar yield of over 65% compared to the historical 30% means that less starting material is required to produce the same amount of intermediate, effectively doubling the throughput efficiency of existing reactor capacity. The simplified post-treatment, which relies on standard extraction and crystallization rather than complex chromatographic purifications, further reduces utility consumption and waste disposal expenses, contributing to a significantly lower cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available and stable reagents such as selenium dioxide and common organic solvents. Unlike specialized catalysts or hazardous gases that may face shipping restrictions or supply shortages, the inputs for this process are commodity chemicals with robust global supply networks. The mild reaction conditions also reduce wear and tear on production equipment, extending asset life and minimizing unplanned maintenance downtime. This reliability ensures that pharmaceutical companies can secure a steady flow of high-purity intermediates, safeguarding their own production schedules for the final antibiotic formulations and preventing stock-outs in the healthcare sector.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) standpoint, this process represents a significant step forward in sustainable manufacturing. The absence of chlorinated waste streams simplifies effluent treatment, reducing the burden on wastewater treatment plants and lowering the environmental footprint of the facility. The ability to run the reaction at near-ambient temperatures (20-30°C) minimizes energy demand for heating or deep cooling, aligning with corporate sustainability goals. Furthermore, the high selectivity of the reaction reduces the generation of organic waste, facilitating easier compliance with increasingly stringent environmental regulations. This 'green' profile makes the technology highly attractive for scaling up to multi-ton production levels without encountering the regulatory hurdles often associated with older, dirtier chemical processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selenium dioxide oxidation technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational safety. Understanding these details is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the long-term value proposition of this manufacturing route.
Q: What are the primary safety advantages of the SeO2 oxidation route over traditional chlorination methods?
A: The novel SeO2 oxidation route completely eliminates the use of elemental chlorine gas, which is classified as a highly dangerous chemical process. This significantly reduces safety risks associated with toxic gas handling and corrosion, while also avoiding the formation of chlorinated by-products that complicate purification.
Q: How does the new synthesis method impact the overall yield and purity of Flomoxef Intermediate 1?
A: By shortening the synthetic route to a single oxidation step, the method achieves a molar yield of greater than 65% and an HPLC purity exceeding 98%. This is a substantial improvement over prior art methods which often suffered from yields around 30% due to multi-step degradation and difficult purification.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process utilizes mild reaction temperatures ranging from 0°C to 35°C and common solvents like dichloromethane or ethyl acetate. The work-up involves standard extraction and crystallization techniques, making it highly scalable and environmentally friendlier compared to routes requiring expensive silver reagents or harsh acidic conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Flomoxef Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the SeO2 oxidation pathway and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering Flomoxef Intermediate 1 with stringent purity specifications, leveraging our rigorous QC labs to ensure every batch meets the >98% HPLC purity benchmark established by this innovative patent. Our facility is equipped to handle the specific solvent systems and crystallization protocols required for this process, guaranteeing a consistent supply of high-quality material for your API synthesis needs.
We invite you to collaborate with us to unlock the full potential of this cost-effective and safe manufacturing route. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this new chemistry can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on the most efficient and reliable foundation available in the industry.
