Advanced One-Pot Synthesis of Quizalofop-P-Ethyl Intermediate for Commercial Herbicide Production
Advanced One-Pot Synthesis of Quizalofop-P-Ethyl Intermediate for Commercial Herbicide Production
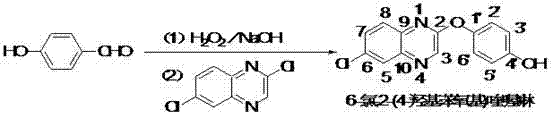
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of key agrochemical intermediates. Patent CN102659694A introduces a transformative one-pot methodology for producing 6-chloro-2-(4-hydroxyphenoxy)quinoxaline, a critical precursor for the widely used herbicide quizalofop-p-ethyl. This technical breakthrough addresses long-standing economic and environmental challenges associated with traditional manufacturing routes. By shifting the starting material from expensive hydroquinone to the more stable and cost-effective p-hydroxybenzaldehyde, the process not only optimizes the cost structure but also simplifies the operational workflow. For R&D directors and procurement strategists, understanding this shift is vital for securing a competitive edge in the agrochemical supply chain. The following analysis dissects the mechanistic advantages and commercial implications of this patented technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 6-chloro-2-(4-hydroxyphenoxy)quinoxaline has relied heavily on the direct condensation of hydroquinone with 2,6-dichloroquinoxaline. While chemically straightforward, this legacy approach suffers from significant economic and logistical drawbacks that hinder modern large-scale manufacturing. The primary bottleneck is the volatility and rising market price of hydroquinone, which directly inflates the cost of goods sold (COGS) for the final herbicide active ingredient. Furthermore, the handling of bulk hydroquinone presents severe occupational health and safety risks due to its toxicity and potential for skin sensitization. From an environmental compliance perspective, processes utilizing stoichiometric amounts of hydroquinone generate wastewater streams with high chemical oxygen demand (COD) loads, specifically containing unreacted phenolic compounds that are notoriously difficult and expensive to treat. These factors collectively create a fragile supply chain vulnerable to raw material price fluctuations and regulatory tightening.
The Novel Approach
The patented one-pot synthesis described in CN102659694A fundamentally reengineers this workflow by introducing an in-situ oxidation strategy. Instead of purchasing and handling hazardous hydroquinone, the process begins with p-hydroxybenzaldehyde, a stable and abundant commodity chemical. In the first stage of the reactor, p-hydroxybenzaldehyde undergoes a Dakin-like oxidation using hydrogen peroxide and sodium hydroxide to generate hydroquinone directly within the reaction medium. This intermediate is never isolated; instead, 2,6-dichloroquinoxaline is introduced immediately into the same vessel. This telescoping of two distinct chemical transformations into a single operational unit eliminates the need for intermediate filtration, drying, and transfer steps. The result is a dramatic reduction in processing time and solvent consumption. By integrating the oxidation and nucleophilic substitution steps, the method achieves a seamless flow that enhances overall yield while minimizing waste generation.
Mechanistic Insights into In-Situ Oxidation and Condensation
The core chemical innovation lies in the precise control of the Dakin oxidation followed by a nucleophilic aromatic substitution (SnAr). In the initial phase, p-hydroxybenzaldehyde reacts with hydrogen peroxide under alkaline conditions. The hydroperoxide anion attacks the carbonyl carbon, forming a tetrahedral intermediate that subsequently rearranges to release formate and generate the phenolate anion of hydroquinone. Critical to this step is the temperature control, which must be maintained below 50°C to prevent the over-oxidation of the phenolic ring or the decomposition of hydrogen peroxide, ensuring high selectivity for the desired dihydroxy species. Once the hydroquinone is formed in situ, the reaction environment is modified by the addition of toluene and 2,6-dichloroquinoxaline. The electron-withdrawing nitrogen atoms in the quinoxaline ring activate the chlorine at the 2-position towards nucleophilic attack. The phenolate oxygen, acting as a strong nucleophile, displaces the chloride ion to form the ether linkage. This SnAr mechanism is driven to completion by the continuous removal of the chloride byproduct and the maintenance of a basic pH, which keeps the phenol in its reactive deprotonated state.
Impurity control in this one-pot system is managed through the sequential addition of reagents and strict thermal profiling. A common side reaction in such systems is the formation of bis-substituted byproducts where both chlorines on the quinoxaline ring are displaced. However, the patent specifies a molar ratio strategy where the oxidant and base are carefully dosed relative to the aldehyde, and the subsequent addition of the dichloroquinoxaline is timed to match the concentration of the generated nucleophile. Furthermore, the use of toluene as a co-solvent in the second stage helps to solubilize the organic reactants while potentially allowing for azeotropic removal of water during the reflux stage, driving the equilibrium forward. The reported gas chromatography (GC) data indicates that this controlled environment suppresses side reactions effectively, yielding a crude product with purity levels consistently exceeding 98% without the need for complex chromatographic purification, which is essential for cost-effective industrial scale-up.
How to Synthesize 6-Chloro-2-(4-hydroxyphenoxy)quinoxaline Efficiently
Implementing this synthesis route requires careful attention to the transition between the oxidative and condensative phases. The process begins by dissolving p-hydroxybenzaldehyde in water, followed by the simultaneous dropwise addition of hydrogen peroxide and sodium hydroxide solution. It is imperative to monitor the exotherm closely, ensuring the internal temperature does not exceed 50°C during this oxidation phase to guarantee complete conversion to the hydroquinone intermediate. Once the oxidation is confirmed complete via GC tracking, the reaction mixture serves as the feed for the second stage without any workup. Distilled water, 2,6-dichloroquinoxaline, and toluene are charged into the reactor. The mixture is then heated, and a second dose of alkali is added to initiate the coupling reaction. The detailed standardized operating procedures, including specific stirring rates, addition times, and quenching protocols required for GMP-compliant manufacturing, are outlined below.
- Dissolve p-hydroxybenzaldehyde in water and simultaneously add hydrogen peroxide and sodium hydroxide while maintaining temperature below 50°C to generate hydroquinone in situ.
- Without isolation, introduce distilled water, 2,6-dichloroquinoxaline, and toluene to the reaction mixture.
- Heat the mixture to 90-95°C, add alkali solution dropwise to induce condensation, and reflux until completion to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this one-pot technology offers tangible strategic benefits beyond simple yield improvements. The shift away from hydroquinone as a direct feedstock mitigates exposure to a volatile raw material market, stabilizing long-term cost projections. Additionally, the consolidation of two reaction steps into a single vessel significantly reduces the capital expenditure required for reactor train capacity, as fewer unit operations are needed to produce the same volume of intermediate. This intensification of the process translates directly into lower utility consumption and reduced labor hours per kilogram of product. The simplified workflow also decreases the risk of cross-contamination and material loss during transfer, enhancing the overall reliability of the supply chain. By adopting this method, manufacturers can secure a more resilient and cost-efficient source of high-purity agrochemical intermediates.
- Cost Reduction in Manufacturing: The substitution of expensive hydroquinone with p-hydroxybenzaldehyde creates a fundamental advantage in raw material costs. Since p-hydroxybenzaldehyde is generally more stable and less prone to price spikes than hydroquinone, the baseline cost of production is lowered. Furthermore, the elimination of the isolation step for the hydroquinone intermediate removes the associated costs of filtration, drying, and packaging equipment. The one-pot nature of the reaction also implies a reduction in solvent usage and waste disposal fees, as there is no need to wash and dry an intermediate solid. These cumulative savings allow for a significantly more competitive pricing structure for the final herbicide active ingredient, providing a buffer against market fluctuations.
- Enhanced Supply Chain Reliability: Relying on p-hydroxybenzaldehyde diversifies the raw material portfolio, reducing dependency on suppliers of hazardous phenols which are often subject to strict transportation and storage regulations. The simplified process flow reduces the number of potential failure points in the manufacturing line, leading to more consistent batch-to-batch quality and on-time delivery performance. With fewer unit operations, the lead time from raw material intake to finished intermediate is drastically shortened. This agility allows suppliers to respond more rapidly to sudden increases in demand from downstream formulators, ensuring continuity of supply even during peak agricultural seasons when herbicide demand surges globally.
- Scalability and Environmental Compliance: The environmental profile of this process is markedly superior to conventional methods. By generating hydroquinone in situ and consuming it immediately, the process avoids the accumulation of toxic phenolic waste streams that require specialized and costly treatment facilities. The reduction in wastewater load and the avoidance of heavy solvent exchanges make this route highly scalable without proportionally increasing the environmental footprint. Regulatory bodies increasingly favor processes that minimize hazard and waste, meaning facilities adopting this technology face fewer permitting hurdles and lower compliance costs. This sustainability advantage future-proofs the manufacturing asset against tightening global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this one-pot synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN102659694A, providing a factual basis for evaluating the feasibility of this route for your specific production needs. Understanding these nuances is critical for making informed decisions about technology transfer and vendor qualification.
Q: Why is p-hydroxybenzaldehyde preferred over hydroquinone for this synthesis?
A: Using p-hydroxybenzaldehyde significantly reduces raw material costs compared to volatile hydroquinone and eliminates the environmental burden of treating high-concentration hydroquinone wastewater, aligning with stricter green chemistry regulations.
Q: What are the critical temperature controls in the one-pot process?
A: The oxidation step must be strictly controlled below 50°C to prevent over-oxidation, while the subsequent condensation with 2,6-dichloroquinoxaline requires heating to 90-95°C to ensure complete nucleophilic substitution.
Q: How does this method impact the purity profile of the intermediate?
A: The one-pot method minimizes intermediate handling losses and exposure to air, consistently achieving GC purities above 98% with yields exceeding 91%, ensuring high quality for downstream herbicide synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Chloro-2-(4-hydroxyphenoxy)quinoxaline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the efficiency of your herbicide production depends on the quality and consistency of your intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative one-pot chemistry described in CN102659694A can be seamlessly translated from the lab to the plant. Our rigorous QC labs and stringent purity specifications guarantee that every batch of 6-chloro-2-(4-hydroxyphenoxy)quinoxaline meets the exacting standards required for agrochemical registration and formulation. We are committed to delivering high-purity agrochemical intermediates that empower your R&D and production teams to succeed.
We invite you to leverage our technical expertise to optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can reduce your overall production costs while ensuring a reliable supply of this critical quizalofop intermediate.
