Advanced Copper-Catalyzed Synthesis of N-aryl-D-glucosamine for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies for the synthesis of complex carbohydrate derivatives, particularly N-aryl-D-glucosamine, which serves as a critical scaffold in the development of novel therapeutic agents. Patent CN102838639A introduces a groundbreaking synthetic method that leverages copper-catalyzed C-N cross-coupling reactions to achieve high-efficiency production of these valuable intermediates. This technology represents a significant leap forward from traditional approaches, offering a universal pathway that accommodates various aryl iodides without the stringent electronic requirements of previous methods. By utilizing 1,3,4,6-O-tetrabenzyl-D-glucosamine hydrochloride as a key starting material, the process ensures high regioselectivity and yield, addressing long-standing challenges in glycosylamine chemistry. For R&D directors and procurement managers alike, this patent data signals a viable route for cost reduction in pharmaceutical intermediates manufacturing, providing a stable foundation for supply chain optimization and the reliable N-aryl-D-glucosamine supplier networks of the future.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical modification of D-glucosamine at the nitrogen position has been heavily reliant on nucleophilic substitution reactions that demand specific electronic environments to proceed effectively. Conventional techniques often require the presence of strong electron-withdrawing groups, such as nitro groups, on the aromatic ring to activate the substrate for reaction, which severely limits the structural diversity of the final products. This narrow substrate scope means that many potentially bioactive aryl variants cannot be synthesized using these older methods, forcing researchers to explore more expensive or hazardous alternatives. Furthermore, the harsh conditions often associated with these traditional substitutions can lead to degradation of the sensitive carbohydrate backbone, resulting in lower overall yields and complex impurity profiles that are difficult to manage during purification. These limitations create significant bottlenecks in the commercial scale-up of complex pharmaceutical intermediates, as the inability to diversify the aryl moiety restricts the potential applications of the final drug candidates.
The Novel Approach
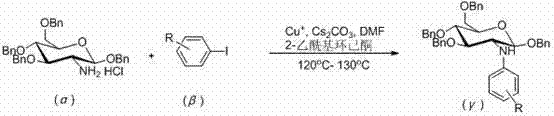
In stark contrast, the novel approach detailed in the patent data utilizes a copper-catalyzed system that dramatically expands the scope of compatible aryl iodides, allowing for the synthesis of a wide range of substituted N-aryl-D-glucosamine derivatives. This method operates under mild conditions, utilizing cesium carbonate as a base and 2-acetylcyclohexanone as a ligand to facilitate the cross-coupling reaction efficiently in DMF solvent. The versatility of this system means that electron-neutral and electron-rich aryl iodides can be employed successfully, overcoming the electronic constraints that plagued previous synthetic routes. By enabling the use of diverse substrates, this technology opens new avenues for medicinal chemistry exploration and process development, ensuring that high-purity N-aryl-D-glucosamine can be produced with greater flexibility. This shift not only enhances the feasibility of the工艺 structure but also provides a strategic advantage for reducing lead time for high-purity N-aryl-D-glucosamines in a competitive market.
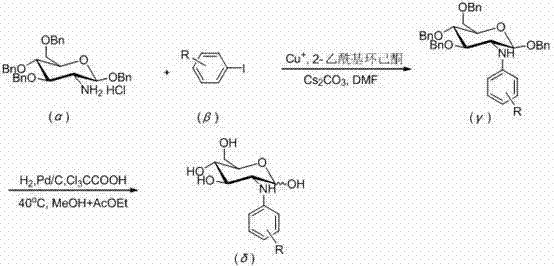
Mechanistic Insights into Copper-Catalyzed C-N Cross-Coupling
The core of this innovative synthesis lies in the mechanistic efficiency of the copper-catalyzed C-N bond formation, which proceeds through a well-defined catalytic cycle involving the activation of the aryl iodide and the glucosamine nucleophile. The use of cuprous salts, such as CuI, CuBr, or CuCl, in conjunction with the beta-diketone ligand 2-acetylcyclohexanone, creates a highly active catalytic species that facilitates the oxidative addition and reductive elimination steps necessary for coupling. This ligand system stabilizes the copper center, preventing aggregation and deactivation, which is crucial for maintaining high turnover numbers throughout the reaction duration of 8 to 20 hours at temperatures between 120°C and 130°C. The reaction environment is carefully controlled under a nitrogen atmosphere to prevent oxidation of the catalyst, ensuring consistent performance and reproducibility across different batches. For technical teams, understanding this mechanism is vital for troubleshooting and optimizing the process, as the interplay between the base, ligand, and solvent dictates the overall success of the transformation.
Following the coupling reaction, the process incorporates a strategic debenzylation step that is equally critical for achieving the final target molecule with the required purity specifications. The intermediate 1,3,4,6-O-tetrabenzyl-N-aryl-D-glucosamine is subjected to catalytic hydrogenation using palladium on carbon in the presence of trichloroacetic acid, which acts as an acid scavenger and promoter for the cleavage of the benzyl ether protecting groups. This step is conducted at a mild temperature of 40°C in a mixed solvent system of methanol and ethyl acetate, which ensures the solubility of the intermediate while maintaining the stability of the glycosidic bond. The careful control of hydrogen pressure and reaction time allows for the complete removal of benzyl groups without over-reduction of the aryl moiety, thereby preserving the structural integrity of the product. This two-stage mechanism, combining robust coupling with selective deprotection, exemplifies a refined approach to impurity control, minimizing the formation of side products that could complicate downstream processing.
How to Synthesize N-aryl-D-glucosamine Efficiently
Implementing this synthesis route requires precise adherence to the stoichiometric ratios and reaction conditions outlined in the patent to ensure optimal yield and purity. The process begins with the preparation of the key starting material, 1,3,4,6-O-tetrabenzyl-D-glucosamine hydrochloride, which must be synthesized with high fidelity to avoid introducing impurities early in the sequence. Once the starting material is secured, the coupling reaction is initiated by combining the glucosamine derivative with the chosen aryl iodide, cesium carbonate, and the copper catalyst system in dry DMF. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios and workup procedures necessary to isolate the intermediate effectively. Following the coupling, the debenzylation step must be monitored closely to ensure complete deprotection, followed by rigorous purification via silica gel chromatography to meet the stringent quality standards required for pharmaceutical applications.
- Perform C-N cross-coupling between 1,3,4,6-O-tetrabenzyl-D-glucosamine hydrochloride and aryl iodide using CuI, Cs2CO3, and 2-acetylcyclohexanone in DMF at 120-130°C.
- Execute debenzylation of the intermediate using H2 and Pd/C in the presence of trichloroacetic acid in a methanol and ethyl acetate solvent system at 40°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic methodology offers substantial benefits for procurement and supply chain teams looking to optimize costs and ensure continuity of supply for critical intermediates. The replacement of expensive noble metal catalysts with readily accessible copper salts significantly lowers the raw material costs associated with the production process, contributing to overall cost reduction in pharmaceutical intermediates manufacturing. Furthermore, the mild reaction conditions reduce the energy consumption required for heating and cooling, while the use of common solvents like DMF and ethyl acetate simplifies the logistics of solvent procurement and waste management. These factors combine to create a more resilient supply chain that is less vulnerable to fluctuations in the prices of specialized reagents or catalysts, enhancing the economic viability of large-scale production.
- Cost Reduction in Manufacturing: The utilization of copper catalysts instead of palladium or other precious metals represents a direct and significant saving in catalyst costs, which is a major component of the overall production budget. Additionally, the high efficiency of the reaction minimizes the loss of valuable starting materials, ensuring that the theoretical yield is closely approached in practice, which further drives down the cost per kilogram of the final product. The elimination of complex purification steps required to remove trace heavy metals also reduces the operational expenses associated with quality control and waste treatment. By streamlining the synthesis pathway, manufacturers can achieve a more competitive pricing structure without compromising on the quality or purity of the N-aryl-D-glucosamine supplied to clients.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable reagents such as aryl iodides and cesium carbonate ensures that the supply chain is not dependent on scarce or geopolitically sensitive materials. This availability reduces the risk of production delays caused by raw material shortages, allowing for more accurate forecasting and planning of inventory levels. The robustness of the reaction conditions also means that the process can be transferred between different manufacturing sites with minimal re-optimization, providing flexibility in sourcing and production capacity. For supply chain heads, this reliability translates into reduced lead time for high-purity N-aryl-D-glucosamines, enabling faster response to market demands and customer orders.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, as the reaction parameters are compatible with standard industrial reactor setups and do not require exotic equipment or extreme pressures. The use of hydrogenation for debenzylation is a well-established unit operation in the fine chemical industry, facilitating easy scale-up from laboratory to commercial production volumes. Moreover, the avoidance of highly toxic reagents and the generation of manageable waste streams align with modern environmental compliance standards, reducing the regulatory burden on the manufacturer. This alignment with green chemistry principles not only mitigates environmental risk but also enhances the corporate sustainability profile of the production facility.
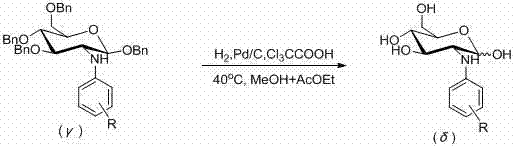
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic route, based on the detailed data provided in the patent documentation. These answers are derived from the specific reaction conditions and outcomes reported, offering clarity on the feasibility and advantages of the technology for potential partners. Understanding these details is essential for making informed decisions about process adoption and supply chain integration, ensuring that all stakeholders have a clear view of the capabilities and limitations of the method.
Q: What are the advantages of the copper-catalyzed method over traditional nucleophilic substitution?
A: The copper-catalyzed method eliminates the need for strong electron-withdrawing groups on the aryl ring, allowing for a much broader substrate scope and milder reaction conditions compared to traditional methods.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the use of inexpensive copper catalysts and readily available reagents like cesium carbonate makes this route highly scalable and cost-effective for industrial manufacturing.
Q: How is the final purity of N-aryl-D-glucosamine ensured in this process?
A: The process includes a rigorous purification step using silica gel chromatography after the coupling reaction and a controlled debenzylation step that minimizes side reactions, ensuring high purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-aryl-D-glucosamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this copper-catalyzed synthesis route for the production of high-value carbohydrate intermediates. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can transition smoothly from development to full-scale manufacturing. Our facilities are equipped with stringent purity specifications and rigorous QC labs to guarantee that every batch of N-aryl-D-glucosamine meets the highest industry standards. We are committed to leveraging this advanced technology to deliver consistent quality and reliability, supporting your R&D and commercial goals with our deep technical expertise and robust infrastructure.
We invite you to collaborate with us to optimize your supply chain and reduce costs through the adoption of this efficient synthetic method. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our capabilities can enhance your production efficiency. By partnering with us, you gain access to a reliable N-aryl-D-glucosamine supplier dedicated to driving innovation and value in the pharmaceutical intermediates sector.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
